Electromagnetic Radiation especially Xrays Biology 555 Andrew J

Electromagnetic Radiation, especially X-rays Biology 555 Andrew J. Howard Based in part on notes from T. C. Irving and T. I. Morrison 11/15/2018 EM radiation, esp. X-rays 1

Electromagnetic Radiation l How do twenty-first-century scientists think about electromagnetic radiation? l The simplest articulation is that of the wave-particle duality: l Light (and other ranges of EM radiation) has both wave-like and particle-like properties l We’ll consider the two sets of properties in turn 11/15/2018 EM radiation, esp. X-rays p. 2 of 54

Why does this matter? l Many of the techniques we have discussed this semester involve interactions between biological macromolecules and electromagnetic radiation l Often the EM radiation we’ll be using is in the X-ray regime, so we’ll pay special attention to that range of energies or wavelengths 11/15/2018 EM radiation, esp. X-rays p. 3 of 54

Particle-like properties l We can view light as being composed of discrete packets of energy that we call photons l Photons have zero rest mass, and their velocity in vacuum is c l Their momenta are calculable: p = E/c = h /c 11/15/2018 EM radiation, esp. X-rays p. 4 of 54

Wavelike properties l When these packets of electromagnetic radiation interact in certain ways with matter and with one another, we observe wavelike phenomena like diffraction and refraction l. These properties were known to seventeenth-century physicists, but they were given a mathematical footing in the nineteenth century 11/15/2018 EM radiation, esp. X-rays p. 5 of 54
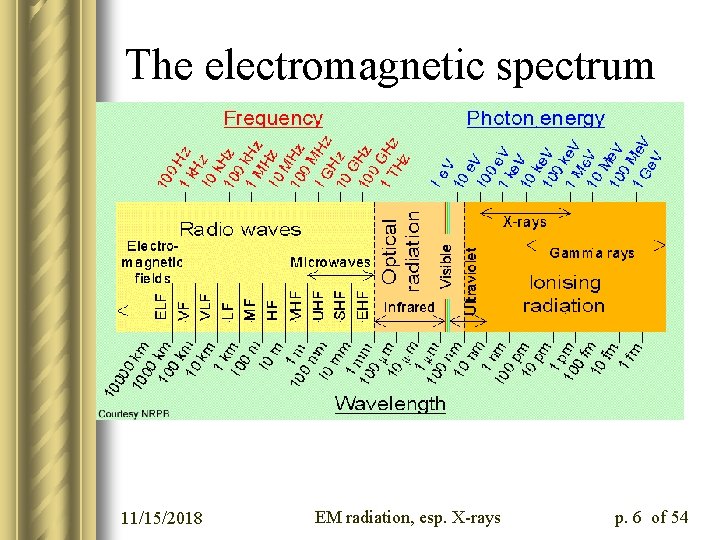
The electromagnetic spectrum 11/15/2018 EM radiation, esp. X-rays p. 6 of 54

Collisions l When EM radiation interacts in other ways with matter, we see scattering phenomena that we can best understand if we view its particle-like nature l EM radiation can transfer momentum to particles of matter in collisions or Compton scattering 11/15/2018 EM radiation, esp. X-rays p. 7 of 54

Somewhat more exotic phenomena… l Twentieth century physics includes experiments in which a photon or a pair of photons can produce a matter-antimatter pair, e. g. an electron and a positron. e- e+ 11/15/2018 EM radiation, esp. X-rays p. 8 of 54

Absorption of photons l When photons are absorbed in matter, some or all of the photon energy is transmitted to an electron and potentially to the rest of the atoms in the system. 11/15/2018 EM radiation, esp. X-rays p. 9 of 54

3 major forms of absorption l Photoelectric effect: light comes in, electron is liberated from atoms + + el Compton scattering: photon enters, scatters off an electron (either free or e bound) + el Pair production (see previous slide) 11/15/2018 EM radiation, esp. X-rays p. 10 of 54

Which one happens when? l Pair production requires that the incoming photon have at least as much energy as the rest energy of the positron and the electron, i. e. , at least 1. 022 Me. V l At various energies, various processes predominate to differing degrees; it also depends on the kinds of atoms that the photons are interacting with. 11/15/2018 EM radiation, esp. X-rays p. 11 of 54

Compton Scattering Energy conservation: Ee-, rest + Eg, in = Eg, out + Ee-, out 1. Energy conservation 2. Momentum conservation in x 3. Momentum conservation in y Momentum conservation in x and y: 11/15/2018 EM radiation, esp. X-rays p. 12 of 54

Compton scattering: equations 11/15/2018 EM radiation, esp. X-rays p. 13 of 54

Compton scattering: results l And therefore h ’ = h / {1 + (1 – cosq)} 11/15/2018 EM radiation, esp. X-rays p. 14 of 54

Attenuation of EM radiation l. Traditionally defined by saying that the fraction lost per unit distance is proportional to the distance traveled through the medium: ld. I/I = -µdx where µ is the linear attenuation coefficient. l. Solving this differential equation with I = I 0 at x = 0 gives I(x) = I 0 exp(-µx) l. So we have derived exponential decay! 11/15/2018 EM radiation, esp. X-rays p. 15 of 54

Practical uses of X rays l Remember that X-rays are simply electromagnetic radiation in the 1 -100 Ke. V range (corresponds to roughly 0. 01 nm - 1 nm) l We have traditionally used them for: – Radiography – X-ray scattering and diffraction – X-ray spectroscopy 11/15/2018 EM radiation, esp. X-rays p. 16 of 54

Emerging uses l X-ray microscopy l X-ray phase contrast imaging l Diffraction-enhanced imaging (developed significantly at IIT) 11/15/2018 EM radiation, esp. X-rays p. 17 of 54

How to make X-rays l Traditional method (goes back to Wilhelm Röntgen, 1895) employs a “reverse Compton effect”: a fast electron collides with a metal target, changing the momentum of the electron and thereby liberating a photon from it. photons Filament (voltage ~ k. V) 11/15/2018 e- target EM radiation, esp. X-rays p. 18 of 54

Wavelengths and energies l Typical reverse-Compton wavelengths range from 0. 01 nm to 10 nm; l Above 1 nm these are ultraviolet; below that they’re X-rays l Remember E = hc/ ~ 12. 398 / if E in ke. V and in Ångström, or E = 1. 2398 / if in nm 11/15/2018 EM radiation, esp. X-rays p. 19 of 54

Two ways to make EM from electrons l. Bremsstrahlung (braking) radiation: Maxwell’s equations say that any charged particle that changes momentum (speed or direction) must radiate, even if it’s just bending or slowing a little l. Characteristic radiation: electron interacts with atom, causing a bound electron to be promoted to a higher energy level. As it falls back to a lower level, it emits a photon with a very specific energy. 11/15/2018 EM radiation, esp. X-rays p. 20 of 54

How does characteristic radiation work? l An electron is promoted from a K or L shell into a higher shell l After a brief interval it decays back to the shell it starts in l As it does so it emits a photon whose energy is exactly the difference between the energies of the two energy levels h e- 11/15/2018 EM radiation, esp. X-rays p. 21 of 54

Characteristic X-ray Emission Lines: Atomic Energy Level Transitions http: //xdb. lbl. gov/Section 1/Sec_1 -2. html 11/15/2018 EM radiation, esp. X-rays p. 22 of 54

Energetics of characteristic radiation l. Most common transitions are k l, k m lk l : (K ) = hc/El- hc/Ek lk m : (K ) = hc/Em- hc/ Ek l. Copper K has = 1. 5418Å; energy is about 8 ke. V l. Molybdenum is ~ 0. 71Å, 15. 7 ke. V l. These are popular because these metals are fairly easy to cool with water 11/15/2018 EM radiation, esp. X-rays p. 23 of 54
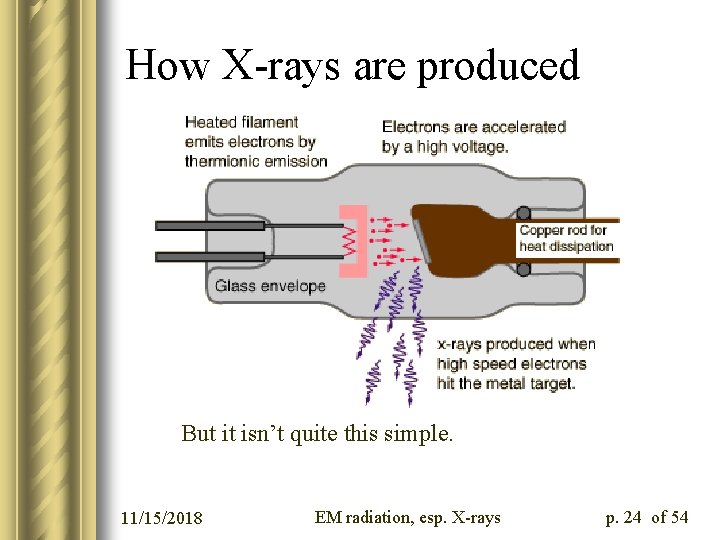
How X-rays are produced But it isn’t quite this simple. 11/15/2018 EM radiation, esp. X-rays p. 24 of 54

An ideal X-ray source l The source should be: – Intense – Spatially small – Non-dispersive – Monochromatic (for most applications) 11/15/2018 EM radiation, esp. X-rays p. 25 of 54

Intensity l More photons on the sample allow for shorter data acquisition times and might help cut down on radiation damage per unit information l But! X-ray production from target metals is inefficient l Total input power = acclerating potential * input beam current l 99% of input power goes into heat 11/15/2018 EM radiation, esp. X-rays p. 26 of 54

Wait! It gets worse! Electron beam l X-rays are produced nearly isotropically from conventional sources, for which the electrons are nonrelativisitic: relatively few go in the direction you want them to go. 11/15/2018 Your experiment EM radiation, esp. X-rays p. 27 of 54

How bad is it? An example: 3 k. W X-ray source 1. 54 Å radiation (8. 05 ke. V) 107 photons/sec (107 *8. 05 *103 *1. 6 *10 -19)/(3*103) ~ 0 realistic efficiency: 4* 10 -12. 11/15/2018 EM radiation, esp. X-rays p. 28 of 54

Power supplies Virtually always the anode floats at 20 -80 KV; the cathode is grounded or held at a small bias 11/15/2018 EM radiation, esp. X-rays p. 29 of 54

Traditional rotating anodes This has typically meant big, heavy supplies with big, heavy transformers (e. g. , 18 k. W = 60 k. V * 300 m. A) http: //www. rigaku. com/protein/ruh 3 r. html 11/15/2018 EM radiation, esp. X-rays p. 30 of 54

Compact, high-brilliance sources l Net power lower but comparable number of photons on sample http: //www. voltronics. com/products/index. html 11/15/2018 EM radiation, esp. X-rays p. 31 of 54

How to cool the target l. Spread the electron beam out l. Shape the target: –so the beam comes off at a glancing angle: –Projected source is: Actual source x sin(takeoff angle) l. Active cooling: Water flows through and past metal target l. Move electron beam along the target (or move the target along the beam): The rotating anode 11/15/2018 EM radiation, esp. X-rays p. 32 of 54

We usually want monochromatic radiation l We can use diffraction from a highly ordered crystal to select out a single and therefore a single wavelength, to within the acceptance of the crystal – Graphite traditionally used with conventional sources – Silicon or germanium more common with storage ring sources l Or we can filter out unwanted wavelengths with an absorber that preferentially absorbs away from the primary energy (Z = Ztarget-1) 11/15/2018 EM radiation, esp. X-rays p. 33 of 54

Can we focus X-rays? l Yes, but: l X-ray lenses per se are difficult because the refractive index of most materials is extremely close to 1 for high energies l But there are ways to use total internal reflection to get the same effect l We can also use diffraction to accomplish focusing 11/15/2018 EM radiation, esp. X-rays p. 34 of 54

What do X-rays do to biological samples? l Three things can happen to a forward pass: two of them are bad. -Bart Starr l Similarly, Xrays can: – Go through the sample unaffected – Get scattered or diffracted – Get absorbed l Absorption leads to sample damage l Two kinds of damage: l Direct: photon displaces atom in the polymer l Indirect: photon produces free radicals that diffuse to the polymer and mess it up 11/15/2018 EM radiation, esp. X-rays p. 35 of 54

How can we mitigate damage? l Not much you can do about direct damage: it’s zeroth-order l Free radical scavengers (ascorbate, glutathione): occasionally work l Cooling! – 110 K reduces radical mobility enough that the polymer survives – Good & bad: atoms don’t move as much, but cooling itself causes some disorder, and the experiment is less representative of biological system – Cryoprotection techniques thoroughly developed; Cryocrystallography and Cryo. EM trade ideas with one another. 11/15/2018 EM radiation, esp. X-rays p. 36 of 54

Special Relativity l Einstein modified galilean relativity, under which velocities are additive. l Galileo: automobile velocity with respect to ground = u = v + w l Einstein says: u < c so we need new rules! 11/15/2018 EM radiation, esp. X-rays p. 37 of 54

Special Relativity: Energy l Einstein’s new rules require that time & distance formulae depend on velocity l Corrections are significant in nuclear reactions, radiation scattering, and accelerators, so we study them here a little l They also give rise to the concept of relativistic energy 11/15/2018 EM radiation, esp. X-rays Hendrick Lorentz p. 38 of 54

Rest energy and mass l If v = 0, E = E 0 = m 0 c 2; this is rest energy if m 0 is the rest mass, i. e. the mass as it is ordinarily defined. l We can summarize the results by defining a relativistic mass m so that we can say E = mc 2 = gm 0 c 2 where m = gm 0 l For v = 0. 1 c, m = 1. 05 m 0 for v = 0. 98 c, m = 5 m 0. l Why do we care about SR here? EM radiation travels with v = c? l Answer: because the electrons that produce it are relativistic! 11/15/2018 EM radiation, esp. X-rays p. 39 of 54

Atomic Structure l JJ Thomson (1897): heavy nucleus with electrons surrounding it. l Rutherford showed that the nucleus had to be very small relative to the atomic size 11/15/2018 EM radiation, esp. X-rays p. 40 of 54

The Bohr Model l Bohr model: quantized angular momentum so that radiation is emitted in quanta equal to difference between energy levels of the atom. Used classical energy calculations! 11/15/2018 EM radiation, esp. X-rays p. 41 of 54

Bohr model: radius l. Quantized angular momentum mvr = nh/2 p l. But this is associated with coulombic attraction for which the centripetal force must equal the coulombic force: F = mv 2/r = k. Ze 2/r 2, so r = k. Ze 2/mv 2 l. Thus v = nh/(2 pmr) = 2 pk. Ze 2/nh so r = n 2 h 2/(4 p 2 k. Ze 2 m) l. For n=Z=1, r = 0. 529*10 -10 m = Bohr radius 11/15/2018 EM radiation, esp. X-rays p. 42 of 54

Bohr model: Electron Energies l Velocity = v = 2 pk. Ze 2/(nh) l Kinetic energy = 1/2 mv 2 = 2 p 2 k 2 Z 2 e 4 m/(n 2 h 2) l Potential energy = -k. Ze 2/r = -4 p 2 k 2 Z 2 e 4 m/(n 2 h 2) l Total energy = KE + PE = -2 p 2 k 2 Z 2 e 4 m/(n 2 h 2) l Photons emerge from transitions from one value of n to another. l Transition from n = 3 to n = 2 gives photon energy = -2 p 2 k 2 Z 2 e 4 m / [(1/9 - 1/4) h 2] = 1. 89 e. V 11/15/2018 EM radiation, esp. X-rays p. 43 of 54

De. Broglie Wave Theory l So: we’ve allowed electromagnetic radiation to behave as a wave and a particle. We can express momentum of light as P = E/c = hn/c = h/l l Can we also talk about matter behaving both as a wave and a particle? Yes. l Particles can exhibit interference effects associated with wave behavior. l Wavelength l = h/P = h / mv 11/15/2018 EM radiation, esp. X-rays p. 44 of 54

Wave Behavior in electrons l Nonrelativistic approximation: KE = (1/2)mv 2 so l = h/(mv) = h(2(KE)m)-1/2 l Further, since the angular momentum mvr is quantized (mvr = nh/(2 p)), we can say 2 pr = n l So we can say that the circumference of the electron’s orbit is an integer multiple of the electron’s wavelength! Standing waves! 11/15/2018 EM radiation, esp. X-rays p. 45 of 54

Synchrotron radiation l Accelerating any charged particle will give rise to electromagnetic radiation l Accelerating electrons that are traveling at close to the speed of light produces radiation in the ultraviolet or X-ray range l A simple way to do this is to accelerate the electrons on a straight path and then squirt them into a roughly circular ring l If acceleration is roughly perpendicular to the direction of motion, we describe the resulting radiation as synchrotron radiation l Since v = c – d, SR applies to these electrons! 11/15/2018 EM radiation, esp. X-rays p. 46 of 54

A very brief history of synchrotron facilities l 1960 -1980: Roughly-circular electron colliders enabled high-energy physics experiments in which electrons collided with each other or with positrons, producing showers of exotic particles l Synchrotron radiation was an unavoidable byproduct of these setups; researchers realized that they could capture the radiation as sources l 1980’s: Synchrotron experiments became so popular that dedicated storage-rings began to be built (SSRL, NSLS, several in Europe, Asia) l 1990’s: High-brilliance facilities containing wigglers or undulators (below) built and instrumented 11/15/2018 EM radiation, esp. X-rays p. 47 of 54

Curves and straight sections l In the curved sections of a storage ring, the X-rays are produced by bending the e-beam with dipole magnets so it curves along the path; this produces a “dipole” source l In the straight sections the beam can be wiggled or undulated through a series of tetrapole or hexapole magnets. If the magnet array exploits resonance between the radiation emitted from one segment and the next, we describe the source as an undulator; if not, it’s a wiggler 11/15/2018 EM radiation, esp. X-rays p. 48 of 54

Bandpass l Bending magnets and wigglers produce a broad spectral distribution; so if you want one wavelength, you need to select it with a monochromator l Undulators can be tuned to produce a narrow bandpass (DE/E~0. 01 -0. 1); can be further monochromatized if necessary 11/15/2018 EM radiation, esp. X-rays p. 49 of 54

Time-resolved studies l Each electron requires 2 pr/c ~ 3. 7*10 -6 sec to make a full circuit l Storage ring electrons aren’t circulating at a constant rate; there’s a time-structure to their emission (ps-scale “buckets” repeated every few ns) l We can capture X-ray photons emitted during parts of that cycle l Not enough photons for monochromatic studies, but for white-beam or pink-beam studies this time-structure can be used. 11/15/2018 EM radiation, esp. X-rays p. 50 of 54

Example of time-resolved MX l Many biochemical reactions can be triggered by ps or fs laser pulses and are fully reversible on a ~msec timescale l So synchronize the X-ray pulse so it happens at a set interval t after the laser pulse is issued l Then let the system relax back to ground state and do it again (laser, then X-ray); rinse, repeat l Assemble the data from all t-interval pulses as one sample-state; and all t’-interval pulses as a different sample-state l Use these to create a “movie” of the reaction 11/15/2018 EM radiation, esp. X-rays p. 51 of 54

Free electron lasers l Remember that lasers are coherent sources of light produced by stimulated emission l If the charged particles producing the coherent radiation are high-energy electrons moving freely through a magnetic array, we call this a free electron laser (microwave -> Xray) l Magnetic lattices are undulators l ~2012: Practical problems overcome for ~10 Ke. V FELs; LCLS at Stanford and a few others built l Very short (fs) pulse lengths, breathtakingly high brilliance 11/15/2018 EM radiation, esp. X-rays p. 52 of 54

Applications of FELs in structural biology l Incredible brilliance means every sample is destroyed when the first pulse hits it, so you expose a stream of crystals past the beam and merge the images to get a complete structure l Ultra-small crystals (< 1 µm) ideal l Room-temperature data collection is fine l First experiments involved ~2*105 xtals; newer injectors require ~200 l Sophisticated flow systems developed at Arizona State and elsewhere l There are non-crystallographic biological applications too 11/15/2018 EM radiation, esp. X-rays p. 53 of 54

Time-structure at XFELs l Pulses are a few fs in duration and each X-ray image is acquired from a single pulse l New pulse delivered once per 100 msec at LCLS; higher rep rate at newer XFELs l Triggering a time-resolved experiment is straightforward l Since every image is a still, you need a repeatable trigger unless you can acquire useful data from one still image 11/15/2018 EM radiation, esp. X-rays p. 54 of 54
- Slides: 54