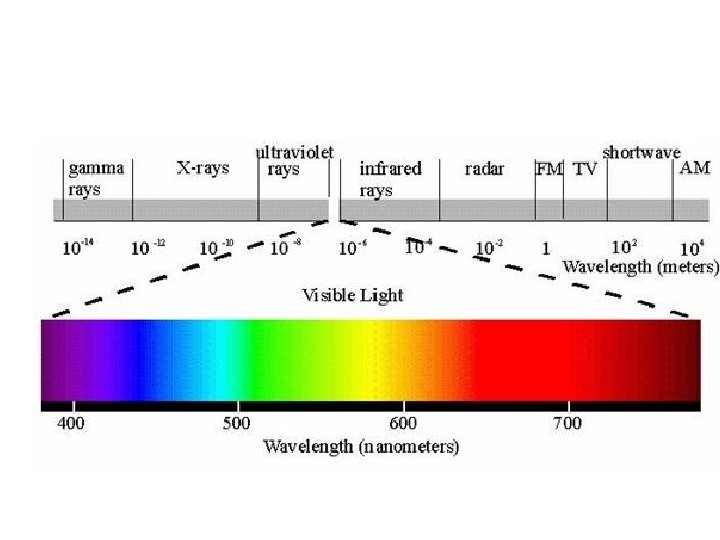
Electromagnetic Spectrum ELECTROMAGNETIC RADIATION electromagnetic radiationUV visible light

Electromagnetic Spectrum

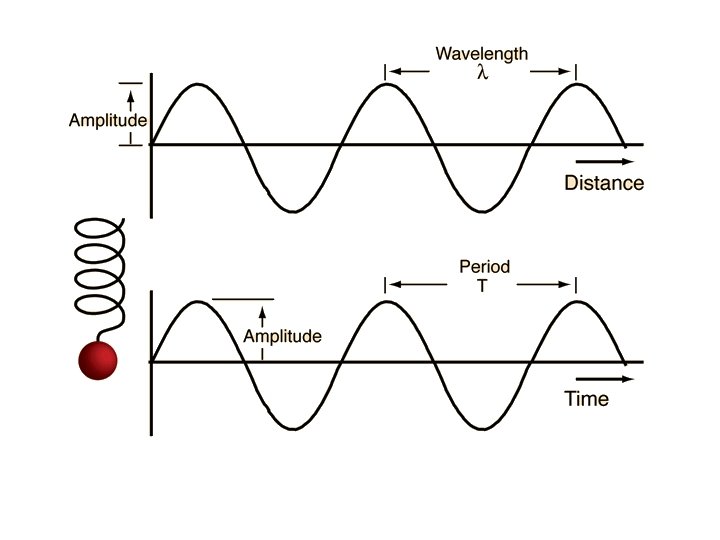
ELECTROMAGNETIC RADIATION • electromagnetic radiation—UV, visible light, IR, microwaves, television and radio signals, and Xrays • wavelength (λ)—(lambda) length between 2 successive crests. • frequency (υ)—number of cycles per second that pass a certain point (Hz-cycles per second) • amplitude—maximum height of a wave • nodes—points of zero amplitude

Electromagnetic Radiation • c= λμ “c”—the speed of light is 3 × 108 m/s; • ALL EM RADIATION TRAVELS AT THIS SPEED! • λ and υ are inversely proportional

A laser emits light of a frequency 4. 74*10 -14. What is the wavelength of the light? • C = 3 x 108 • 3 x 108 /4. 74 x 1014 = 6. 3 * 10 -6

Plank’s Constant • h is a proportionality constant called Planck's constant: • h = 6. 6260755 × 10 -34 joule • seconds.

De. Broglie • De. Boglie equation: • λ=h/mv • All matter has wave properties!

De Broglie Cont. • The more massive the object, the smaller its associated wavelength and vice versa! • ANY moving particle has an associated wavelength. • E is then really a form of matter, and ALL matter shows the same types of properties. That is, all matter exhibits both particulate and wave properties.

Practice • A racecar weighing 1496 kg and traveling at 89. 4 m/s would have a De. Broglie wavelength of _____. • 4. 95 * 10 -39
- Slides: 10