Dissolving of Salt in Water Na ions Water






- Slides: 15

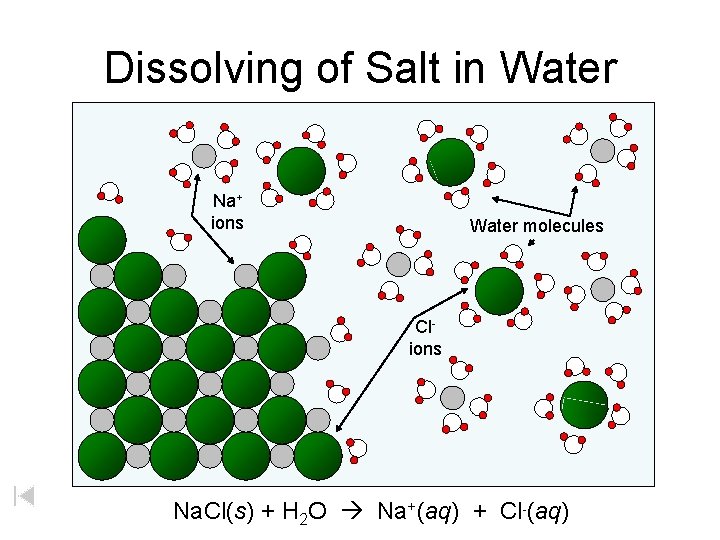
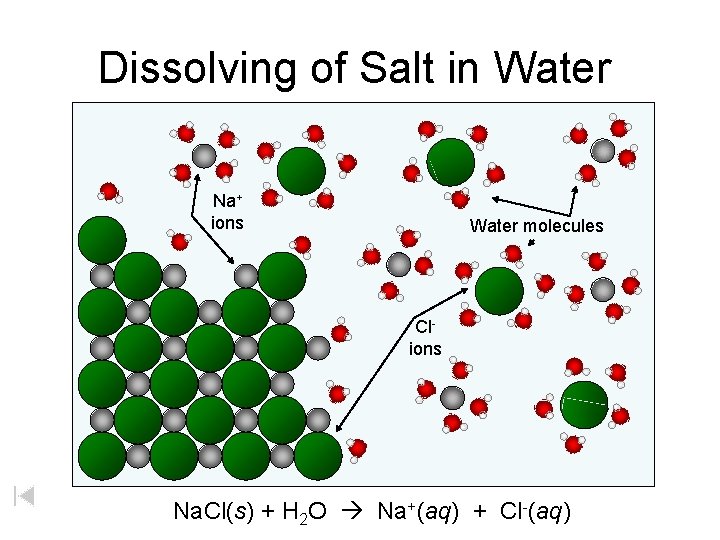
Dissolving of Salt in Water Na+ ions Water molecules Clions Na. Cl(s) + H 2 O Na+(aq) + Cl-(aq)

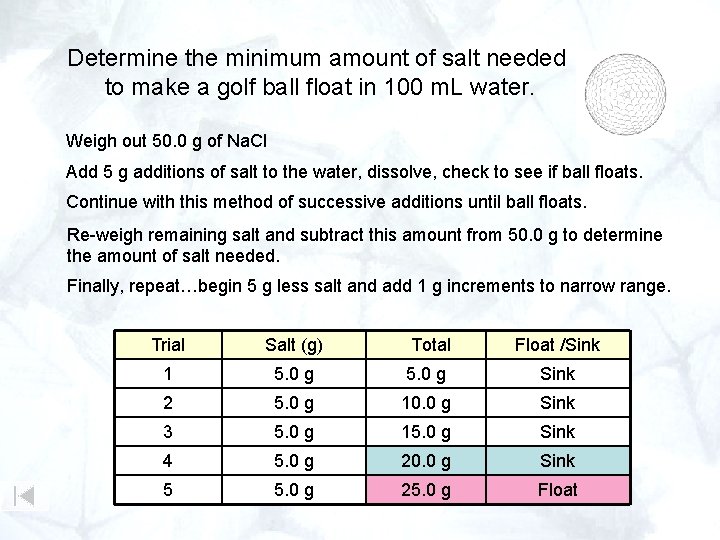
Determine the minimum amount of salt needed to make a golf ball float in 100 m. L water. Weigh out 50. 0 g of Na. Cl Add 5 g additions of salt to the water, dissolve, check to see if ball floats. Continue with this method of successive additions until ball floats. Re-weigh remaining salt and subtract this amount from 50. 0 g to determine the amount of salt needed. Finally, repeat…begin 5 g less salt and add 1 g increments to narrow range. Trial Salt (g) Total Float /Sink 1 5. 0 g Sink 2 5. 0 g 10. 0 g Sink 3 5. 0 g 15. 0 g Sink 4 5. 0 g 20. 0 g Sink 5 5. 0 g 25. 0 g Float

Theorize, but Verify …We must trust in nothing but facts. These are presented to us by nature and cannot deceive. We ought in every instance to submit our reasoning to the test of experiment. It is especially necessary to guard against the extravagances of imagination which incline to step beyond the bounds of truth. Antoine Laurent Lavoisier, 1743 - 1794 Jaffe, New World of Chemistry, 1955, page 1

Theory Guides, Laboratory Decides! Density of water = 1. 0 g/m. L Need to determine density of a golf ball. mass =______ g (electronic balance) volume = ______ m. L (water displacement method) or formula? Density of golf ball cannot be made to decrease. Therefore, you need to increase the density of the water by dissolving salt into the water. Limiting Factor: accurate determination of volume of golf ball Solubility Curve of salt in water. Water has a limit to how much salt can be dissolved. Saturation – point at which the solution is full and cannot hold anymore solute.

Packing of Na. Cl Ions Electron Microscope Photograph of Na. Cl

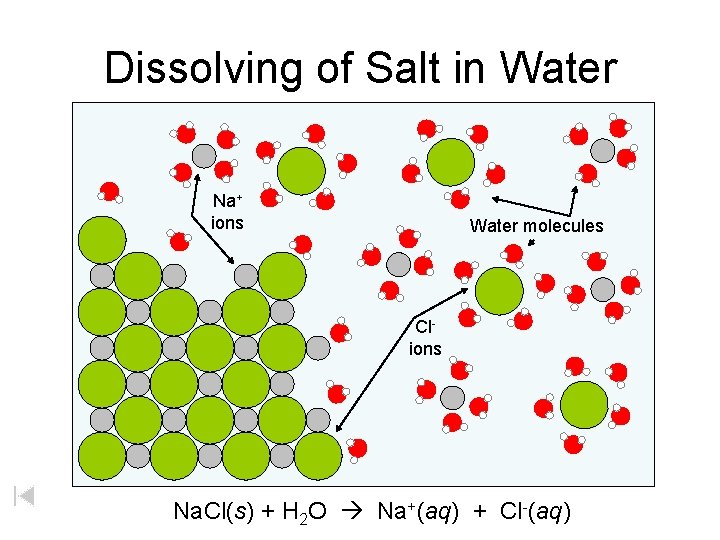
Dissolving of Salt in Water Na+ ions Water molecules Clions Na. Cl(s) + H 2 O Na+(aq) + Cl-(aq)

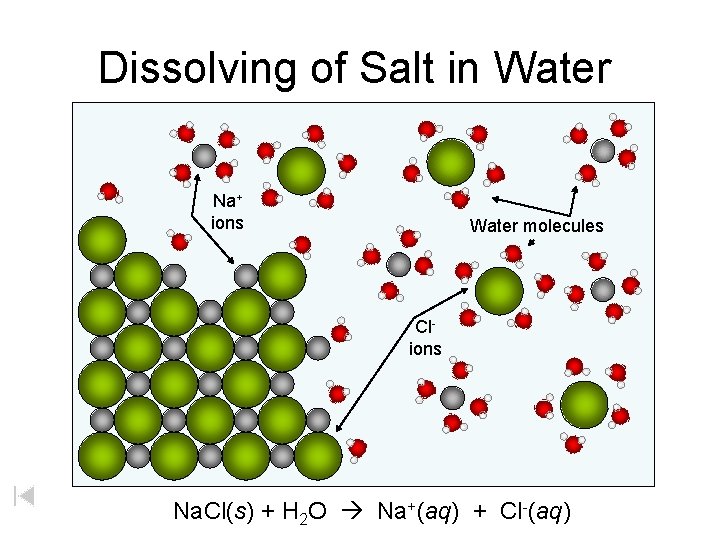
Dissolving of Salt in Water Na+ ions Water molecules Clions Na. Cl(s) + H 2 O Na+(aq) + Cl-(aq)

Dissolving of Salt in Water Na+ ions Water molecules Clions Na. Cl(s) + H 2 O Na+(aq) + Cl-(aq)

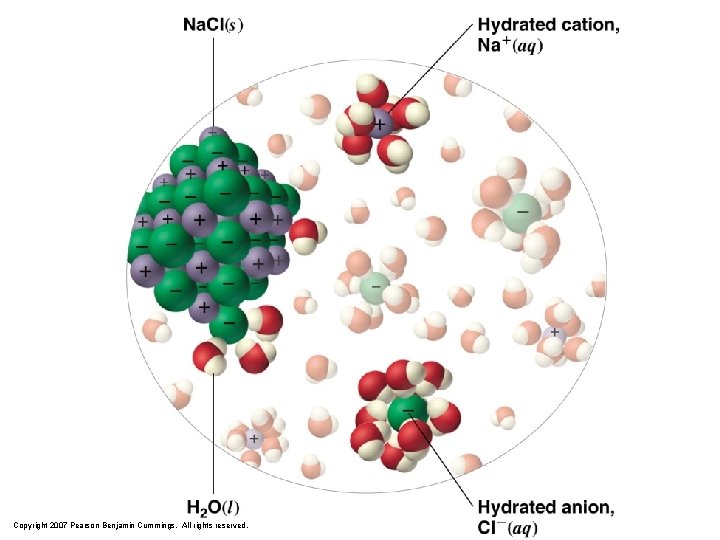
Copyright 2007 Pearson Benjamin Cummings. All rights reserved.

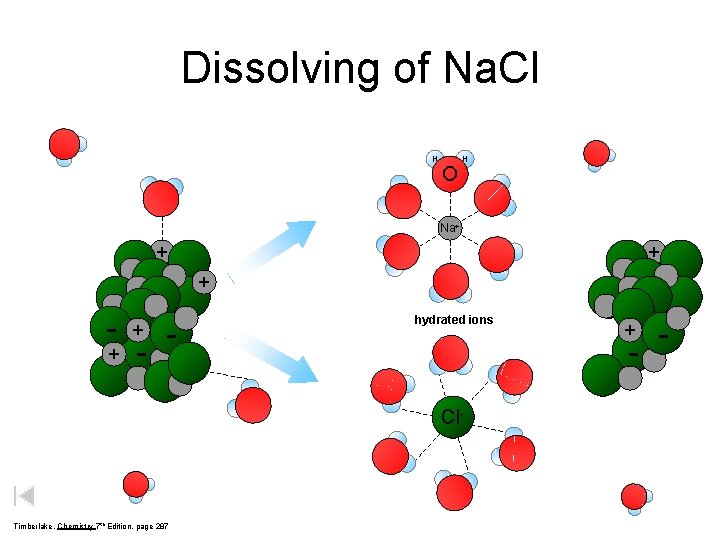
Dissolving of Na. Cl O H H Na+ hydrated ions - Cl - + + + - + + Timberlake, Chemistry 7 th Edition, page 287

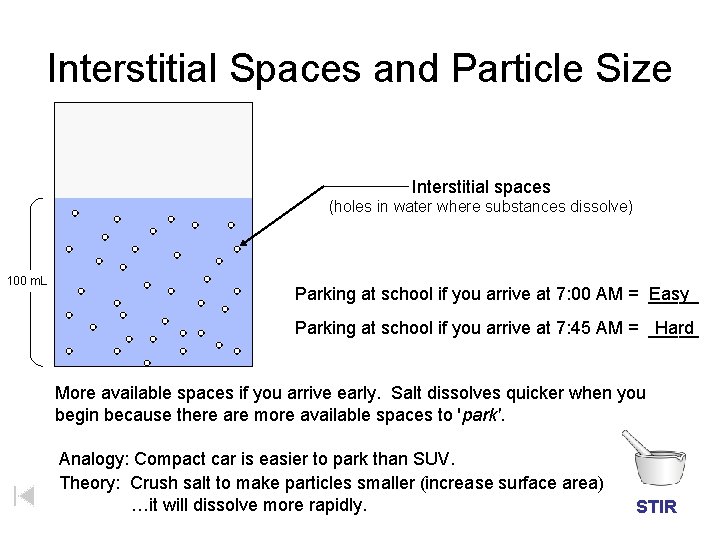
Interstitial Spaces and Particle Size Interstitial spaces (holes in water where substances dissolve) 100 m. L Parking at school if you arrive at 7: 00 AM = _____ Easy Parking at school if you arrive at 7: 45 AM = _____ Hard More available spaces if you arrive early. Salt dissolves quicker when you begin because there are more available spaces to 'park'. Analogy: Compact car is easier to park than SUV. Theory: Crush salt to make particles smaller (increase surface area) …it will dissolve more rapidly. STIR

100 m. L of water = 100 g density of water= 1. 00 g/m. L You determine the density of golf ball to be 1. 18 g/m. L Add 19 g salt to 100 g water = 119 g salt + water Volume remains 100 m. L (saltwater) Density = Mass volume or 119 g 100 m. L Density (saltwater) = 1. 19 g/m. L If golf ball doesn’t float, add 2 m. L additions of salt until it floats. Add 3. 0 m. L water, stir…float Add 3. 0 m. L water, stir…sink

Goals and Objectives: a. Given materials and problem, formulate and test a hypothesis to determine if a golf ball can float in salt water. b. Collect accurate data and compare own data to other class data. Evaluate own results. Investigation Procedure: a. Design an experiment to accurately determine how dense salt water must be in order for a golf ball to float. Use metric units. Be sure to control as many variables as possible. b. Write down the procedure that you and your partner(s) are going to use prior to lab day. Record any researched facts that may be useful in knowing before conducting your experiment. c. Carefully run your experiment, make observations and record your measurements in a data table. Use grams and milliliters in your measurements. Include a calculation column in your data table. d. Critique your own procedure, discuss and compare your process with another group, then modify your own steps as needed. e. Repeat your experiment to check for accuracy, if time allows.

Discussion Questions for Understanding: a. How did you determine the density of your golf ball? b. Why does a golf ball normally sink to the bottom of a pond at the golf course? c. What variables were difficult or impossible for you to control during this experiment? How much salt can be dissolved in 100 m. L of water? (saturated) effect of temperature on solubility Surface area of salt may affect rate of dissolving (may need to crush salt finely) d. What variables may have changed as time went on that could have affected the outcome of your results? e. Did you improve the accuracy of your results after conferring with another group? f. Describe your sources of error. (Human error and faulty equipment are unacceptable answers)

Materials: electronic balance mortar / pestle golf ball 250 m. L beaker 100 m. L & 500 m. L graduated cylinder glass stirring rod salt (Kosher, iodized table salt, table salt) Extension: a. Research the manufacturing of golf balls to determine why they sink in pond water. b. Research to determine which body of salt water in the world would float a golf ball the highest. Lab Report : (10 - 12 point font two page maximum length) Background / problem Hypothesis (if. . . then) Procedure (protocol) Data (table, graph) Analysis Conclusions / Future directions (limitations) Sample calculations - Appendix Do not use references to yourself or others in your writing of a lab report (except for citing past research). OR Poster (25 words or less) A picture is worth 1000 words!