BONDING VALENCE ELECTRONS Highest energy electrons outer shell

BONDING

VALENCE ELECTRONS � Highest energy electrons (outer shell) � Only s and p block elements contain valence electrons, so how many valence electrons can there be? ? ? � Noble gas=most stable � How � Octet many electrons? 8 Rule: 8 valence electron’s are most stable. � Elements will find the easiest route to get 8 electrons (either gain or lose electrons)

LEWIS DOT STRUCTURES � Lewis Dot Structures/ Structural Formulas � Write the atomic symbol; place the appropriate number of dots around the element to show the valence electrons. � C, � Ionic K, Cl, Kr Compounds � Write the elements that are bonding, place appropriate number of valence dots, use an arrow to show where the electrons get traded, end equation with ions, one complete, one empty element � Example: Na. Cl, Ca. Br 2

LEWIS DOT STRUCTURES/COVALENT CMPD � Covalent Compounds � Lewis Dot: same concept as ionic, but electrons do not transfer � Structural Formulas: place a line in between bonded elements, these lines represent paired electrons, left over dots are unpaired electrons. � Number � Single of bonds: bond: one line, one pair of electrons � Double bond: two lines, two pairs of electrons � Triple Bond: three lines, three pairs of electrons

LEWIS DOT STRUCTURES/COVALENT CMPD � Rules of writing Covalent Compounds: � � � Count valence electrons (add for negative charges, subtract for positive charges) write skeleton with single bonds already in place Place remaining valence electrons around elements make stable octet, increase bond number if needed E. G. : CO 2, N 2, CH 4 Exceptions: � � H, Li satisfied with 2 electrons Boron family satisfied with 6 electrons (metals) P, S more than 8 electrons is OK When one element is satisfied, but partner is not, the satisfied element with donate a pair of electrons to make another bond = Coordinate Covalent Bonds

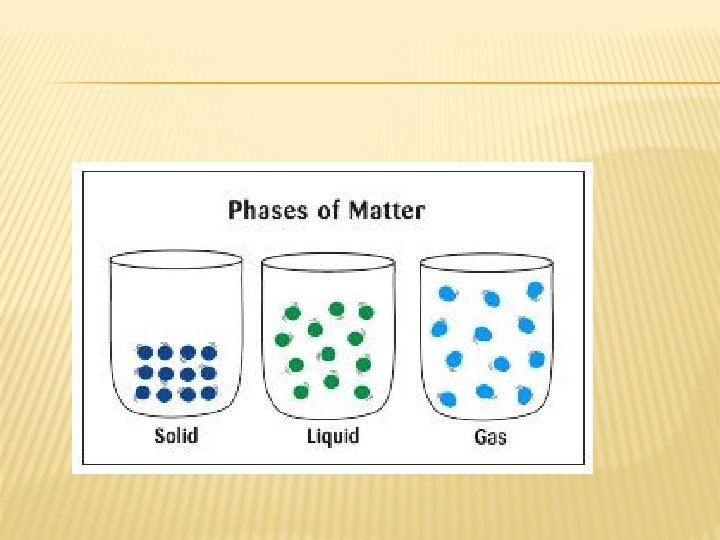
Solids, liquids, and gases STATES OF MATTER

KINETIC-MOLECULAR THEORY Particles are hard spheres with insignificant volume, mostly empty space, no attractive or repulsive forces � Particles in constant, rapid motion � All collisions are perfectly elastic �

SOLIDS � Particles vibrate about fixed points � Highly organized, dense, and incompressible � Do NOT take shape of container

LIQUIDS � Particles are in motion and are free to slide past one another � Vibrate and spin as they move � Particles are attracted to each other through intermolecular forces � Holds shape of container � Less dense than a solid � Not enough energy to escape as a gas

GASES � Particles � Take are in constant motion shape of container � Less dense than solids or liquids � Forces of attraction are weaker
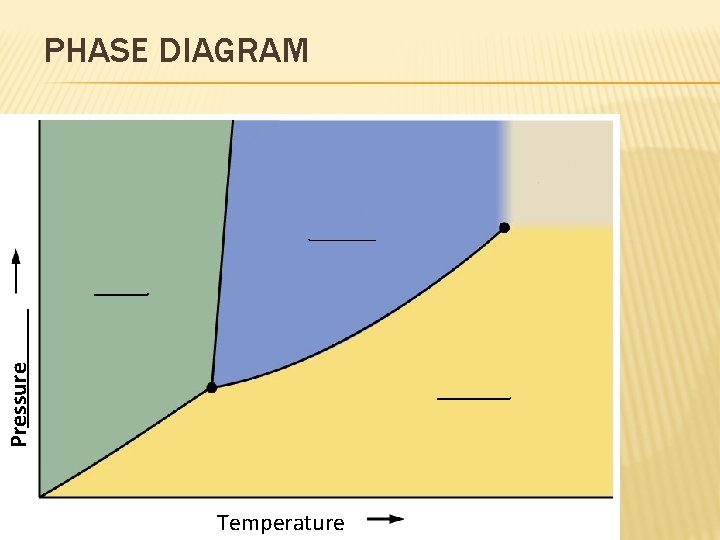
Pressure PHASE DIAGRAM Temperature

TRIPLE POINT VIDEO � http: //www. youtube. com/watch? v=BLRqp. JN 9 ze. A
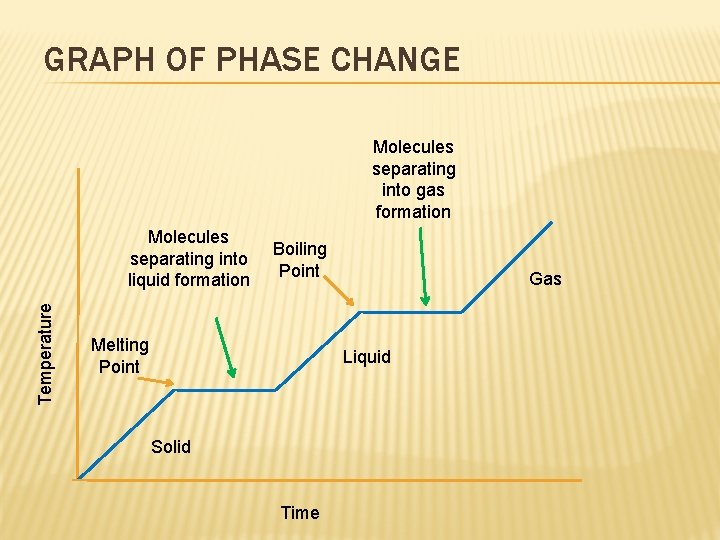
GRAPH OF PHASE CHANGE Molecules separating into gas formation Temperature Molecules separating into liquid formation Boiling Point Melting Point Gas Liquid Solid Time

BOND POLARITY

BOND POLARITY � Covalent bonds differ in how bonded atoms share electrons � � Non-polar covalent bond: � � Electrons are being pulled by both nuclei Bonds are pulled equally (usually between two like atoms) H 2 Polar Covalent Bonds: Two different atoms bond and electrons are shared unequally � More electronegative atoms will have the stronger electron attraction, will acquire a slightly negative charge (Example) � Less electronegative atom will acquire a slightly positive charge(Example) � Charges are represented by lower case delta (the atoms charges are less than +1 and -1) (Example) �

DETERMINING POLAR MOLECULES �A molecule is polar when the atoms do not share electrons equally � This creates a slightly positive charge on one end and slightly negative charge on the other � Look for: � Unshared electrons on the central atom (called lone pairs) � Different atoms surrounding the central atom � Examples: � What NH 3 about H 2 O? CH 2 Cl 2

Ionic, metallic, and network OTHER TYPES OF BONDS
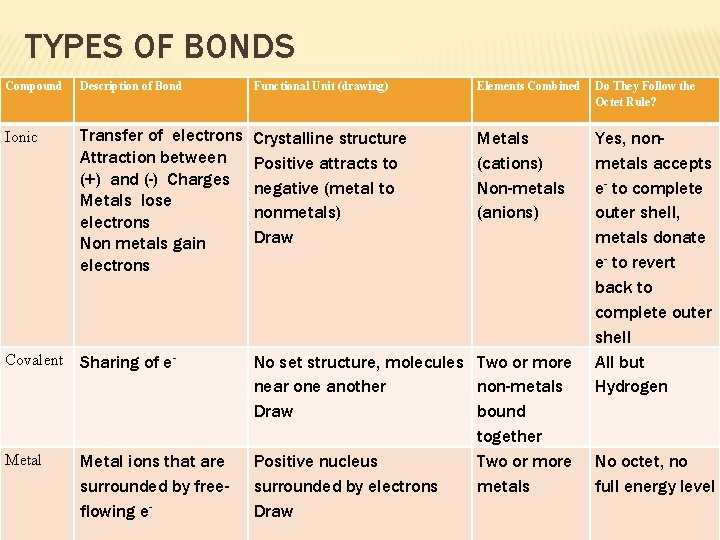
TYPES OF BONDS Compound Description of Bond Functional Unit (drawing) Elements Combined Do They Follow the Octet Rule? Ionic Transfer of electrons Attraction between (+) and (-) Charges Metals lose electrons Non metals gain electrons Crystalline structure Positive attracts to negative (metal to nonmetals) Draw Metals (cations) Non-metals (anions) Covalent Sharing of e- Metal ions that are surrounded by freeflowing e- No set structure, molecules Two or more near one another non-metals Draw bound together Positive nucleus Two or more surrounded by electrons metals Draw Yes, nonmetals accepts e- to complete outer shell, metals donate e- to revert back to complete outer shell All but Hydrogen No octet, no full energy level
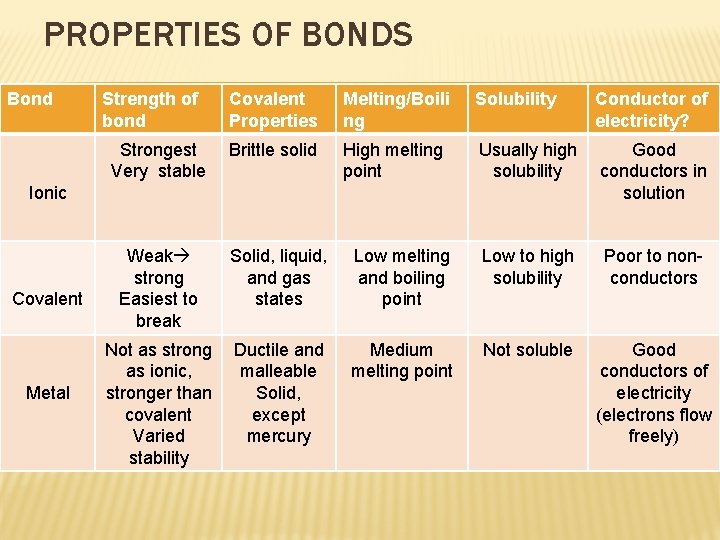
PROPERTIES OF BONDS Bond Strength of bond Strongest Very stable Covalent Properties Melting/Boili ng Solubility Conductor of electricity? Brittle solid High melting point Usually high solubility Good conductors in solution Ionic Covalent Metal Weak strong Easiest to break Solid, liquid, and gas states Low melting and boiling point Low to high solubility Poor to nonconductors Not as strong as ionic, stronger than covalent Varied stability Ductile and malleable Solid, except mercury Medium melting point Not soluble Good conductors of electricity (electrons flow freely)

FORCES

FORCES BETWEEN MOLECULES � Intermolecular forces � Attraction between molecules � Weaker than atom bonding � Used to determine physical properties of the molecule (such as state of the molecule solid, liquid, or gas) � Strongest Weak Ionic bond Hydrogen Bonds dipole Interaction Van der Waals
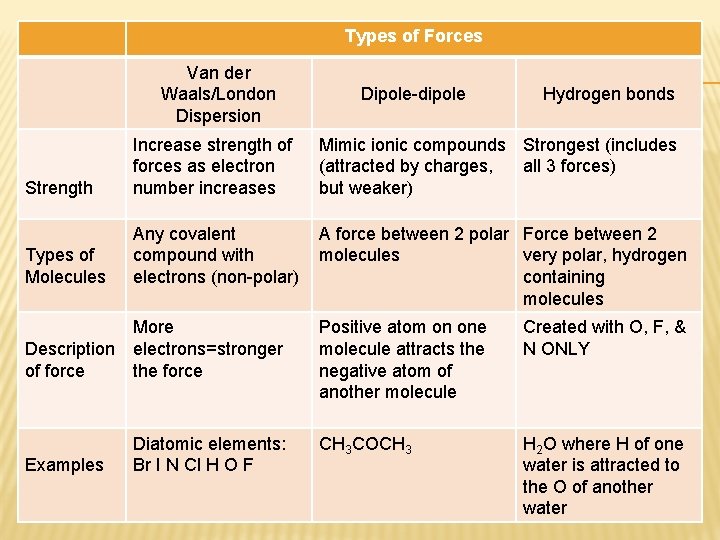
Types of Forces Van der Waals/London Dispersion Dipole-dipole Hydrogen bonds Strength Increase strength of forces as electron number increases Mimic ionic compounds Strongest (includes (attracted by charges, all 3 forces) but weaker) Types of Molecules Any covalent compound with electrons (non-polar) A force between 2 polar Force between 2 molecules very polar, hydrogen containing molecules More Description electrons=stronger of force the force Examples Diatomic elements: Br I N Cl H O F Positive atom on one molecule attracts the negative atom of another molecule Created with O, F, & N ONLY CH 3 COCH 3 H 2 O where H of one water is attracted to the O of another water

FORCES AND PROPERTIES � Physical properties depend on type of bond (ionic or covalent) � The variety in covalent compounds are due to intermolecular forces � Properties � Melting point: only the weak attractions need to be broken. � Boiling Point: break apart molecules of liquid � Solubility: stronger the intermolecular force the harder it is to be soluble.

QUESTIONS � Define the following: � Boiling point � Melting point � Evaporation � How do each of the above relate to the type of bond in a molecule? � What role does intermolecular forces play in the strength of bonds? � Draw Isopropyl Alcohol ((CH 3)2 CHOH), methanol (CH 3 OH), acetone (CH 3 COCH 3), and water.
- Slides: 25