Electron Configurations Electrons in an atom Electrons in
































- Slides: 50

Electron Configurations

Electrons in an atom

Electrons in an atom • Energy level - the ring of electrons an electron is found in.

Electrons in an atom • Energy level - the ring of electrons an electron is found in. • Sublevel – the specific shaped cloud that the electron is found in.

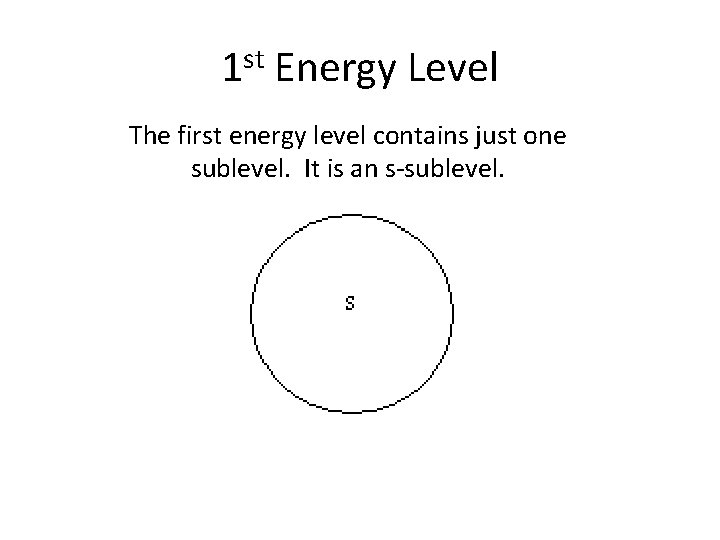
1 st Energy Level The first energy level contains just one sublevel. It is an s-sublevel.

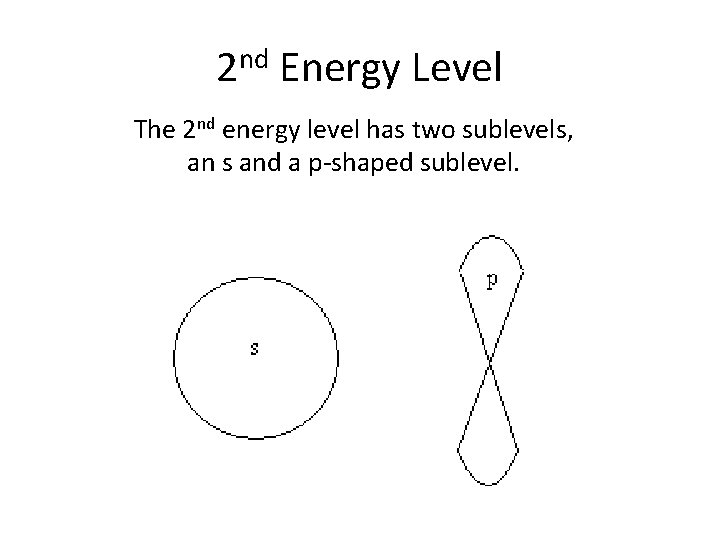
2 nd Energy Level The 2 nd energy level has two sublevels, an s and a p-shaped sublevel.

3 rd Energy Level • The 3 rd energy level contains 3 sublevels.

3 rd Energy Level • The 3 rd energy level contains 3 sublevels. • s, p, and d (the shape of the d-sublevel is more complicated)

Energy Levels 4 -7 • The 4 th-7 th energy levels contains 4 sublevels each.

Energy Levels 4 -7 • The 4 th-7 th energy levels contains 4 sublevels each. • s, p, d, and f (the shape of the f-sublevel is also very complicated)

Electrons in an atom • Energy level - the ring of electrons an electron is found in. • Sublevel – the specific shaped cloud that the electron is found in. • Orbital - a region of the sublevel that can be occupied by no more than 2 electrons.

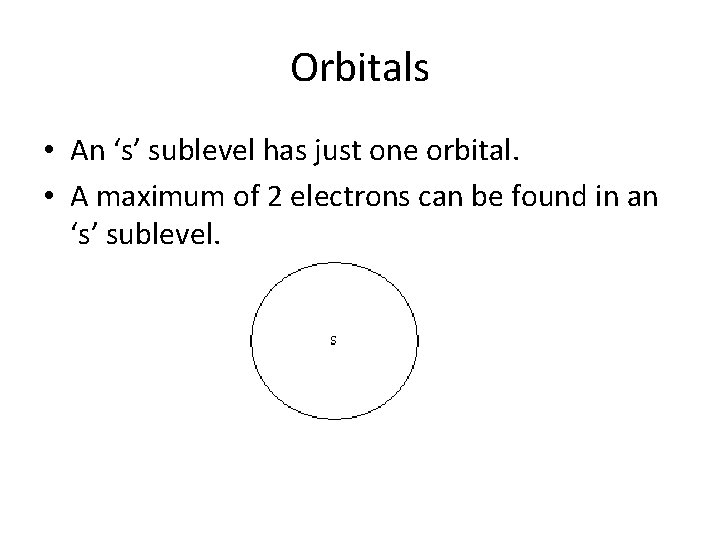
Orbitals • An ‘s’ sublevel has just one orbital.

Orbitals • An ‘s’ sublevel has just one orbital. • A maximum of 2 electrons can be found in an ‘s’ sublevel.

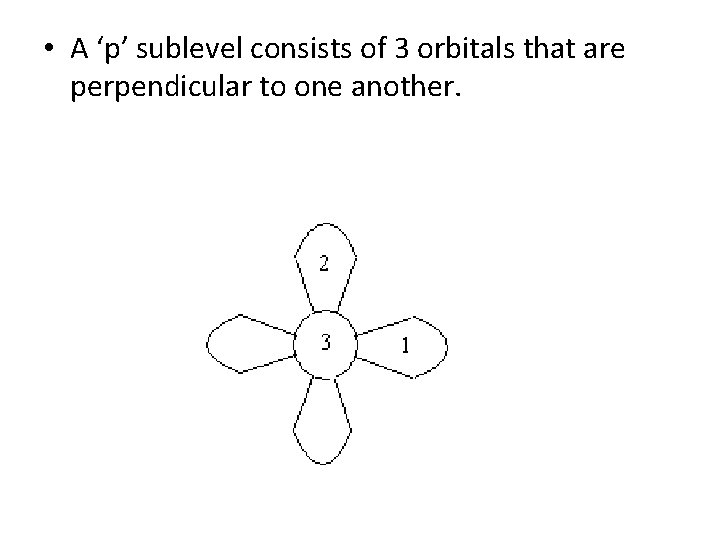
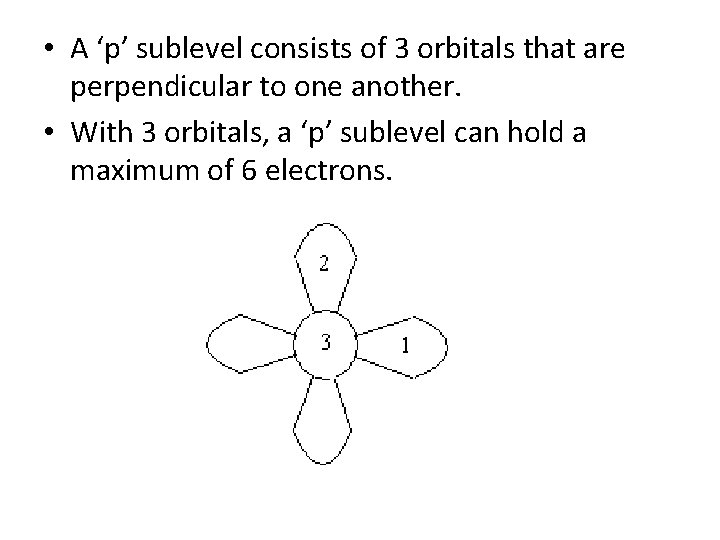
• A ‘p’ sublevel consists of 3 orbitals that are perpendicular to one another.

• A ‘p’ sublevel consists of 3 orbitals that are perpendicular to one another. • With 3 orbitals, a ‘p’ sublevel can hold a maximum of 6 electrons.

• A ‘d’ sublevel contains a total of 5 orbitals.

• A ‘d’ sublevel contains a total of 5 orbitals. • The maximum number of electrons in a ‘d’ sublevel is 10.

• An ‘f’ sublevel contains a total of 7 orbitals.

• An ‘f’ sublevel contains a total of 7 orbitals. • The maximum number of electrons in a ‘f’ sublevel is 14.

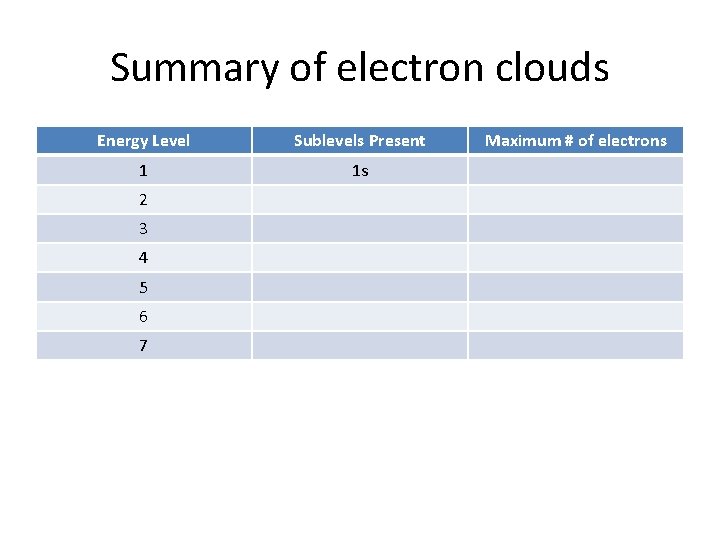
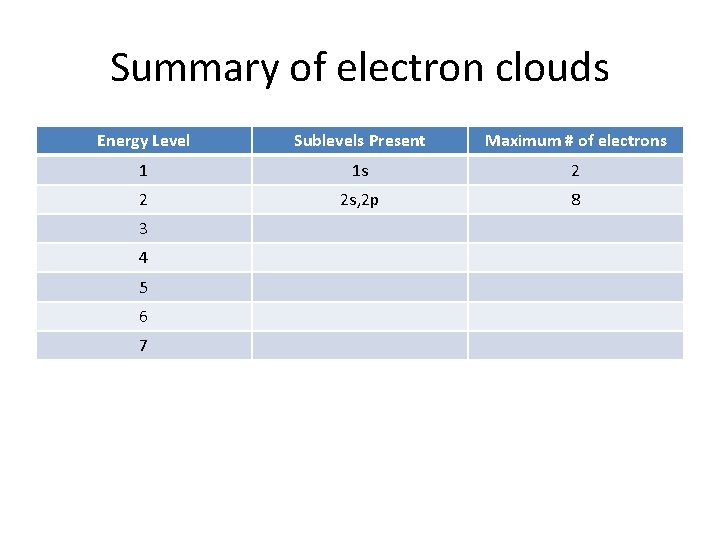
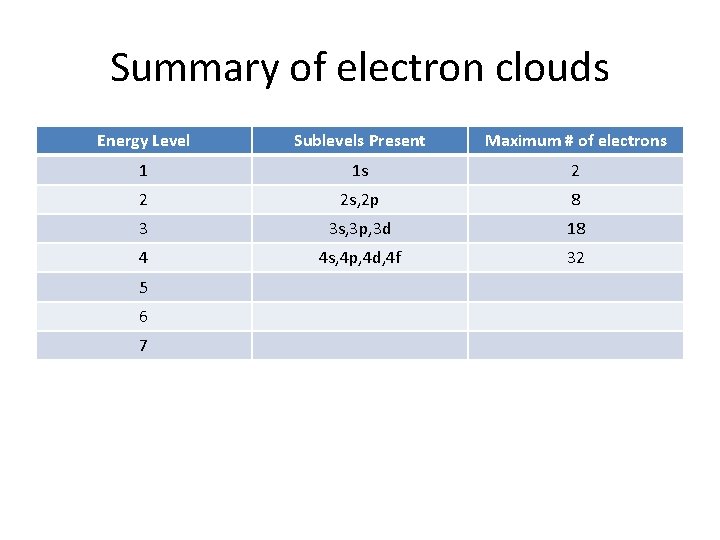
Summary of electron clouds Energy Level Sublevels Present 1 1 s 2 3 4 5 6 7 Maximum # of electrons

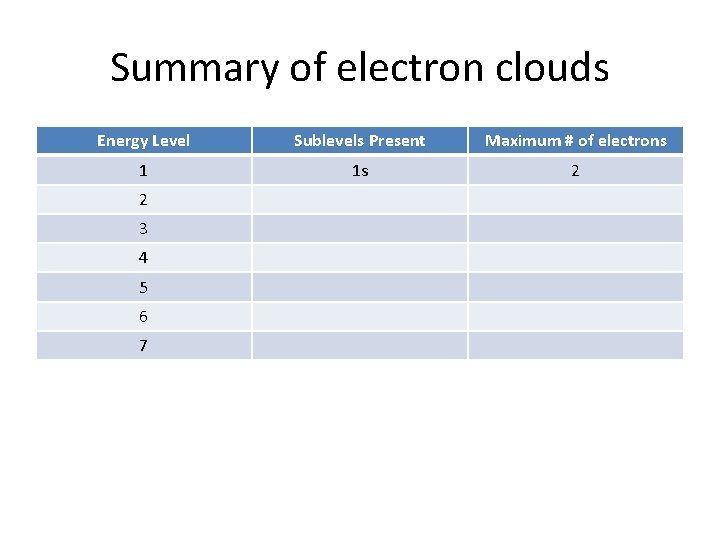
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 3 4 5 6 7

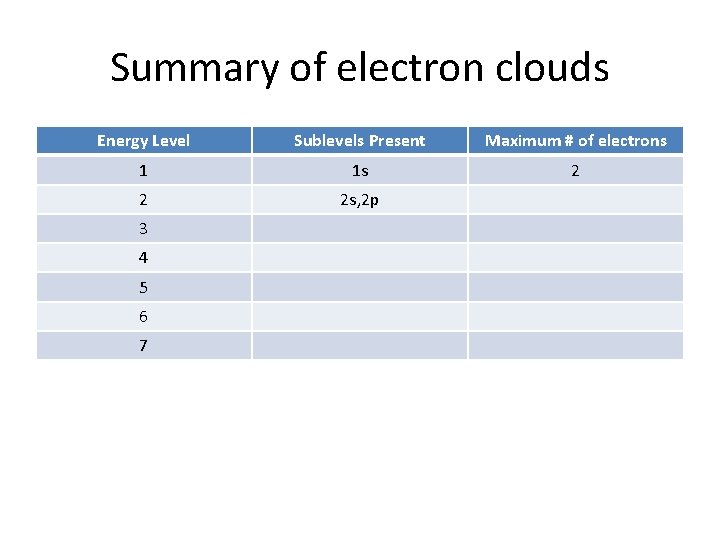
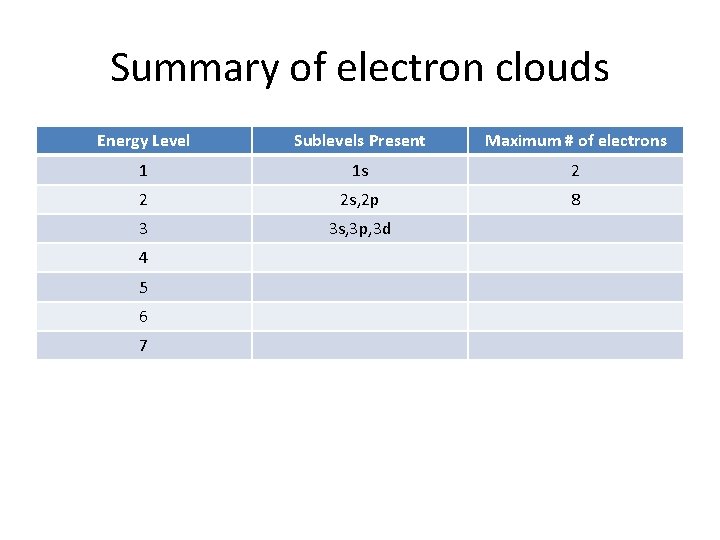
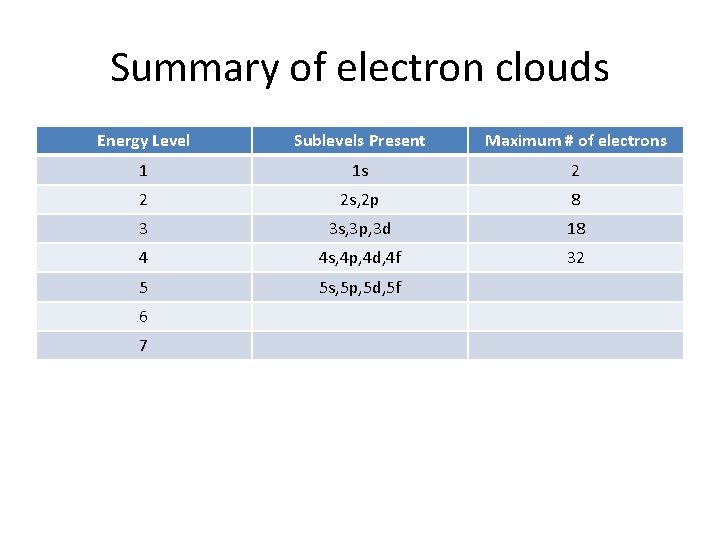
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 3 4 5 6 7

Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 4 5 6 7

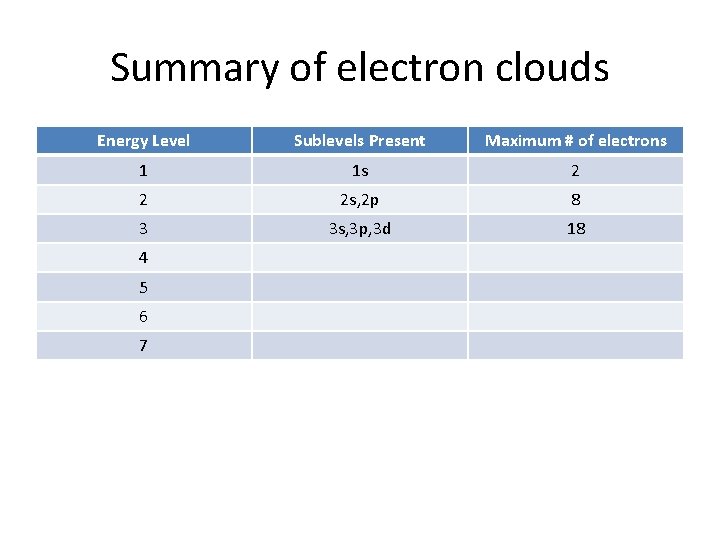
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 4 5 6 7

Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 5 6 7

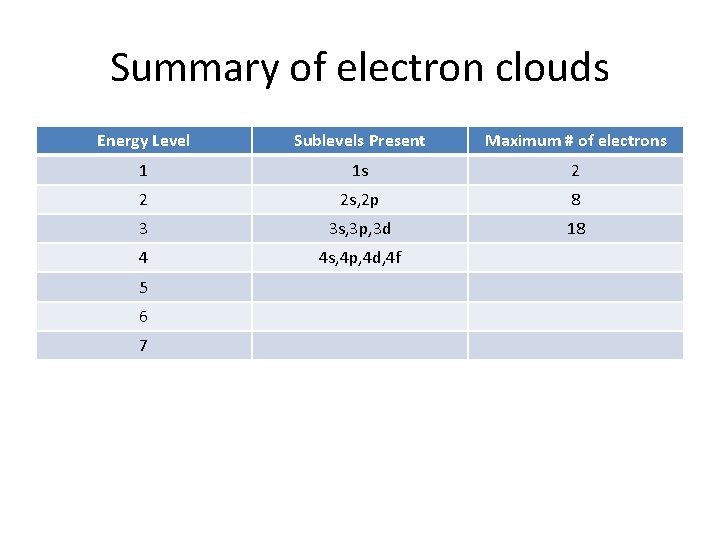
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 4 s, 4 p, 4 d, 4 f 5 6 7

Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 4 s, 4 p, 4 d, 4 f 32 5 6 7

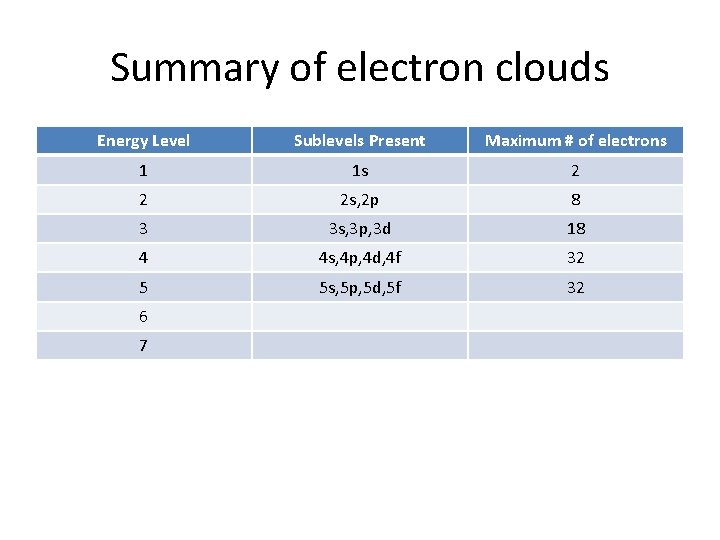
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 4 s, 4 p, 4 d, 4 f 32 5 5 s, 5 p, 5 d, 5 f 6 7

Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 4 s, 4 p, 4 d, 4 f 32 5 5 s, 5 p, 5 d, 5 f 32 6 7

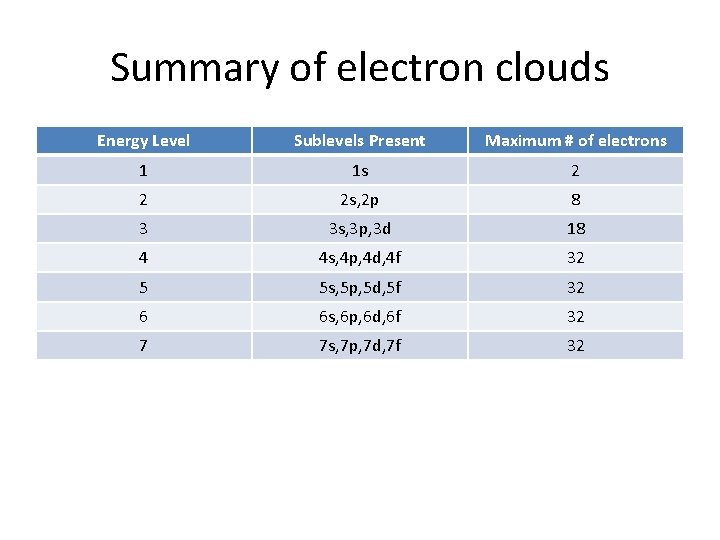
Summary of electron clouds Energy Level Sublevels Present Maximum # of electrons 1 1 s 2 2 2 s, 2 p 8 3 3 s, 3 p, 3 d 18 4 4 s, 4 p, 4 d, 4 f 32 5 5 s, 5 p, 5 d, 5 f 32 6 6 s, 6 p, 6 d, 6 f 32 7 7 s, 7 p, 7 d, 7 f 32

Diagonal Rule • The diagonal rule allows you to determine the order in which sublevels will be filled with electrons.

Diagonal Rule • The diagonal rule allows you to determine the order in which sublevels will be filled with electrons. • Begin by writing out all of the possible sublevels in 4 columns, one for each letter.


Diagonal Rule • The diagonal rule allows you to determine the order in which sublevels will be filled with electrons. • Begin by writing out all of the possible sublevels in 4 columns, one for each letter. • Starting at the top, draw parallel diagonal lines so that each sublevel is crossed.


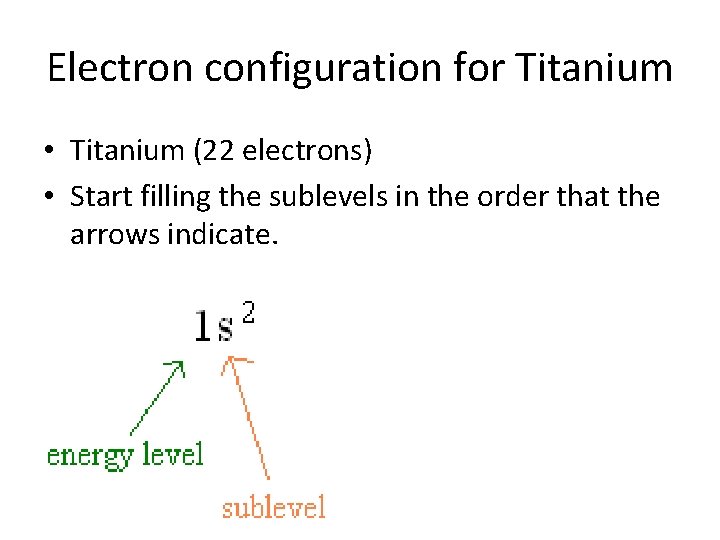
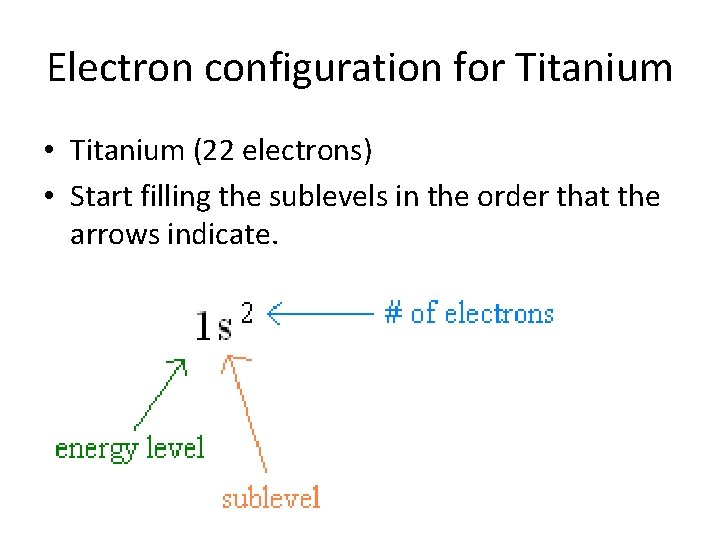
Electron configuration for Titanium • Titanium (22 electrons)

Electron configuration for Titanium • Titanium (22 electrons) • Start filling the sublevels in the order that the arrows indicate.

Electron configuration for Titanium • Titanium (22 electrons) • Start filling the sublevels in the order that the arrows indicate.

Electron configuration for Titanium • Titanium (22 electrons) • Start filling the sublevels in the order that the arrows indicate.

Electron configuration for Titanium • Titanium (22 electrons) • Start filling the sublevels in the order that the arrows indicate.

Electron configuration for Titanium • Titanium (22 electrons) • Start filling the sublevels in the order that the arrows indicate.

• Continue until all 22 electrons are accounted for.

• Continue until all 22 electrons are accounted for. • 1 s 22 p 63 s 23 p 64 s 23 d 2

• Continue until all 22 electrons are accounted for. • 1 s 22 p 63 s 23 p 64 s 23 d 2 • Even though the d sublevel can hold 10 electrons, we only had 2 electrons left.

• Continue until all 22 electrons are accounted for. • 1 s 22 p 63 s 23 p 64 s 23 d 2 • Even though the d sublevel can hold 10 electrons, we only had 2 electrons left. • Being in period 4, the 4 th energy level is the valence level for titanium.

• Continue until all 22 electrons are accounted for. • 1 s 22 p 63 s 23 p 64 s 23 d 2 • Even though the d sublevel can hold 10 electrons, we only had 2 electrons left. • Being in period 4, the 4 th energy level is the valence level for titanium. • The two electrons in the 4 s sublevel are valence electrons.

Checking for accuracy • 1 s 22 p 63 s 23 p 64 s 23 d 2

Checking for accuracy • 1 s 22 p 63 s 23 p 64 s 23 d 2 • The d in the last term means we should find titanium in the d-block of the periodic table (groups 3 -12).

Checking for accuracy • 1 s 22 p 63 s 23 p 64 s 23 d 2 • The d in the last term means we should find titanium in the d-block of the periodic table (groups 3 -12). • The exponent 2 indicates that it should be in the 2 nd column of the d-block (group 4).

Checking for accuracy • 1 s 22 p 63 s 23 p 64 s 23 d 2 • The d in the last term means we should find titanium in the d-block of the periodic table (groups 3 -12). • The exponent 2 indicates that it should be in the 2 nd column of the d-block (group 4). • The energy level (3) indicates the period number for the element if it is in the s or p block. The d block is always one period behind and the f block is 2 behind. Titanium should be in period 4.