Electromagnetic Radiation and XRays Its of no use
![Electromagnetic Radiation and X-Rays "It's of no use whatsoever[. . . ] this is Electromagnetic Radiation and X-Rays "It's of no use whatsoever[. . . ] this is](https://slidetodoc.com/presentation_image_h2/1d5aaa7ac321327a9bba9a020d0ec30f/image-1.jpg)



















- Slides: 23
![Electromagnetic Radiation and XRays Its of no use whatsoever this is Electromagnetic Radiation and X-Rays "It's of no use whatsoever[. . . ] this is](https://slidetodoc.com/presentation_image_h2/1d5aaa7ac321327a9bba9a020d0ec30f/image-1.jpg)
Electromagnetic Radiation and X-Rays "It's of no use whatsoever[. . . ] this is just an experiment that proves Maestro Maxwell was right - we just have these mysterious electromagnetic waves that we cannot see with the naked eye. But they are there. " Heinrich Hertz 1

Spectroscopy and X-Ray Analysis n Electromagnetic Radiation ¨ Electromagnetic waves ¨ Calculations involving waves ¨ The electromagnetic spectrum ¨ Light and Optics ¨ Refraction and diffraction n X-Rays ¨ Discovery of X-rays ¨ Generation of X-rays ¨ Quantum Numbers ¨ Electron Energy Transitions 2

The Electromagnetic Waves Insert electromagnetic wave image here Light waves are self propagating waves that consist of both an electronic and magnetic component. 3

Formulas for Waves Propagation Speed c = λf Energy E = hf c is speed of propagation, (m/s) Where: E is the energy of the photon λ is wavelength, (m) h is Planck’s constant f is frequency (/s, Hz, s-1) f is the frequency of the radiation Period T = 1/f f = 1/T Where: T is the period (s) f is the frequency (Hz) For light c is constant and equal to 2. 998 x 108 m/s 4

The Electromagnetic Spectrum Insert electromagnetic spectrum picture here 5

EM Radiation Activity n You will each be assigned one of the following types of electromagnetic radiation. Look it up. Report the following information for it: ¨ Wavelength ¨ How it is generated ¨ What it are some common uses n Gamma rays, X-rays, Ultraviolet radiation, Light, Infra-red radiation, Microwaves, Radio waves (FM, AM, ELF), Gravity waves. 6

Calculations Calculate the frequency of a red laser pointer light with wavelength 655 nm. 7

Calculations Calculate the wavelength and type of electromagnetic radiation you would expect to produce from a 3 GHz computer. 8

Calculations A common unit in spectroscopy is the “wave number” which is usually defined as the number of waves per cm. How many wave cycles per cm (wave numbers) would you expect to find in radiation produced from a microwave oven operating at a frequency of 2450 MHz? 9

Calculations Copper emits a kα X-ray of 8. 04 ke. V. What would the wavelength be? 10

Light and Optics n Electromagnetic radiation ¨ What we see as light is part of the electromagnetic spectrum. ¨ Photon: a unit of electromagnetic energy (light). Photons have no electric charge, they have zero “rest mass” but they do have momentum and energy. ¨ http: //hyperphysics. phy-astr. gsu. edu/hbase/emwav. html#c 1 http: //en. wikipedia. org/wiki/Electromagnetic_radiation 11

Discovery of X-rays 1895 Insert Wilhelm Roentgen image here Insert image of the first X-ray here Wilhelm Röntgen http: //en. wikipedia. org/wiki/X-ray 12

X-ray Tube Insert X-ray tube image here 13

Two methods for generating X-rays Bremsstrahlung / Braking Insert image Ionization / Characteristic Insert image http: //www. antonine-education. co. uk/Physics_A 2/Options/Module_6/Topic_7/topic_7_x. htm 14

X-Ray Analysis n n n Quantum numbers Electron Shells Allowed electron transitions Insert image http: //www 4. nau. edu/microanalysis/Microprobe/Probe. html 15

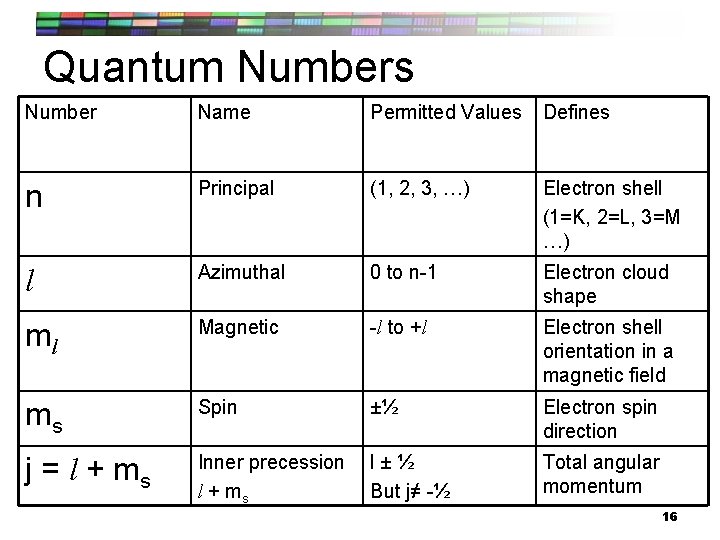
Quantum Numbers Number Name Permitted Values Defines n Principal (1, 2, 3, …) Electron shell (1=K, 2=L, 3=M …) l Azimuthal 0 to n-1 Electron cloud shape ml Magnetic -l to +l Electron shell orientation in a magnetic field ms Spin ±½ Electron spin direction j = l + ms Inner precession l + ms l±½ But j≠ -½ Total angular momentum 16

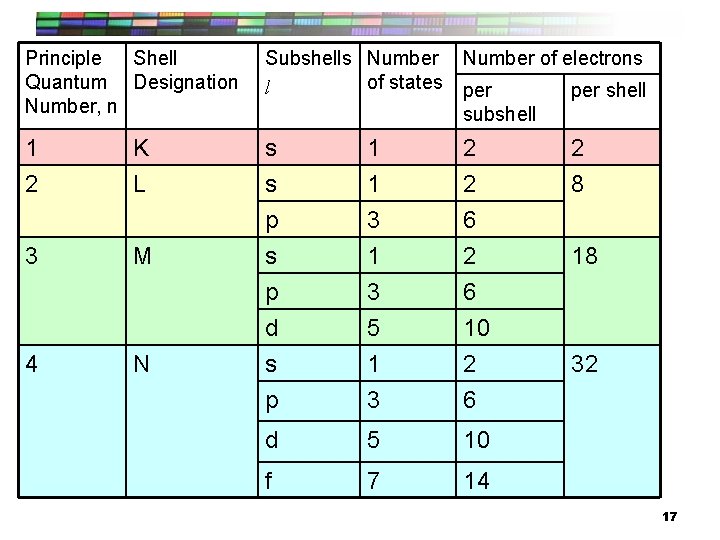
Principle Shell Quantum Designation Number, n Subshells Number of states l Number of electrons per subshell per shell 1 2 s s 1 1 2 2 2 8 p s p d s p 3 1 3 5 1 3 6 2 6 10 2 6 d 5 10 f 7 14 K L 3 M 4 N 18 32 17

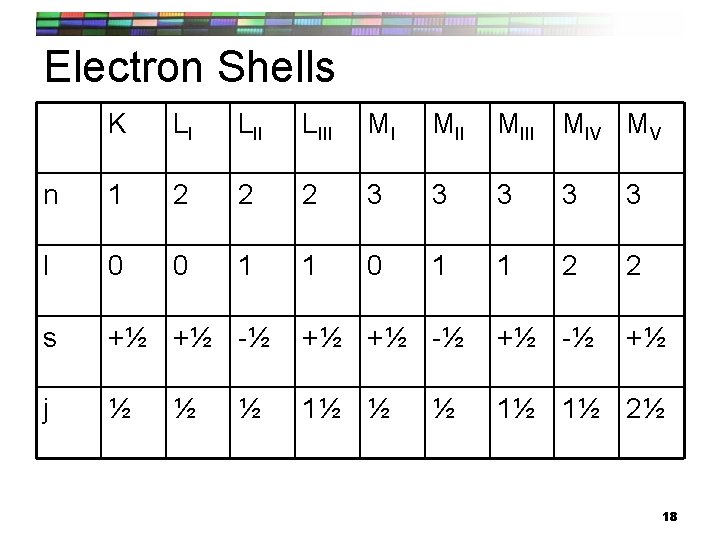
Electron Shells K LI LIII MI MIII MIV MV n 1 2 2 2 3 3 3 l 0 0 1 1 2 2 s +½ +½ -½ j ½ 1½ 1½ 2½ ½ +½ 18

Electron Shells K 1 s LI 2 s LII 2 p -½ LIII MI 2 p 3 s +½ MII 3 p -½ MIII MIV MV 3 p 3 d 3 d +½ -½ +½ 19

Electron Transitions 1. 2. 3. The change in n must be ≥ 1 (Δn ≠ 0) The change in l can only be ± 1 The change in j can only be ± 1 or 0 20

Calculation 1. 2. 3. The change in n must be ≥ 1 (Δn ≠ 0) The change in l can only be ± 1 The change in j can only be ± 1 or 0 2 p +½ to 1 s Quantu m# n l ml ms j Δ 21

Example of Electron Transitions Insert image 22

Spectroscopy and X-Ray Analysis n Electromagnetic Radiation ¨ Electromagnetic waves ¨ Calculations involving waves ¨ The electromagnetic spectrum ¨ Light and Optics ¨ Refraction and diffraction n X-Rays ¨ Discovery of X-rays ¨ Generation of X-rays ¨ Quantum Numbers ¨ Electron Energy Transitions 23