1 Clip 3 Radioactivity is the spontaneous disintegration
















- Slides: 24

1 Clip

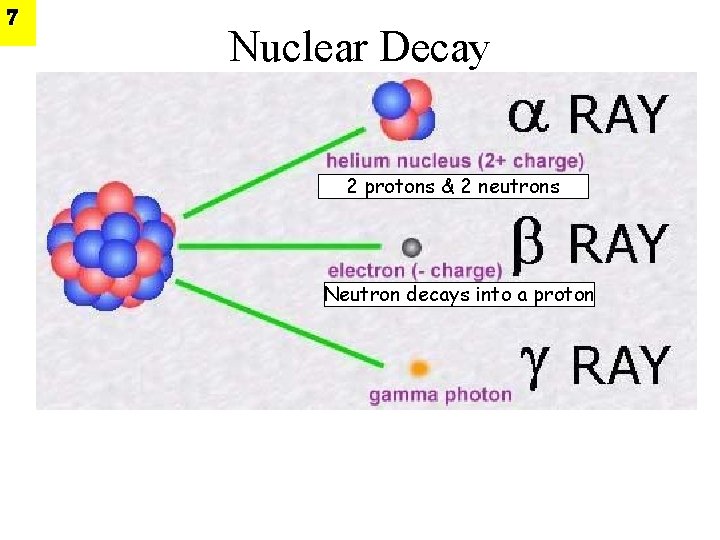
3 Radioactivity is the spontaneous disintegration of atomic nuclei. The nucleus emits α particles, ß particles, or electromagnetic rays during this process. After decaying, radioactive atoms “change” into other atoms Clip

4 • Why does the atom do this? – the nucleus of an atom attempts to become more stable • In some instances, a new element is formed and in other cases, a new form of the original element, called an isotope, appears. – this process of change is often referred to as the decay of atoms. • The rate of Radioactive decay is described in halflives

5 Energy is released during radioactive decay

6 Types of Nuclear Radiation • When an unstable nucleus decays, particles and energy are given off from the decaying nucleus. • α and β radiation is in the form of particles • γ radiation is in the form of waves-kind of like light but higher frequency

7 Nuclear Decay 2 protons & 2 neutrons Neutron decays into a proton

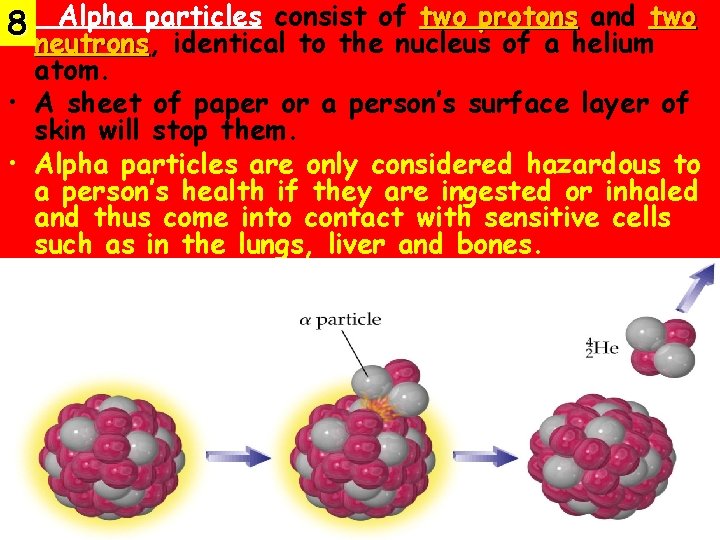
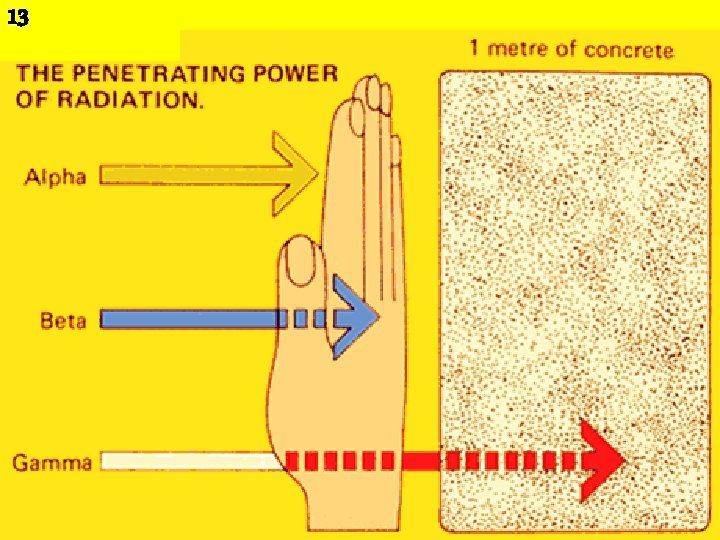
• Alpha particles consist of two protons and two 8 neutrons, neutrons identical to the nucleus of a helium atom. • A sheet of paper or a person’s surface layer of skin will stop them. • Alpha particles are only considered hazardous to a person’s health if they are ingested or inhaled and thus come into contact with sensitive cells such as in the lungs, liver and bones.

9

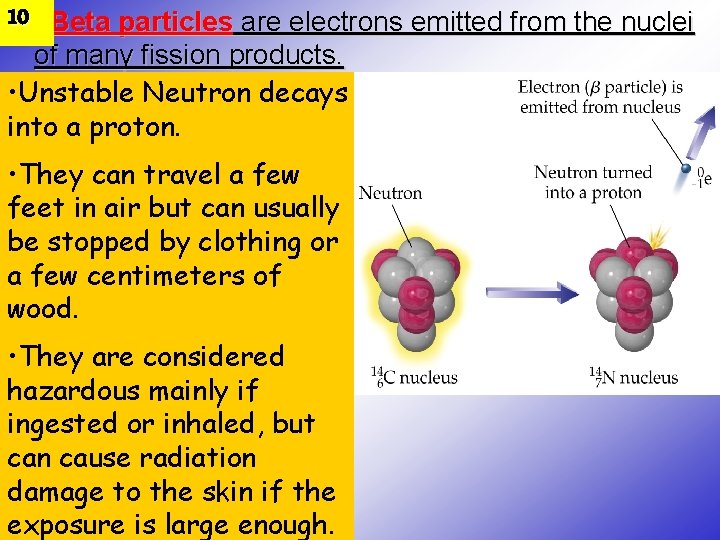
10 • Beta particles are electrons emitted from the nuclei of many fission products. • Unstable Neutron decays into a proton. • They can travel a few feet in air but can usually be stopped by clothing or a few centimeters of wood. • They are considered hazardous mainly if ingested or inhaled, but can cause radiation damage to the skin if the exposure is large enough.

11

• Gamma rays are a form of electromagnetic radiation (like light, radio, and television) that come from the nucleus of a radioactive atom. – Occurs when an unstable nucleus emits electromagnetic radiation. The radiation has no mass, and so its emission does not change the element. – They penetrate matter easily and are best stopped by water or thick layers of lead or concrete. – Gamma radiation is hazardous to people inside and outside of the body. • However, gamma radiation often accompanies alpha and beta emission, which do change the element's identity. • Gamma rays have the lowest ionizing power, but the highest penetrating power. 12

13

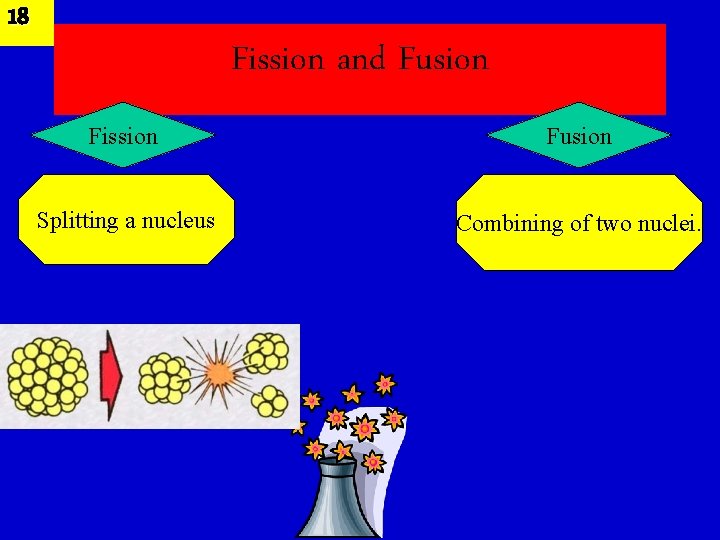
18 Fission and Fusion Fission Fusion Splitting a nucleus Combining of two nuclei.

19 • Nuclear power can come from the fission of uranium, plutonium or thorium or the fusion of hydrogen into helium. • Today it is almost all uranium. • The fission of an atom of uranium produces 10 million times the energy produced by the combustion of an atom of carbon from coal.

21 FUSION • A fusion reaction occurs when nuclei of light elements, specifically hydrogen and its isotopes (deuterium, or "heavy water, " and tritium), are forced together at extremely high temperatures and densities until they fuse into nuclei of heavier elements and release enormous amounts of energy. • If fusion is to yield net energy, the fuel must be heated in the form of plasma (a highly ionized gas) to a very high temperature and the plasma must then be held together for a sufficiently long time such that the number of fusion reactions occurring releases more energy than was required to heat the fuel.

Nuclear Isotope Notations

Nuclear Isotope Notations • Nuclear Isotope notation is used so we can keep track of the type of decay of an atom • Alpha and beta particles can be included in chemical equations to show nuclear radiation

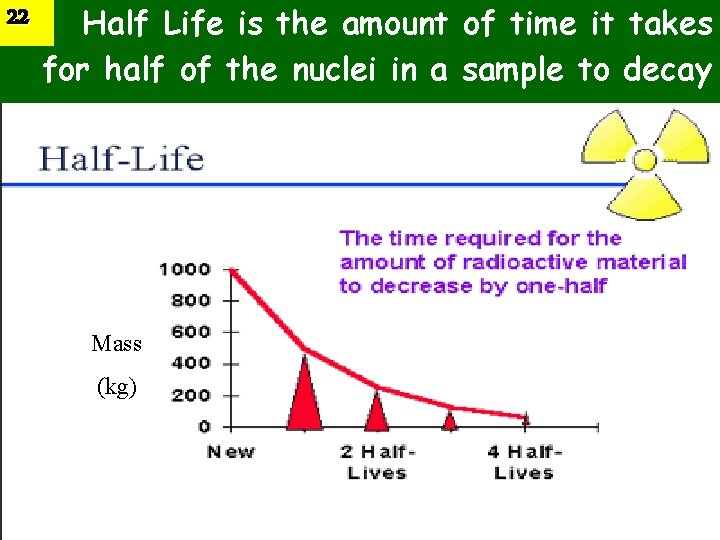
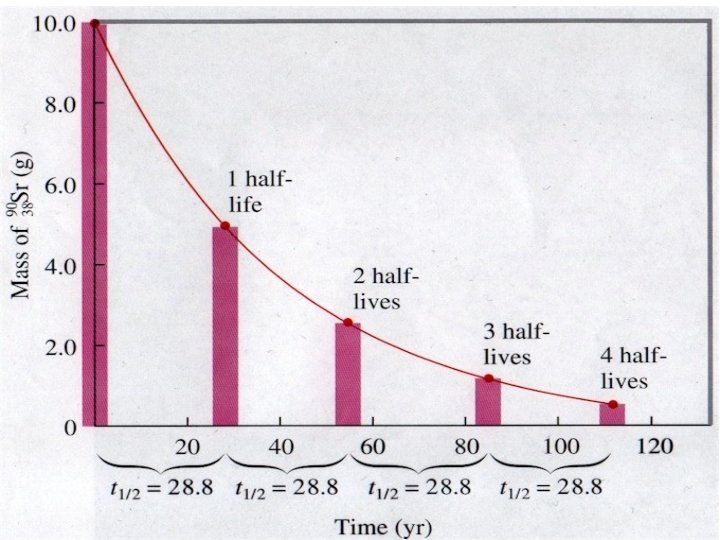
22 Half Life is the amount of time it takes for half of the nuclei in a sample to decay Mass (kg)


12 C mass number 14 C 13 C • Carbon occurs naturally in three isotopes. • All of these atoms have the same number of protons but different numbers of neutrons. • The number of neutrons and protons determines the mass, so the masses are different. • 14 C is radioactive.

14 C • Radioactive 14 C acts chemically just like 12 C, so it becomes incorporated into plants an animals. • When the animal/plant dies the 14 C begins to decay into 14 N at a know rate, so we can determine how long ago the organism died. • This is called Carbon Dating. • It’s only good for about 50, 000 years.

26 • The half life of 14 C is 5, 730 years. • If a sample originally contained 100 g, how much would be left after 11, 460 years? 50 g 25 g


Older Dating Methods • The isotopes 235 U and 238 U can be used to date objects billions of years old. • 235 U has a half life of 704 million years. • 238 U has a half life of 4. 5 billion years. • Mainly used for rocks.