DR JOSEPH SEBASTIAN Senior Consultant Neurologist Caritas Hospital

DR JOSEPH SEBASTIAN Senior Consultant Neurologist Caritas Hospital, Kottayam DM (neurology) from PGIMER, Chandigarh PHOTO YOUR Fellowship in Cerebrovascular Diseases from University of Alberta, Canada Received Award from ICMR, for the clinical research paper - Clinical outcome of Cerebral Venous Sinus Thrombosis - A CT Perfusion Correlate A certified practitioner by American Society of Neuroimaging in Neurosonology Published articles in international peer reviewed Journals

NOAC DOAC NO AC DR JOSEPH SEBASTIAN MD, DM(NEUROLOGY)(PGI) Fellowship in Vascular Neurology (University Of Alberta, Canada) DEPARTMENT OF NEUROSCIENCES CARITAS HOSPITAL

Objectives § NOACS VS WARFARIN § Which NOAC TO CHOOSE § Anticoagulation in Cerebral Amyloid Angiopathy § Reinitiation of ANTICOAGULATION after IC BLEED § Role in Cervical Artery Dissection
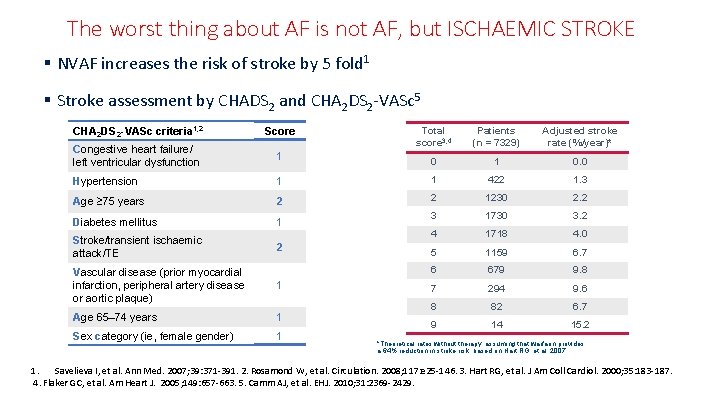
The worst thing about AF is not AF, but ISCHAEMIC STROKE § NVAF increases the risk of stroke by 5 fold 1 § Stroke assessment by CHADS 2 and CHA 2 DS 2 -VASc 5 CHA 2 DS 2 -VASc criteria 1, 2 Score Congestive heart failure/ left ventricular dysfunction Total score 3, 4 Patients (n = 7329) Adjusted stroke rate (%/year)* 1 0. 0 Hypertension 1 1 422 1. 3 Age 75 years 2 2 1230 2. 2 Diabetes mellitus 1 3 1730 3. 2 Stroke/transient ischaemic attack/TE 4 1718 4. 0 2 5 1159 6. 7 6 679 9. 8 7 294 9. 6 8 82 6. 7 9 14 15. 2 Vascular disease (prior myocardial infarction, peripheral artery disease or aortic plaque) 1 Age 65– 74 years 1 Sex category (ie, female gender) 1 *Theoretical rates without therapy; assuming that warfarin provides a 64% reduction in stroke risk, based on Hart RG, et al. 2007. 1. Savelieva I, et al. Ann Med. 2007; 39: 371 -391. 2. Rosamond W, et al. Circulation. 2008; 117: e 25 -146. 3. Hart RG, et al. J Am Coll Cardiol. 2000; 35: 183 -187. 4. Flaker GC, et al. Am Heart J. 2005; 149: 657 -663. 5. Camm AJ, et al. EHJ. 2010; 31: 2369 -2429.
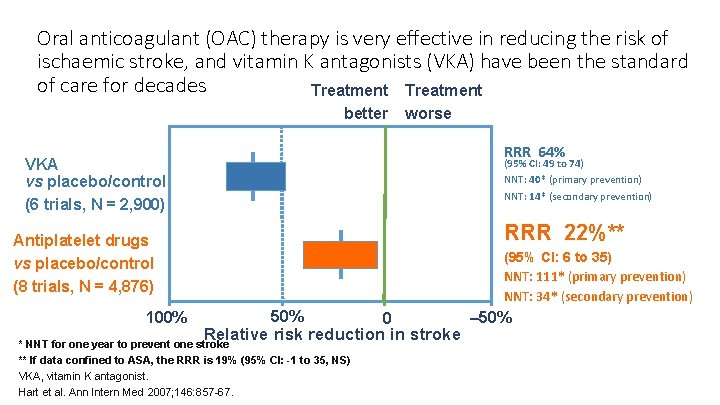
Oral anticoagulant (OAC) therapy is very effective in reducing the risk of ischaemic stroke, and vitamin K antagonists (VKA) have been the standard of care for decades Treatment better worse RRR 64% VKA vs placebo/control (6 trials, N = 2, 900) (95% CI: 49 to 74) NNT: 40* (primary prevention) NNT: 14* (secondary prevention) RRR 22%** Antiplatelet drugs vs placebo/control (8 trials, N = 4, 876) 100% (95% CI: 6 to 35) NNT: 111* (primary prevention) NNT: 34* (secondary prevention) 50% 0 Relative risk reduction in stroke * NNT for one year to prevent one stroke ** If data confined to ASA, the RRR is 19% (95% CI: -1 to 35, NS) VKA, vitamin K antagonist. Hart et al. Ann Intern Med 2007; 146: 857 -67. – 50%

Serious side effects and limitations of VKA therapy Unpredictable response Narrow therapeutic window (INR range 2 -3) Routine coagulation monitoring Slow onset/offset of action VKA therapy has several limitations that make it difficult to use in practice Intracranial Bleeding 1. Ansell J, et al. Chest 2008; 133; 160 S-198 S; 2. Umer Ushman MH, et al. J Interv Card Electrophysiol 2008; 22: 129 -137; Nutescu EA, et al. Cardiol Clin 2008; 26: 169 -187. Frequent dose adjustments Numerous fooddrug interactions Numerous drug interactions Warfarin resistance CYP 2 C 9, VKORC 1 genetic polymorphisms

Selecting appropriate antithrombotic therapy for a patient is an important management decision in AF BALANCING RISK Stroke Bleeding Aim: Reducing the risk of thrombotic events with the minimal risk of bleeding complications Fang. Ann Intern Med 2011; 155: 636 -7.
NOACS or DOAC

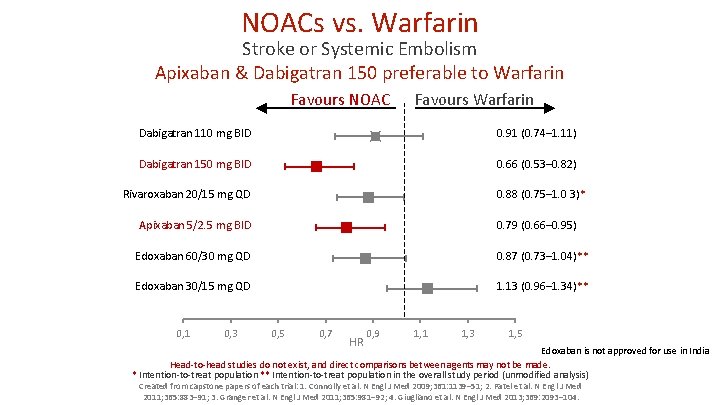
NOACs vs. Warfarin Stroke or Systemic Embolism Apixaban & Dabigatran 150 preferable to Warfarin Favours NOAC Favours Warfarin Dabigatran 110 mg BID 0. 91 (0. 74– 1. 11) Dabigatran 150 mg BID 0. 66 (0. 53– 0. 82) Rivaroxaban 20/15 mg QD 0. 88 (0. 75– 1. 0 3)* Apixaban 5/2. 5 mg BID 0. 79 (0. 66– 0. 95) Edoxaban 60/30 mg QD 0. 87 (0. 73– 1. 04)** Edoxaban 30/15 mg QD 1. 13 (0. 96– 1. 34)** 0, 1 0, 3 0, 5 0, 7 HR 0, 9 1, 1 1, 3 1, 5 Edoxaban is not approved for use in India Head-to-head studies do not exist, and direct comparisons between agents may not be made. * Intention-to-treat population ** Intention-to-treat population in the overall study period (unmodified analysis) Created from capstone papers of each trial: 1. Connolly et al. N Engl J Med 2009; 361: 1139– 51; 2. Patel et al. N Engl J Med 2011; 365: 883– 91; 3. Granger et al. N Engl J Med 2011; 365: 981– 92; 4. Giugliano et al. N Engl J Med 2013; 369: 2093– 104.
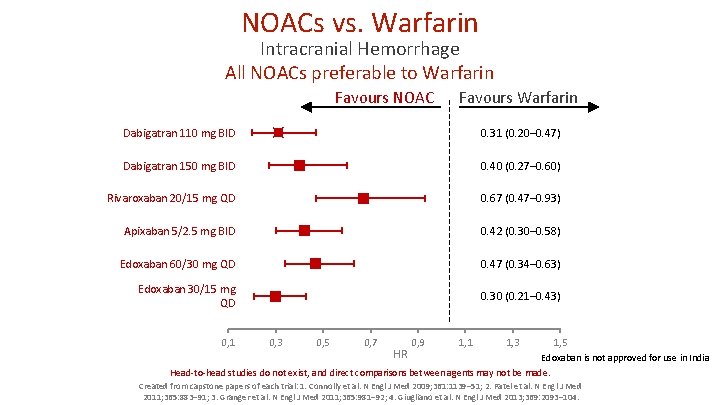
NOACs vs. Warfarin Intracranial Hemorrhage All NOACs preferable to Warfarin Favours NOAC Favours Warfarin Dabigatran 110 mg BID 0. 31 (0. 20– 0. 47) Dabigatran 150 mg BID 0. 40 (0. 27– 0. 60) Rivaroxaban 20/15 mg QD 0. 67 (0. 47– 0. 93) Apixaban 5/2. 5 mg BID 0. 42 (0. 30– 0. 58) Edoxaban 60/30 mg QD 0. 47 (0. 34– 0. 63) Edoxaban 30/15 mg QD 0. 30 (0. 21– 0. 43) 0, 1 0, 3 0, 5 0, 7 HR 0, 9 1, 1 1, 3 1, 5 Edoxaban is not approved for use in India Head-to-head studies do not exist, and direct comparisons between agents may not be made. Created from capstone papers of each trial: 1. Connolly et al. N Engl J Med 2009; 361: 1139– 51; 2. Patel et al. N Engl J Med 2011; 365: 883– 91; 3. Granger et al. N Engl J Med 2011; 365: 981– 92; 4. Giugliano et al. N Engl J Med 2013; 369: 2093– 104.
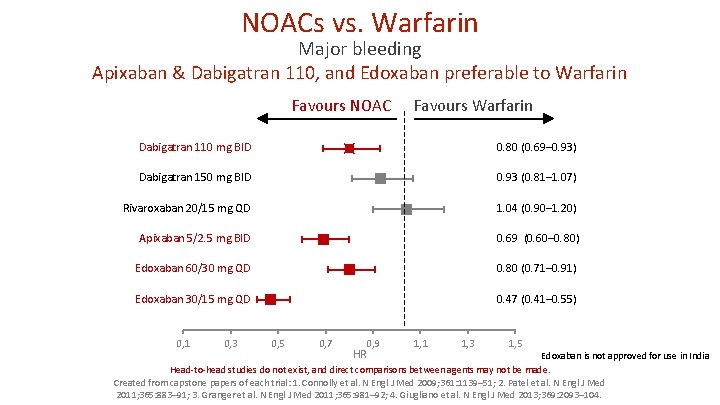
NOACs vs. Warfarin Major bleeding Apixaban & Dabigatran 110, and Edoxaban preferable to Warfarin Favours NOAC Favours Warfarin Dabigatran 110 mg BID 0. 80 (0. 69– 0. 93) Dabigatran 150 mg BID 0. 93 (0. 81– 1. 07) Rivaroxaban 20/15 mg QD 1. 04 (0. 90– 1. 20) Apixaban 5/2. 5 mg BID 0. 69 (0. 60– 0. 80) Edoxaban 60/30 mg QD 0. 80 (0. 71– 0. 91) Edoxaban 30/15 mg QD 0. 47 (0. 41– 0. 55) 0, 1 0, 3 0, 5 0, 7 0, 9 HR 1, 1 1, 3 1, 5 Edoxaban is not approved for use in India Head-to-head studies do not exist, and direct comparisons between agents may not be made. Created from capstone papers of each trial: 1. Connolly et al. N Engl J Med 2009; 361: 1139– 51; 2. Patel et al. N Engl J Med 2011; 365: 883– 91; 3. Granger et al. N Engl J Med 2011; 365: 981– 92; 4. Giugliano et al. N Engl J Med 2013; 369: 2093– 104.

GUIDELINES
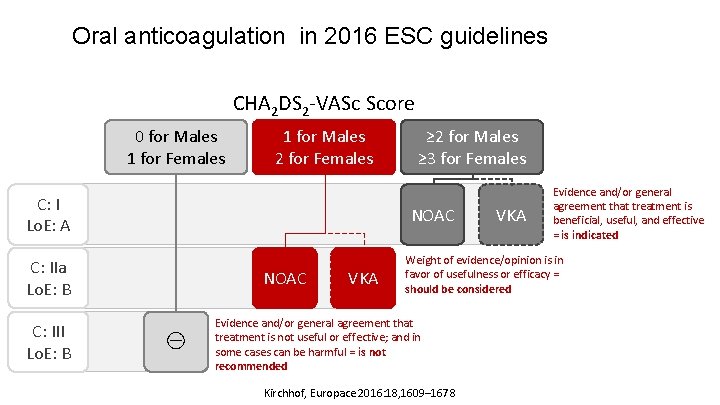
Oral anticoagulation in 2016 ESC guidelines CHA 2 DS 2 -VASc Score 0 for Males 1 for Females 1 for Males 2 for Females C: I Lo. E: A NOAC C: IIa Lo. E: B C: III Lo. E: B ≥ 2 for Males ≥ 3 for Females NOAC ⊖ VKA Evidence and/or general agreement that treatment is beneficial, useful, and effective = is indicated Weight of evidence/opinion is in favor of usefulness or efficacy = should be considered Evidence and/or general agreement that treatment is not useful or effective; and in some cases can be harmful = is not recommended Kirchhof, Europace 2016: 18, 1609– 1678
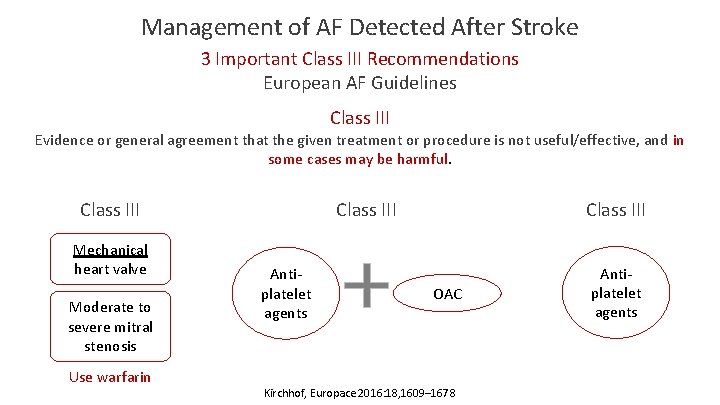
Management of AF Detected After Stroke 3 Important Class III Recommendations European AF Guidelines Class III Evidence or general agreement that the given treatment or procedure is not useful/effective, and in some cases may be harmful. Class III Mechanical heart valve Moderate to severe mitral stenosis Use warfarin Class III Antiplatelet agents OAC Kirchhof, Europace 2016: 18, 1609– 1678 Antiplatelet agents
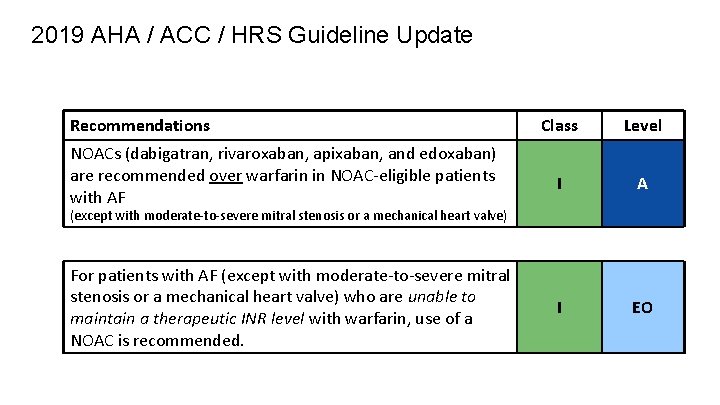
2019 AHA / ACC / HRS Guideline Update Recommendations Class Level NOACs (dabigatran, rivaroxaban, apixaban, and edoxaban) are recommended over warfarin in NOAC-eligible patients with AF I A For patients with AF (except with moderate-to-severe mitral stenosis or a mechanical heart valve) who are unable to maintain a therapeutic INR level with warfarin, use of a NOAC is recommended. I EO (except with moderate-to-severe mitral stenosis or a mechanical heart valve)
Which NOAC to choose? Dabigatran Apixaban Rivaroxaban Edoxaban* *Not approved in India

NOACs in Elderly Patients
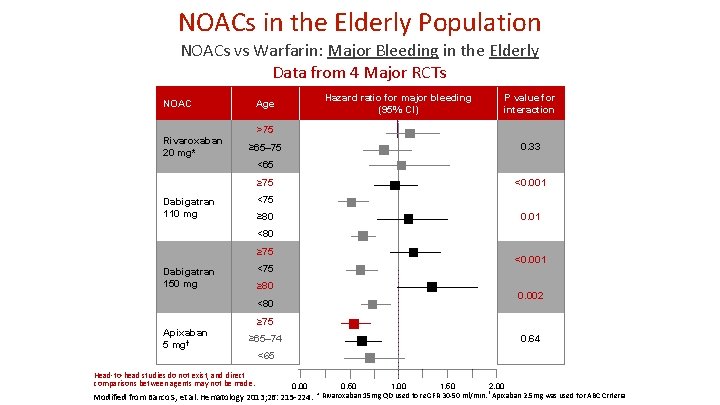
NOACs in the Elderly Population NOACs vs Warfarin: Major Bleeding in the Elderly Data from 4 Major RCTs NOAC Rivaroxaban 20 mg* Hazard ratio for major bleeding (95% CI) Age P value for interaction >75 0. 33 ≥ 65– 75 <65 ≥ 75 <0. 001 <75 Dabigatran 110 mg ≥ 80 0. 01 <80 ≥ 75 <0. 001 <75 Dabigatran 150 mg ≥ 80 0. 002 <80 ≥ 75 Apixaban 5 mg† ≥ 65– 74 Head-to-head studies do not exist, and direct comparisons between agents may not be made. 0. 64 <65 0. 00 0. 50 1. 00 1. 50 2. 00 Modified from Barco S, et al. Hematology 2013; 26: 215 -224. * Rivaroxaban 15 mg QD used for e. GFR 30 -50 ml/min. † Apixaban 2. 5 mg was used for ABC Criteria

NOACs in the Elderly Population Expert Recommendation >75 years Dabigatran 110 mg. Second BID First Choice Apixaban 5 mg twice daily Dabigatran 110 mg BID Edoxaban 60 mg daily Diener, et al. Eur Heart J 2017; 38: 860 -868. Edoxaban is not approved for use in India

NOACs in Renal Failure
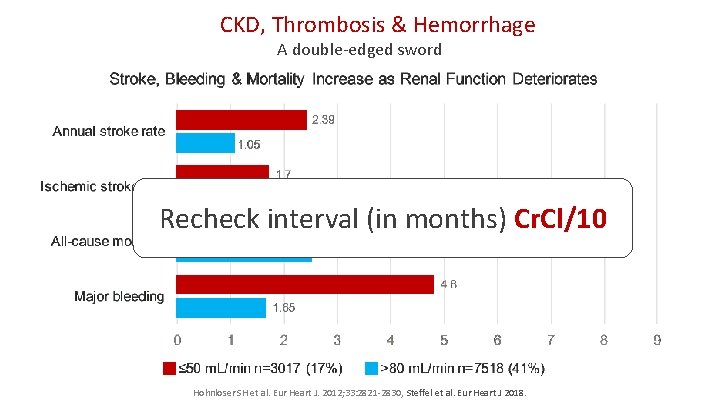
CKD, Thrombosis & Hemorrhage A double-edged sword Recheck interval (in months) Cr. Cl/10 Hohnloser SH et al. Eur Heart J. 2012; 33: 2821 -2830, Steffel et al. Eur Heart J 2018.
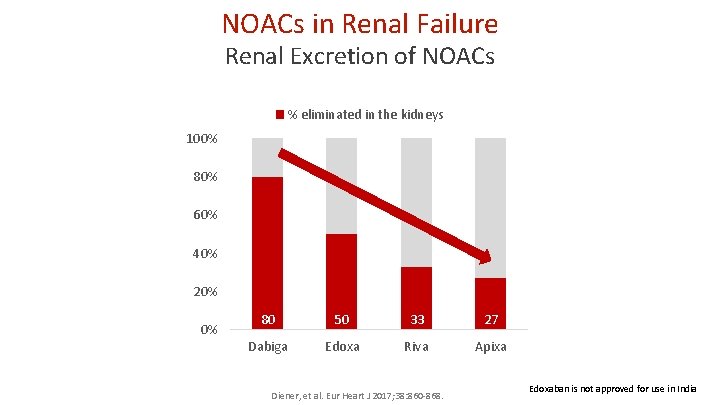
NOACs in Renal Failure Renal Excretion of NOACs % eliminated in the kidneys 100% 80% 60% 40% 20% 0% 80 50 33 27 Dabiga Edoxa Riva Apixa Diener, et al. Eur Heart J 2017; 38: 860 -868. Edoxaban is not approved for use in India
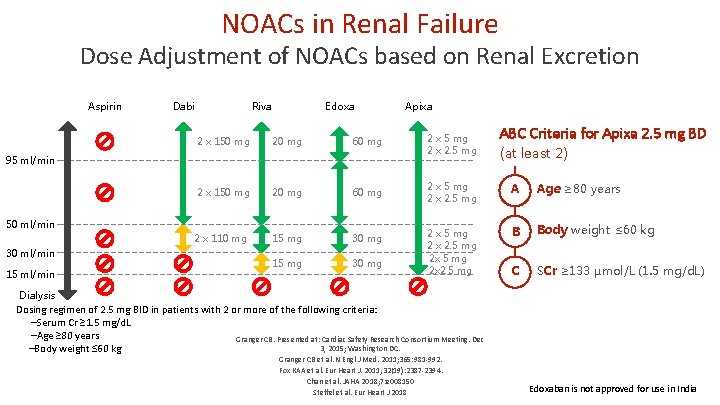
NOACs in Renal Failure Dose Adjustment of NOACs based on Renal Excretion Aspirin Dabi Riva Edoxa 20 mg 60 mg 2 x 5 mg 2 x 2. 5 mg 2 x 150 mg 20 mg 60 mg 2 x 5 mg 2 x 2. 5 mg A Age ≥ 80 years 2 x 110 mg 15 mg 30 mg B Body weight ≤ 60 kg 15 mg 30 mg 2 x 5 mg 2 x 2. 5 mg 2 x 2. 5 mg C SCr ≥ 133 µmol/L (1. 5 mg/d. L) 50 ml/min 15 ml/min ABC Criteria for Apixa 2. 5 mg BD (at least 2) 2 x 150 mg 95 ml/min 30 ml/min Apixa Dialysis Dosing regimen of 2. 5 mg BID in patients with 2 or more of the following criteria: –Serum Cr ≥ 1. 5 mg/d. L –Age ≥ 80 years Granger CB. Presented at: Cardiac Safety Research Consortium Meeting. Dec 3, 2015; Washington DC. –Body weight ≤ 60 kg Granger CB et al. N Engl J Med. 2011; 365: 981 -992. Fox KAA et al. Eur Heart J. 2011; 32(19): 2387 -2394. Chan et al. JAHA 2018; 7: e 008150 Steffel et al. Eur Heart J 2018 Edoxaban is not approved for use in India

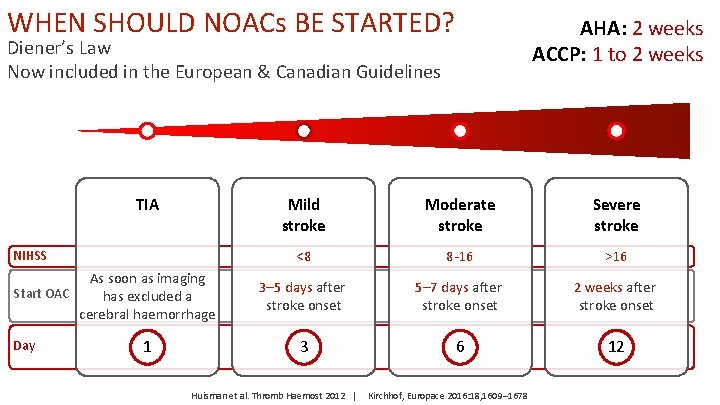
WHEN SHOULD NOACs BE STARTED? AHA: 2 weeks ACCP: 1 to 2 weeks Diener’s Law Now included in the European & Canadian Guidelines TIA NIHSS As soon as imaging Start OAC has excluded a cerebral haemorrhage Day 1 Mild stroke Moderate stroke Severe stroke <8 8 -16 >16 3– 5 days after stroke onset 5– 7 days after stroke onset 2 weeks after stroke onset 3 6 12 Huisman et al. Thromb Haemost 2012 | Kirchhof, Europace 2016: 18, 1609– 1678

WHEN SHOULD OACs BE STARTED? Other factors to be considered Waiting 14 d Early Anticoagulation Low NIHSS (<8): Small/no brain infarction on imaging High recurrence risk (e. g. cardiac thrombus) No need for gastrostomy No need for carotid surgery No hemorrhagic transformation Clinically stable Young patient Blood pressure is controlled No microbleeds Delayed Anticoagulation High NIHSS (≥ 8): Large/moderate brain infarction on imaging Lower recurrence risk Needs gastrostomy or major surgery Needs carotid surgery Hemorrhagic transformation Neurologically unstable Elderly patient Uncontrolled hypertension Microbleeds Modified from Kirchhof, Europace 2016: 18, 1609– 1678
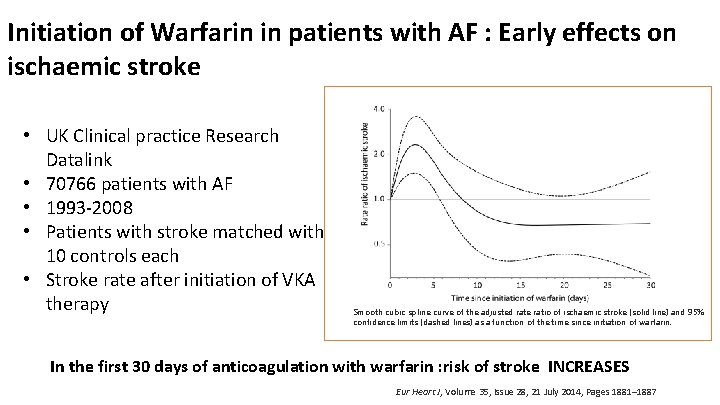
Initiation of Warfarin in patients with AF : Early effects on ischaemic stroke • UK Clinical practice Research Datalink • 70766 patients with AF • 1993 -2008 • Patients with stroke matched with 10 controls each • Stroke rate after initiation of VKA therapy Smooth cubic spline curve of the adjusted rate ratio of ischaemic stroke (solid line) and 95% confidence limits (dashed lines) as a function of the time since initiation of warfarin. In the first 30 days of anticoagulation with warfarin : risk of stroke INCREASES Eur Heart J, Volume 35, Issue 28, 21 July 2014, Pages 1881– 1887

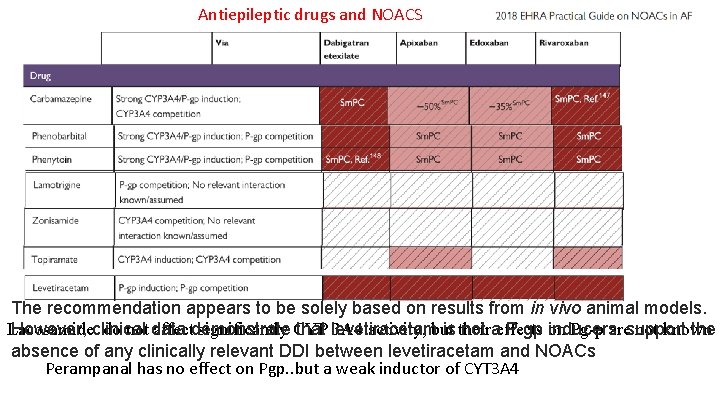
Antiepileptic drugs and NOACS The recommendation appears to be solely based on results from in vivo animal models. Lacosamide do not data affectdemonstrate significantly CYP 3 A 4 activity, but Pg-p aresupport not known However, clinical that levetiracetam is their not aeffects P-gp on inducers. the absence of any clinically relevant DDI between levetiracetam and NOACs Perampanal has no effect on Pgp. . but a weak inductor of CYT 3 A 4

EHRA guidance on the management of bleeding in patients using NOACs Bleeding while using a NOAC • • • Inquire about last NOAC intake Blood sample to determine Cr. Cl, haemoglobin, white blood cells Inquire with lab about possibility of rapid coagulation assessment Moderate/severe bleeding Mild bleeding Delay or discontinue next dose Reconsider concomitant medication • • Heidbuchel et al Eurospace 2015 Supportive measures Mechanical compression Endoscopic haemostasis if GI bleed Surgical haemostasis Fluid replacement (colloids if needed) RBC transfusion if needed FFP (as plasma expander) Platelet transfusion (if platelet count ≤ 60× 109/L) Dabigatran-treated patients • Idarucizumab 5 g IV • Maintain adequate diuresis • Consider haemodialysis Life-threatening bleeding Consider • For dabigatran-treated patients, idarucizumab 5 g IV • PCC 50 U/kg + 25 U/kg if indicated • a. PCC 50 U/kg; max 200 U/kg/day • r. FVIIa 90 µg/kg

Management of AIS in patient on NOAC • J Steffel et al. 2018 EHRA Practical Guide on NOACs in AF. European Heart Journal (2018) 39, 1330– 1393
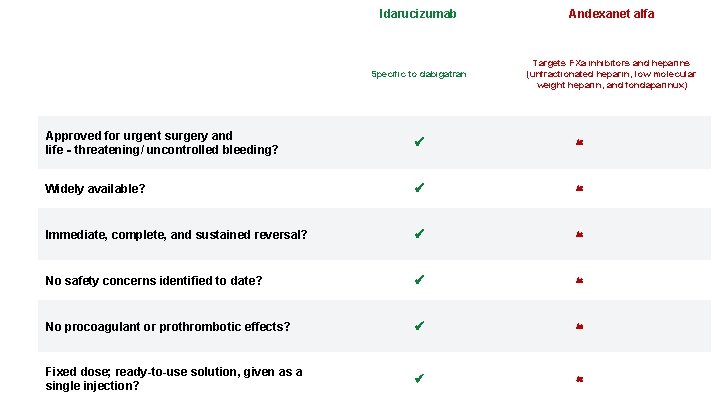
Idarucizumab Andexanet alfa Specific to dabigatran Targets FXa inhibitors and heparins (unfractionated heparin, low molecular weight heparin, and fondaparinux) Approved for urgent surgery and life‐threatening/ uncontrolled bleeding? Widely available? Immediate, complete, and sustained reversal? No safety concerns identified to date? No procoagulant or prothrombotic effects? Fixed dose; ready-to-use solution, given as a single injection?
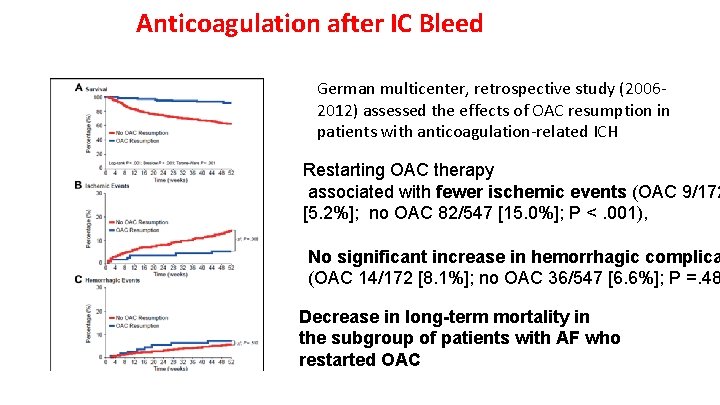
Anticoagulation after IC Bleed German multicenter, retrospective study (20062012) assessed the effects of OAC resumption in patients with anticoagulation-related ICH Restarting OAC therapy associated with fewer ischemic events (OAC 9/172 [5. 2%]; no OAC 82/547 [15. 0%]; P <. 001), No significant increase in hemorrhagic complica (OAC 14/172 [8. 1%]; no OAC 36/547 [6. 6%]; P =. 48 Decrease in long-term mortality in the subgroup of patients with AF who restarted OAC

When is it safe to resume anticoagulation after IC BLEED? NO published studies 2 ONGOING TRIALS…restarting NOACS The most recent AHA/ASA guidelines recommend 4 weeks after ICH (patients with prosthetic heart valve are excluded) In a recent survey of field experts, 4 -14 day delay for OAC resumption in prosthetic heart valve Reinitiation with NOACS is theoretically preferable given marked reduction in ICH rates Al. Kherayf F. , Xu Y. , Westwick H. Timing of anticoagulant re-initiation following intracerebral hemorrhage in mechanical heart valves: survey of neurosurgeons and thrombosis experts. Clin. Neurol. Neurosurg. 2017; 154: 23– 27.
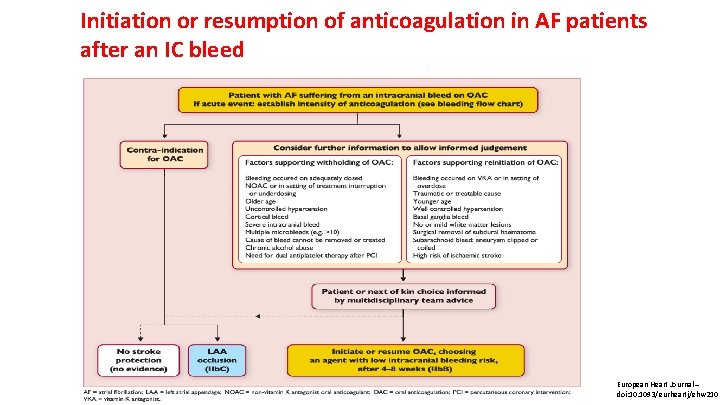
Initiation or resumption of anticoagulation in AF patients after an IC bleed European Heart Journal – doi: 10. 1093/eurheartj/ehw 210

Comorbid AF IN CEREBRAL AMYLOID ANGIOPATHY(CAA) • CAA - spontaneous lobar ICH in elderly • High risk of recurrence of -7%/year • In clinical practice, 25% of CAA-ICH patients with AF receive resumption of the oral anticoagulation after the ICH • Pooled analysis of 3 observational cohorts reported 70% RR in year mortality and 3 fold i likelihood of a favourable outcome (m. RS 0 -3) in possible and probable CAA who were started on anticoagulation(warfarin) NOACS might be an attractive option in CAA related ICH survivors - 50% lower risk of ICH compared to warfarin Cerebral amyloid angiopathy, cerebral microbleeds and implications for anticoagulation decisions: The need for a balanced approach Charidimou et al. Int J Stroke 2018; 13: 117 -120 Oral anticoagulation and functional outcome after ICH Biffi A et al Ann Neurol 2017 ; 82: 755 -765
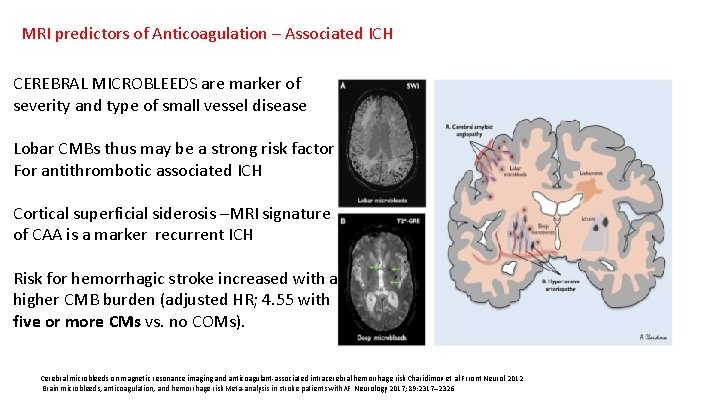
MRI predictors of Anticoagulation – Associated ICH CEREBRAL MICROBLEEDS are marker of severity and type of small vessel disease Lobar CMBs thus may be a strong risk factor For antithrombotic associated ICH Cortical superficial siderosis –MRI signature of CAA is a marker recurrent ICH Risk for hemorrhagic stroke increased with a higher CMB burden (adjusted HR; 4. 55 with five or more CMs vs. no COMs). Cerebral microbleeds on magnetic resonance imaging and anticoagulant-associated intracerebral hemorrhage risk Charidimov et al Frront Neurol 2012 Brain microbleeds, anticoagulation, and hemorrhage risk Meta-analysis in stroke patients with. AF Neurology 2017; 89: 2317– 2326

ANTIPLATELET VS ANTICOAGULATION IN CERVICAL ARTERY DISSECTION CADISS trial final results: 250 patients were randomized (118 carotid and 132 vertebral), 126 to AP and 124 to AC. The recurrent stroke rate at 1 year was 6 of 250 (2. 4%). No difference between treatment groups Of the 181 patients with confirmed dissection and complete imaging at baseline and 3 months, there was no difference recanalisation between those receiving AP or AC (P =. 97). The Cervical Artery Dissection in Stroke Study (CADISS) Randomized Clinical Trial Final Results. JAMA Neurol. 2019; 76(6): 657 -664. 200 patients with a confirmed diagnosis of extracranial carotid (n = 132) or vertebral (n = 68) artery dissection treated with AP (n = 136) or AC (n = 64). Six (4. 41%) patients in the AP and 1 patient (1. 56%) in the AC had recurrent ischemic events (P = 0. 434); SICH was more frequent in the anticoagulant group (9. 4% vs 3. 7%, P = 0. 185) Vineetha V S, Sreedharan SE, Sarma P S, Sylaja P N. Antiplatelets versus Anticoagulants in the Treatment of Extracranial Carotid and Vertebral Artery Dissection. Neurol India 2019; 67: 1056 -9

Any subgroup that would benefit from anticoagulation in extracranial dissection? May be beneficial in patients with greater risk of embolic events – • Microembolic signals(MES) in TCD. The presence of MES was associated with increased frequency of cortical infarctions in cervical artery dissection • Presence of mobile thrombus in arterial lumen • ? Severe stenotic and occlusive dissections Very limited information on the treatment of cervical dissection with NOACS Treatment of Extracranial Arterial Dissection: the Roles of Antiplatelet Agents, Anticoagulants, and Stenting Curr Treat Options Neurol(2019) 21: 48

Novel Oral Anticoagulants (NOACs) vs. Warfarin in the Treatment of Cerebral Venous Sinus Thrombosis (CVST) RESPECT CVT TRIAL: Patients treated with NOACs for CVST achieve similar rates of good functional outcome and recanalization as those treated with warfarin. While not statistically significant, higher rates of major hemorrhage in the warfarin group suggest a favorable safety profile of NOACs. Limited evidence for a recommendation

Thank you
- Slides: 42