10 4 Energy Levels of Electrons Electrons move





















![Example for you to try • You try Ba 2 • Ba = [Xe] Example for you to try • You try Ba 2 • Ba = [Xe]](https://slidetodoc.com/presentation_image_h2/196322404eacee79071414ec38f34c30/image-25.jpg)
- Slides: 25

10. 4 Energy Levels of Electrons • Electrons move in definite energy levels; these are labeled 1 - 7 • Each level has sublevel(s) which are probability shapes that show where the electrons may be at any one time. Also known as orbitals. • S orbital can hold up to 2 electrons (0, 1, 2) • p orbital can hold up to 6 electrons (0 -6) • d orbital can hold up to 10 electrons (0 -10) • f orbital can hold up to 14 electrons (0 -14) • Aufbau chart shows how electrons fill into the main energy levels and the sublevels or orbitals

Energy Levels and Sublevels • • 1 s 2 s 3 s 4 s 5 s 6 s 7 s 2 p 3 p 4 p 5 p 6 p 7 p 3 d 4 d 5 d 6 d 7 d 4 f 5 f 6 f 7 f

Aufbau Diagram or Chart • • 1 s 2 s 3 s 4 s 5 s 6 s 7 s START HERE and follow 2 p the arrows! 3 p 3 d 4 p 4 d 4 f 5 p 5 d 5 f 6 p 6 d 6 f 7 p 7 d 7 f

Electron Configuration Be • 1 s 2 2 s 2 • 15 P • 1 s 22 p 63 s 23 p 3 • 25 Mn • 1 s 22 p 63 s 23 p 64 s 23 d 5 • 4

Pauli Exclusion Principle • Pauli exclusion principle states that no more than 2 electrons can be in the same suborbital. Even so, this would cause them to have precisely the same quantum address. So Pauli decided there has to be a way to tell one electron from another. In other words, they must differ by at least one quantum number!

Pauli Exclusion Principle • So they invented spin (+1/2 or -1/2) called spin up and spin down. Has nothing to do with the direction of the electron--we don’ t know how they move just where they may be at with 90% chance of finding it inside the energy level and orbital designated.

Hund’s Rule • Hund’s rule states that electrons fill unpaired until there is no more room then they will pair (applies to p, d and f orbitals)

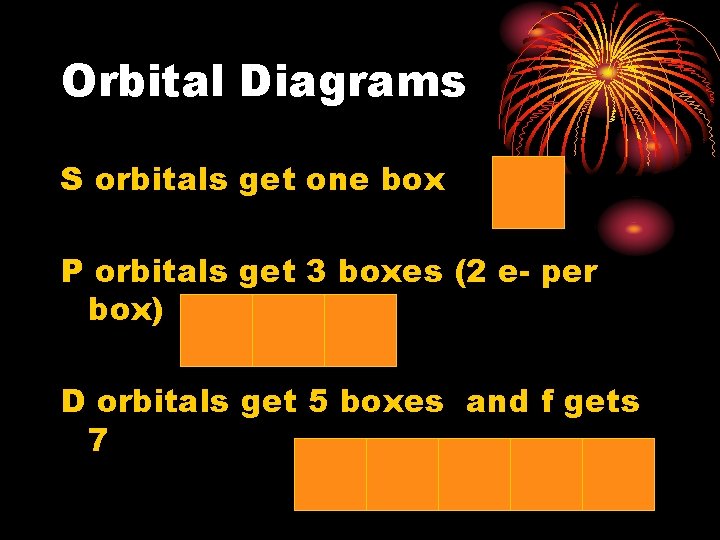
Orbital Diagrams S orbitals get one box P orbitals get 3 boxes (2 e- per box) D orbitals get 5 boxes and f gets 7

Orbital Diagrams (cont. ) • Insert electrons (using arrows into each box according to Hund’s and Pauli) 2 p 3

Answer • 2 p 3 (arrows can all point up or down) • Now try 4 f 10

Answer to 10 4 f • Arrows may point up or down if they are in boxes individually; however, if there are 2 electrons in a box, one must point up and one down.

Electrons and the Periodic Table Revisited History of the Table Periodic Law Important People

Mendeleev • Mendeleev was a Russian chemist who arranged the known elements in vertical columns in order of increasing mass and noticed a pattern in physical and chemical properties

Mosley • Mosley was a British physicist who determined the atomic number (number of protons) of the atoms of elements and then arranged the elements according to their atomic number.

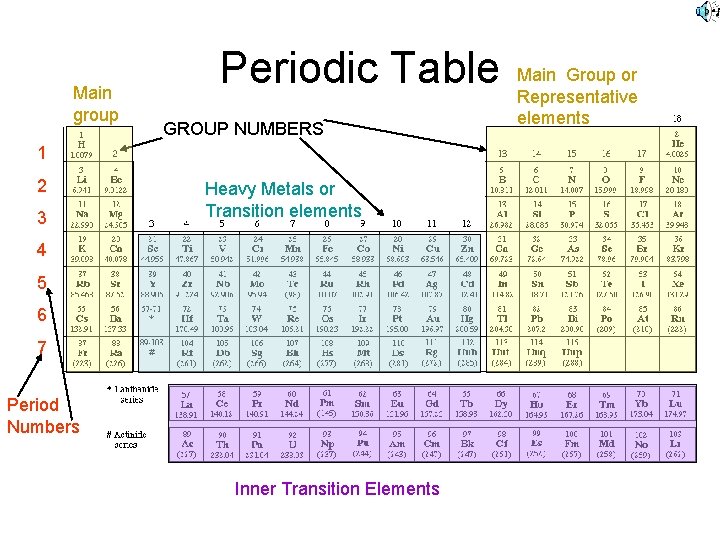
Periods and Groups • Periods of the periodic table are the rows across • Groups or Families are columns on the periodic table. • Currently we have 18 groups. We will use the 1 -18 designations not the A/B or Roman Numerals

Areas of the Periodic Table • Representative elements or Main Group are those that are in Groups 1, 2, 13, 14, 15, 16, 17, 18 • Transition elements are Groups 3 - 12 , also called the Heavy Metals

Inner Transition • Rare Earth elements that are located in the bottom two rows (away from the rest of the table) of the periodic table

Main group Periodic Table GROUP NUMBERS 1 2 3 Heavy Metals or Transition elements 4 5 6 7 Period Numbers Inner Transition Elements Main Group or Representative elements

Groups with names • • Group 1 = Alkali Metals 2 = Alkaline Earth Metals 18 = Inert or Noble Gases 17 = Halogens

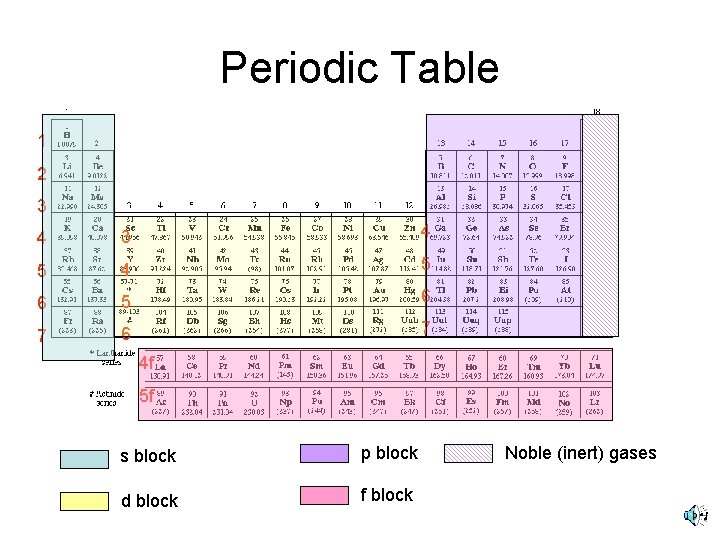
Periodic Table and Electron Configuration • The light metals compose the s block. • The transition elements are the d block. • The nonmetals are p block. • The inner transition (rare earth) metals are the f block.

Periodic Table 1 2 3 4 5 4 5 6 7 6 7 4 f 5 f s block p block d block f block Noble (inert) gases

• • Complete the electron configurations for the Noble Gases (Hint: Group 18) • Xe = He 1 s 2 Ne 1 s 22 p 6 Ar = 1 s 22 p 63 s 23 p 6 Kr = 1 s 22 p 63 s 23 p 64 s 23 d 1 04 p 6 1 s 22 p 63 s 23 p 64 s 23 d 10 4 p 65 s 24 d 105 p 6 • Rn = 1 s 22 p 63 s 23 p 64 s 23 d 10 4 p 65 s 24 d 105 p 66 s 24 f 145 d 1 06 p 6 Except He, do you see a trend in all of the noble gas configurations? • What do they all end in?

Shorthand Notation • We use the noble gases in shorthand notation • Find the closest noble gas that has an atomic number LESS than that of the element

Example • • Ex. K What is K’s atomic number? 19 Closest noble gas? Ar What is Ar’s atomic number? 18 = 1 s 22 p 63 s 23 p 6 = [Ar] 4 s 1 = Means the first 18 electrons are arranged like argon and the last electron is called the VALENCE ELECTRON (outermost shell)
![Example for you to try You try Ba 2 Ba Xe Example for you to try • You try Ba 2 • Ba = [Xe]](https://slidetodoc.com/presentation_image_h2/196322404eacee79071414ec38f34c30/image-25.jpg)
Example for you to try • You try Ba 2 • Ba = [Xe] 6 s • Try Pb • Pb = [Xe] 6 s 24 f 145 d 106 p 2