ELECTRONS IN ATOMS 2 ELECTRON ORBITALS Cartoon courtesy

ELECTRONS IN ATOMS #2
ELECTRON ORBITALS Cartoon courtesy of lab-initio. com

THE BOHR MODEL OF THE ATOM I pictured electrons orbiting the nucleus much like planets orbiting the sun. But I was wrong! They’re more like bees around a hive. Neils Bohr

THE BOHR MODEL • Bohr proposed that an electron is found only in specific circular paths, or orbits, around the nucleus. • Each possible e- orbit has a fixed energy. • The fixed energies are called energy levels. • To move from one energy level to another, an e- must gain or lose just the right amount of energy. • Higher energy levels are generally farther from the nucleus.

• Quantum = the amount of energy required to move an electron from one energy level to another energy level. (Max Plank) • Bohr’s model worked great to explain Hydrogen, but failed to explain the energies absorbed and emitted by the other atoms

QUANTUM MECHANICAL MODEL OF THE ATOM Mathematical laws can identify the regions outside of the nucleus where electrons are most likely to be found. These laws are beyond the scope of this class…

THE QUANTUM MECHANICAL MODEL • LOUIS DE BROGLIE PROPOSED THAT MOVING PARTICLES LIKE ELECTRONS HAVE SOME PROPERTIES OF WAVES. • SCHRODINGER (AUSTRIAN PHYSICIST) USED A MATHEMATICAL EQUATION TO DESCRIBE THE MOTIONS OF ELECTRONS. • QUANTUM MECHANICAL MODEL = THE MODERN DESCRIPTION OF THE E- IN ATOMS, BASED ON SCHRODINGER’S WORK.

• Heisenberg Uncertainty Principle: The position and momentum of a particle cannot be simultaneously measured. • Like the Bohr model, the quantum model predicts quantized energy levels for electrons, however it does not describe the exact path the electron takes around the nucleus. • It is concerned with the probability, or likelihood, of finding an electron in a certain position.

HEISENBERG UNCERTAINTY PRINCIPLE - THIS HEISENBERG “One cannot simultaneously determine both the position and momentum of an electron. ” Werner Heisenberg You can find out where the electron is, but not where it is going. OR… You can find out where the electron is going, but not where it is!

The main difference between Bohr’s model and the quantum mechanical model is the electron orbitals. Bohr’s orbitals were circular and fixed. In the quantum mechanical model the electron orbital are regions in space where the electron is likely to be found. The quantum mechanical model is often called the “electron cloud model” (fuzzy boundaries of e- orbits)

QUANTUM NUMBERS • • A QUANTUM NUMBER IS USED TO DEFINE THE ENERGY AND LOCATION OF AN ELECTRON IN THE QUANTUM MECHANICAL ATOM. A TOTAL OF FOUR QUANTUM NUMBERS ARE NECESSARY TO COMPLETELY DEFINE THE ENERGY AND LOCATION OF AN ELECTRON. THE FIRST THREE NUMBERS GIVE THE LOCATION OF THE ELECTRON. THE FOURTH QUANTUM NUMBER DESCRIBES THE SPIN OF AN ELECTRON IN AN ORBITAL.

9 -22 -15 • GET OUT YOUR NOTES FROM FRIDAY ABOUT QUANTUM NUMBERS • GET READY TO BOOGIE • ****POLY ATOMIC IONS****

FIRST QUANTUM NUMBER � Principal quantum number � n = 1, 2, 3, 4, (positive integer starting with 1) � Describes the energy level that the electron occupies. � Basically tells you how far the electron is from the nucleus. n =3 n =2 n =1 � Generally, the larger the value on n, the farther away from the nucleus and the higher the energy of the electron. **Number of e- that can fit in a shell: 2 n 2

Electron Orbitals AN ORBITAL IS A REGION WITHIN AN ENERGY LEVEL WHERE THERE IS A PROBABILITY OF FINDING AN ELECTRON. Orbital shapes are defined as the surface that contains 90% of the total electron probability.

SECOND QUANTUM NUMBER • • • Describes the shapes of atomic orbitals. l = 0, 1, 2 , 3 s, p, d, f Orbital Shapes s = sphere p= dumbbell d & f = complex Each principal energy level can have a maximum of one s orbital (2 e-), three p orbitals (6 e-), five d orbitals (10 e-), and seven f orbitals (14 e-). Within each principal energy level, the group of same shape/energy orbitals is a sublevel. • Sublevels one s three p five d seven f Orbital orbitals

S ORBITAL SHAPE The s orbital has a spherical shape centered around the origin of the three axes in space.
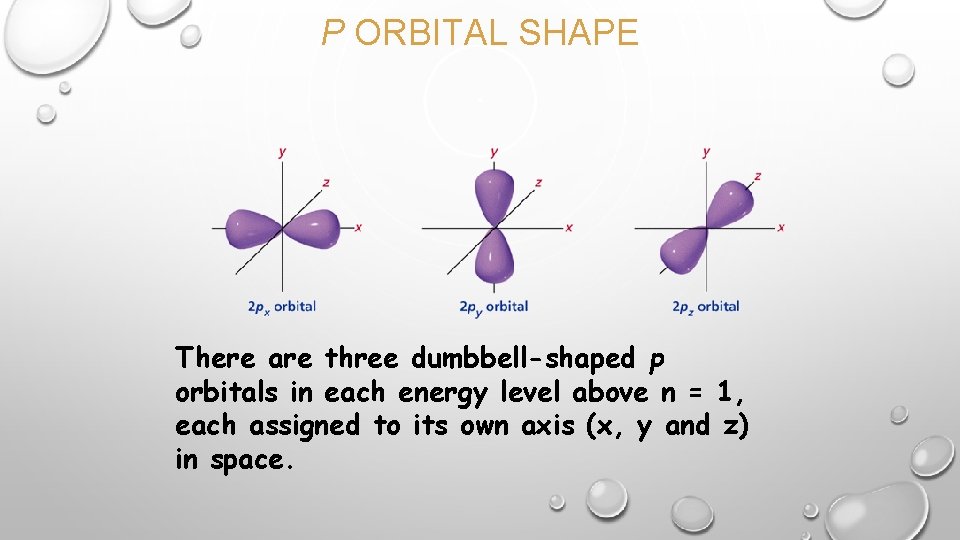
P ORBITAL SHAPE There are three dumbbell-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.
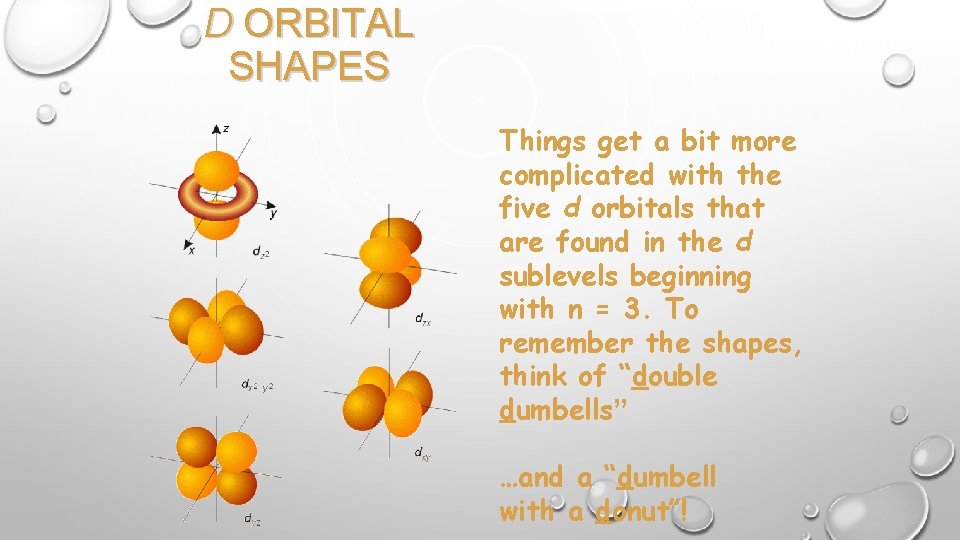
D ORBITAL SHAPES Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbells” …and a “dumbell with a donut”!
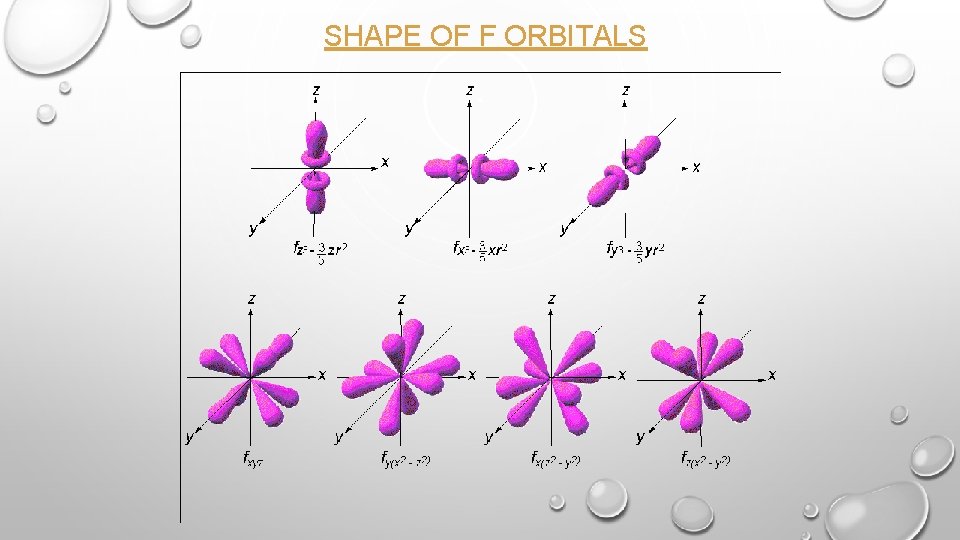
SHAPE OF F ORBITALS
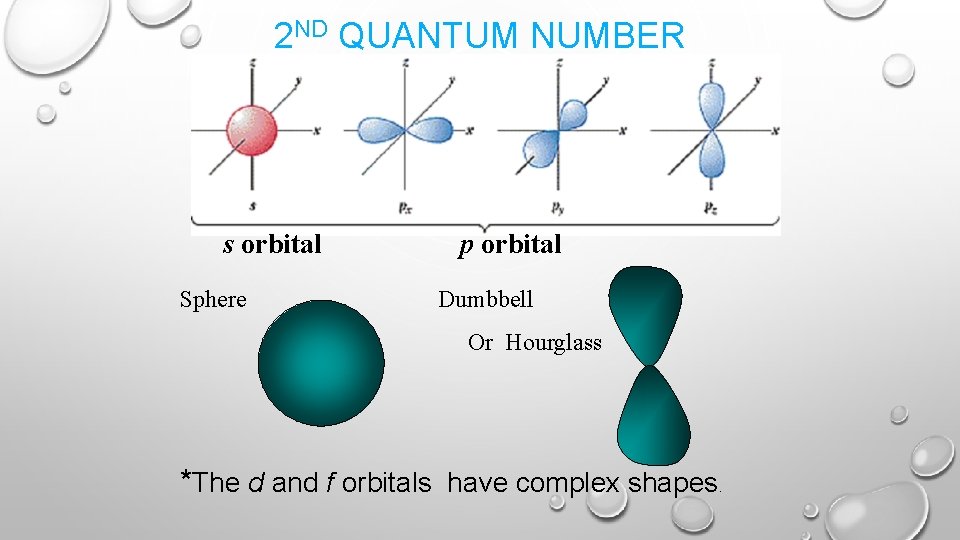
2 ND QUANTUM NUMBER s orbital Sphere p orbital Dumbbell Or Hourglass *The d and f orbitals have complex shapes.

THIRD QUANTUM NUMBER Magnetic quantum number (ml) Tells you the electron position by designating the orientation in space of the orbital that is occupies. Ex: p orbital can have three spatial orientations. (x, y, z

FOURTH QUANTUM NUMBER • Spin quantum number(ms ) • Labels the orientation of the electrons within the orbital (the direction of spin) • Clockwise & Counter clockwise. • No two e- have an identical set of four quantum numbers.
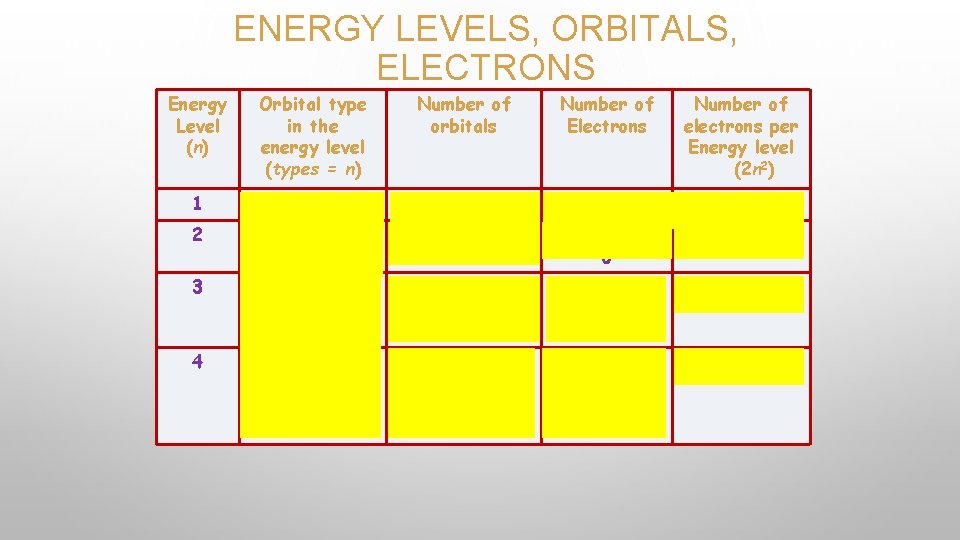
ENERGY LEVELS, ORBITALS, ELECTRONS Energy Level (n) Orbital type in the energy level (types = n) Number of orbitals Number of Electrons Number of electrons per Energy level (2 n 2) 1 s 1 2 2 2 s p 1 3 2 6 8 3 s p d 1 3 5 2 6 10 18 4 s p d f 1 3 5 7 2 6 10 14 32

REMEMBER…. • THE PRINCIPAL QUANTUM NUMBER ALWAYS EQUALS THE NUMBER OF SUBLEVELS WITHIN THAT PRINCIPAL ENERGY LEVEL. 1 1 s 2 3 2 s & 2 p 3 s, 3 p, & 3 d 4 4 s, 4 p, 4 d, 4 f

ELECTRON ARRANGEMENT IN ATOMS • IN THE ATOM, ELECTRONS AND THE NUCLEUS INTERACT TO MAKE THE MOST STABLE ARRANGEMENT POSSIBLE. • THE WAYS THAT ELECTRONS ARE ARRANGED INTO VARIOUS ORBITALS AROUND THE NUCLEUS ARE CALLED ELECTRON CONFIGURATIONS.

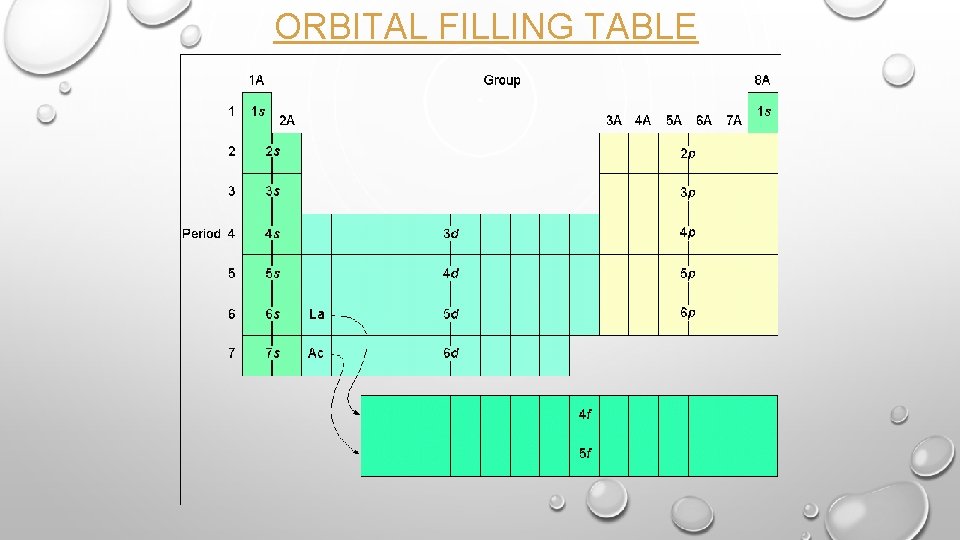
THE AUFBAU PRINCIPLE • Electrons occupy orbitals of lowest energy first. • Within each principal energy level: Lowest-energy s<p<d<f highest-energy • The range of one principal energy level can overlap another level, so the electrons fill the orbitals in the following order, according to energy: 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s 4 d 5 p 6 s 4 f 5 d 6 p 7 s 5 f 6 d 7 p

PAULI EXCLUSION PRINCIPLE Two electrons occupying the same orbital must have opposite spins Wolfgang Pauli

PAULI EXCLUSION PRINCIPLE AN ATOMIC ORBITAL MAY DESCRIBE, AT MOST, TWO ELECTRONS. IN ORDER TO OCCUPY THE SAME ORBITAL, TWO ELECTRONS MUST HAVE OPPOSITE SPINS. • Hund’s rule: When electrons occupy orbitals of equal energy (the same sublevel), one electron enters each orbital until all the orbitals contain one electron with the same spin direction. • Electrons spread out as much as they can with one electron in each orbital, until all of the orbitals have one electron. Second electrons then occupy each orbital so that their spins are paired (opposite) with the first electron in the orbital.

WARM UP 4 -14 -15 • NAME (AND BRIEFLY DESCRIBE THE) 3 PRINCIPLES/RULES FOR WRITING ELECTRON CONFIGURATION) – USE NOTES NOT PEOPLE) – IT IN UP FRONT Do NOT USETURN MY PAPER FOR THE WARM UP Get a periodic table from the front (and 2 sheets of paper) and get out 4 colored markers, or highlighters etc.

THE AUFBAU PRINCIPLE • Electrons occupy orbitals of lowest energy first. • Within each principal energy level: Lowest-energy s<p<d<f highest-energy • The range of one principal energy level can overlap another level, so the electrons fill the orbitals in the following order, according to energy: 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s 4 d 5 p 6 s 4 f 5 d 6 p 7 s 5 f 6 d 7 p

PAULI EXCLUSION PRINCIPLE AN ATOMIC ORBITAL MAY DESCRIBE, AT MOST, TWO ELECTRONS. IN ORDER TO OCCUPY THE SAME ORBITAL, TWO ELECTRONS MUST HAVE OPPOSITE SPINS. • Hund’s rule: When electrons occupy orbitals of equal energy (the same sublevel), one electron enters each orbital until all the orbitals contain one electron with the same spin direction. • Electrons spread out as much as they can with one electron in each orbital, until all of the orbitals have one electron. Second electrons then occupy each orbital so that their spins are paired (opposite) with the first electron in the orbital.

HOW TO WRITE ELECTRON CONFIGURATIONS • Determine how many electrons are in the atom? What is the atomic number? • Ex: Ga has 31 e- because its atomic # is 31. O has 8 e- because its atomic # is 8. Is it an ion? If so, subtract the charge from the atomic number. • Ex: Mg 2+ has 12 – 2 = 10 e- ; O 2 - has 8 - ( -2) = 10 e. Fill orbitals in the proper order w/ e-. • Use the Aufbau diagram at first. 2 electrons can fit in each box = orbital. • Ex: Chlorine has 17 e- so its configuration is • 1 s 2 2 p 6 3 s 2 3 p 5 • Check that the total # of e- (superscripts) in the e- configuration equals the # of e- in the element.

7 s 6 p 6 s 5 p 5 s 4 p 4 s 3 p 3 s 2 p 2 s 1 s 6 d 5 f 5 d 4 f 4 d 3 d
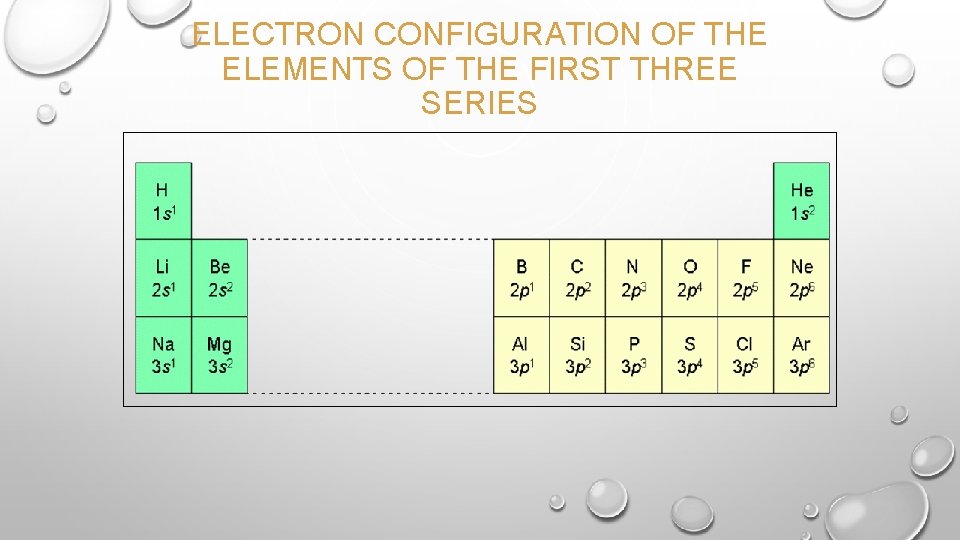
ELECTRON CONFIGURATION OF THE ELEMENTS OF THE FIRST THREE SERIES

E- CONFIGURATION W/ THE PERIODIC TABLE • FOR S & P ; COEFFICIENT = PERIOD # • FOR D ; COEFFICIENT = PERIOD -1 • FOR F ; COEFFICIENT = PERIOD – 2 • COUNT EACH ELEMENT IN THE SECTION TO DETERMINE THE EXPONENT
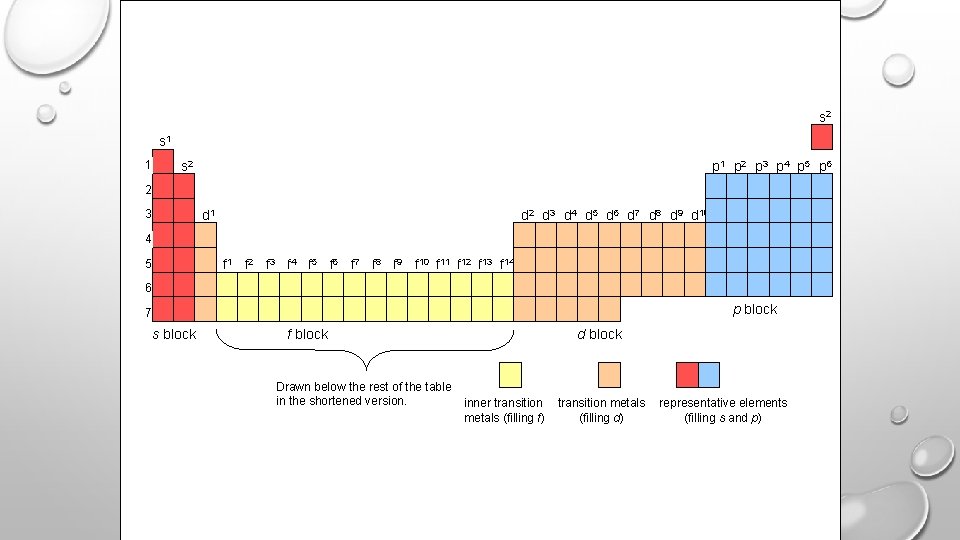
s 2 s 1 1 s 2 p 1 p 2 p 3 p 4 p 5 p 6 2 3 d 1 d 2 d 3 d 4 d 5 d 6 d 7 d 8 d 9 d 10 4 5 f 1 f 2 f 3 f 4 f 5 f 6 f 7 f 8 f 9 f 10 f 11 f 12 f 13 f 14 6 p block 7 s block f block Drawn below the rest of the table in the shortened version. d block inner transition metals (filling f) transition metals (filling d) representative elements (filling s and p)
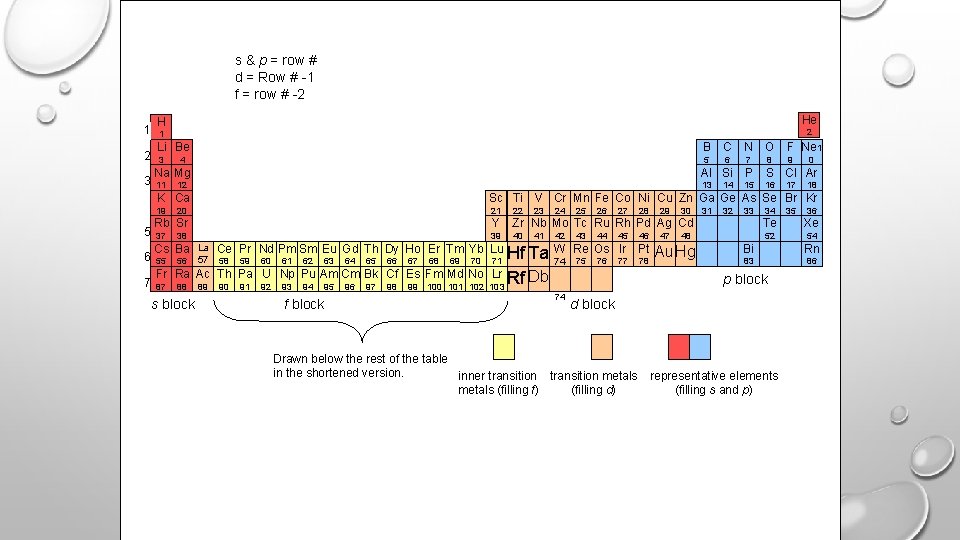
s & p = row # d = Row # -1 f = row # -2 1 2 3 5 6 7 He H 2 1 Li Be B C N O F Ne 1 3 5 6 7 9 4 8 0 Na Mg Al Si P S Cl Ar 11 13 16 12 14 15 17 18 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 19 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd Te Xe 39 40 52 54 37 38 Cs Ba 55 56 La 57 88 s block 89 41 24 42 43 26 44 27 45 28 46 Re Os Ir Pt 58 75 59 60 61 62 63 64 65 66 67 68 69 70 90 91 92 93 94 95 f block 96 97 98 99 100 101 102 Hf Ta W 74 Lr Rf Db 103 25 Ce Pr Nd Pm Sm Eu Gd Th Dy Ho Er Tm Yb Lu Fr Ra Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No 87 23 71 74 76 77 78 29 47 30 31 32 33 48 Au Hg 34 36 Bi Rn 83 86 p block d block Drawn below the rest of the table in the shortened version. inner transition metals (filling f) (filling d) 35 representative elements (filling s and p)
ORBITAL FILLING TABLE

ELECTRON ENERGY LEVEL DIAGRAM • BOX & ARROW NOTATION = USE LABELED BOXES TO REPRESENT EACH ORBITAL AND AN ARROW TO REPRESENT EACH ELECTRON. • BE SURE TO FOLLOW HUND’S RULE • EX: PHOSPHORUS
![Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1](http://slidetodoc.com/presentation_image_h2/5d337462895213b0fd09bcc19ae43786/image-40.jpg)
Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 ____ 1 s Beryllium ____ 2 p ____ 2 s ____ 2 p ____ [He]2 s 2 p 2 ____ 2 s ____ 2 p ____ 1 s 22 p 3 [He]2 s 2 p 3 ____ 2 s ____ 2 p ____ 1 s 22 p 4 [He]2 s 2 p 4 ____ 2 s ____ 2 p ____ 1 s 22 p 5 [He]2 s 2 p 5 ____ 1 s Neon ____ 2 s 1 s 22 p 2 ____ 1 s Fluorine ____ [He]2 s 2 p 1 ____ 1 s Oxygen ____ 2 p 1 s 22 p 1 ____ 1 s Nitrogen ____ [He]2 s 2 ____ 1 s Carbon ____ 2 s 1 s 22 s 2 ____ 1 s Boron Noble gas notation ____ 2 s ____ 2 p ____ 1 s 22 p 6 [He]2 s 2 p 6 ____ 1 s ____ 2 s ____ 2 p ____

VALENCE ELECTRONS • VALENCE E- ARE THE OUTERMOST ELECTRONS-ELECTRONS WITH THE HIGHEST PRINCIPAL QUANTUM NUMBER. • VALENCE E- WILL ONLY INCLUDE S & P ORBITALS, BECAUSE THEY WILL ALWAYS HAVE THE HIGHEST PRINCIPAL QUANTUM NUMBER.
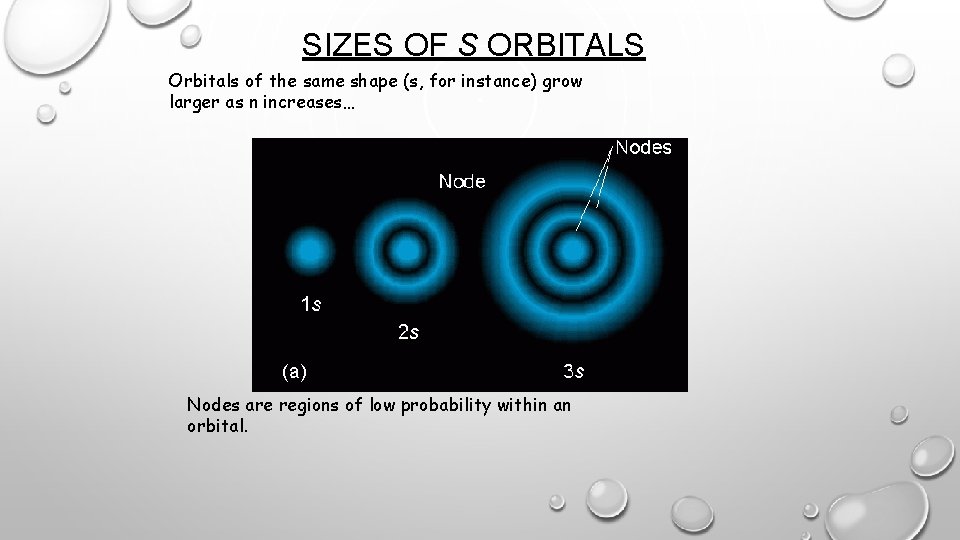
SIZES OF S ORBITALS Orbitals of the same shape (s, for instance) grow larger as n increases… Nodes are regions of low probability within an orbital.
- Slides: 42