Radioactivity The unstable nucleus Radioactivity Is the spontaneous

Radioactivity The unstable nucleus!!!!!

Radioactivity Is the spontaneous breaking up of an unstable nucleus with the emission of radiation

Radioactivity • Emission of energy from unstable nuclei which are trying to become stable • Natural effect - radioactive isotopes are found everywhere. • As the nucleus of an unstable atom breaks down, it gives out rays and particles called emissions. • The breaking down of unstable nuclei happens spontaneously – it is unaffected by heat, pressure, or whether the element is solid, liquid or gas

Revision of Atomic Structure • The Nucleus – This is the centre of the atom. It contains protons and neutrons. The mass of the atom is concentrated in the nucleus. • Protons – These have a positive charge and a mass of 1. • Neutrons – These don’t have a charge and have a mass of 1. • Electrons – These move around the nucleus. They have virtually no mass and have a negative charge.

Henri Becquerel • French scientist • Worked with Uranium • Saw how it effected photographic film left in the drawer of his desk. The uranium left an image of the crystals that it was composed of on the photographic film. • He is credited with the discovery of Radioactivity

Marie & Pierre Curie • Continued the work carried out by Becquerel on Uranium salts • Discovered Polonium and Radium (which are much more radioactive than Uranium) • Shared the Noble prize in Physics with her husband Becquerel in 1903 for the discovery of radioactivity • Her long term exposure to radiation results in health problems and she dies in 1934

The 3 types of radiation Alpha Radiation Gamma Radiation Beta Radiation

Types of particles

Alpha Radiation

Alpha Particles • Are positively charged particle, • Emitted by some radioactive nuclei, • That consists of two protons and two neutrons. • Alpha particles are the biggest type of particle can barely pass trough a piece of paper.

Alpha Radiation • 2 protons and 2 neutrons (= helium nucleus) ejected from the nucleus • Positive charge of +2 • Very high ionising power – this means it collides with lots of atoms and knocks electrons off them, making them ions • Short range in air – a few centimetres • Stopped by a piece of paper.
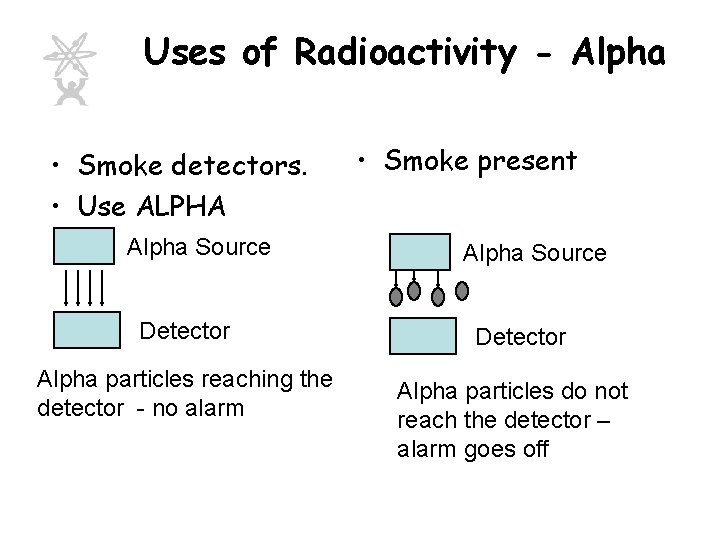
Uses of Radioactivity - Alpha • Smoke detectors. • Use ALPHA Alpha Source Detector Alpha particles reaching the detector - no alarm • Smoke present Alpha Source Detector Alpha particles do not reach the detector – alarm goes off

Beta Radiation

Beta Radiation • An neutron that breaks down into a proton and an electron • Negative charge of – 1 • Low ionising power • Stopped by a piece of aluminium foil

Uses of Beta Radiation Carbon-14 Dating: – All living things contain a fixed proportion of radioisotope Carbon-14 (14 C) – When animals and plants die, the proportion of 14 C starts to fall, because decaying 14 C is no longer being replaced by 14 C being taken in e. g. food – Scientists can work out the age of ancient organic substances e. g. . bones, by comparing the amount of 14 C left to the proportion in living organisms, and using halflife ITS Chemistry

Gamma Radiation ITS Chemistry

Gamma Radiation • Not made of protons or electrons • A high-energy electromagnetic wave • Emitted from nuclei changing from a high energy level to a lower one • The most penetrating atomic radiation – can travel huge distance through air • Stopped by several feet of lead ITS Chemistry
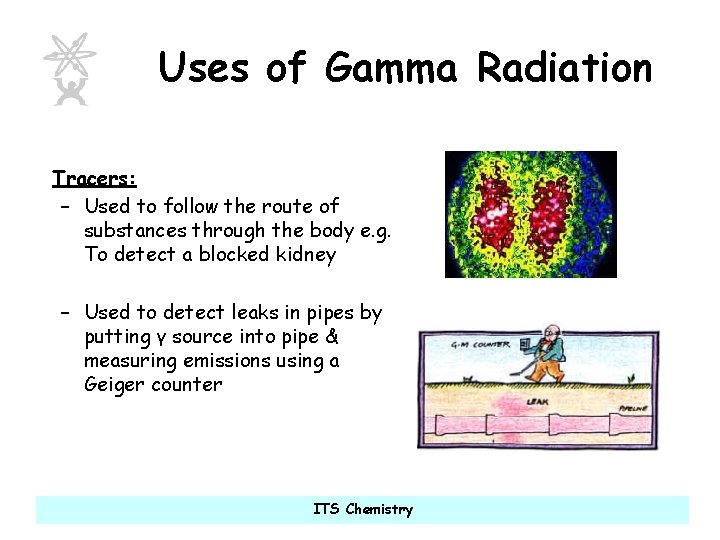
Uses of Gamma Radiation Tracers: – Used to follow the route of substances through the body e. g. To detect a blocked kidney – Used to detect leaks in pipes by putting γ source into pipe & measuring emissions using a Geiger counter ITS Chemistry

Uses of Gamma Radiation • Radiotherapy: – Cancer cells are exposed to gamma rays which kills them off – Makes the patient feel unwell – Correct dose vital – too much can kill healthy cells, too little won’t prevent spread of cancer • Sterilisation: – Civil use – γ radiation used to kill bacteria on some foods. Prolongs shelf-life but may change the taste. – Medical use – sterilises medical equipment that would be damaged by heat e. g. Syringes • Welding – Gamma emissions passed through metal onto photographic film, to check for bubbles ITS Chemistry
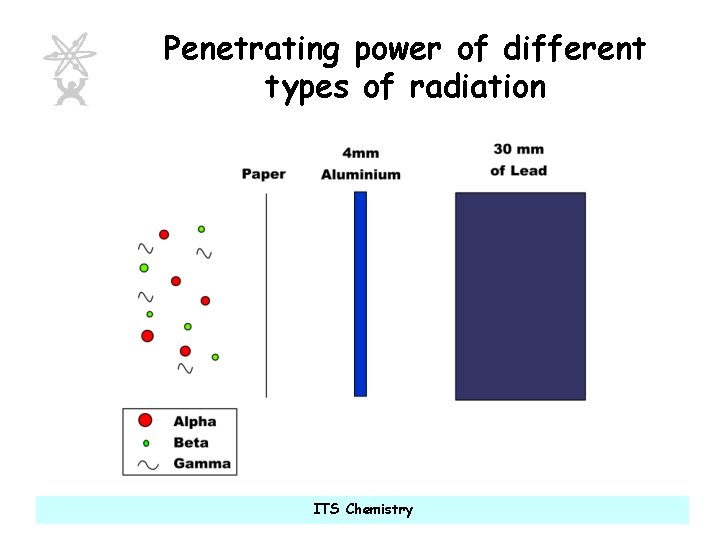
Penetrating power of different types of radiation ITS Chemistry

Penetrating power of different types of radiation ITS Chemistry

Detection of Radiation • Geiger counter • Photographic film • Cloud chamber ITS Chemistry

Nuclear Equations ITS Chemistry
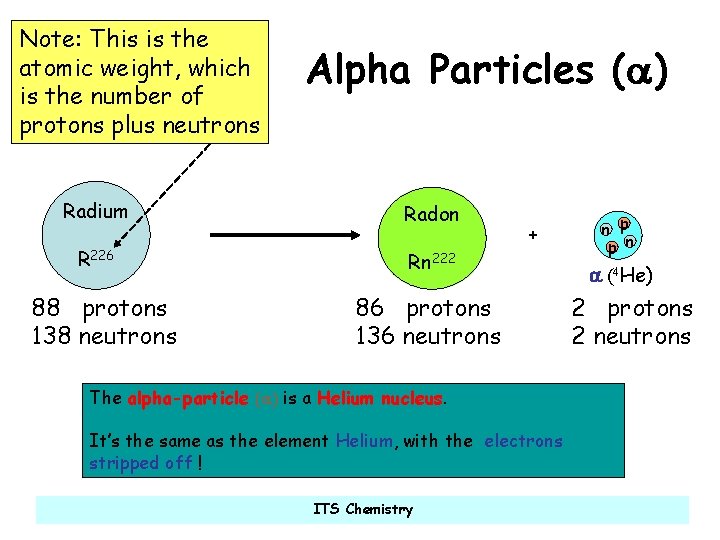
Note: This is the atomic weight, which is the number of protons plus neutrons Alpha Particles (a) Radium Radon R 226 Rn 222 88 protons 138 neutrons + 86 protons 136 neutrons The alpha-particle (a) is a Helium nucleus. It’s the same as the element Helium, with the electrons stripped off ! ITS Chemistry n p p n a (4 He) 2 protons 2 neutrons
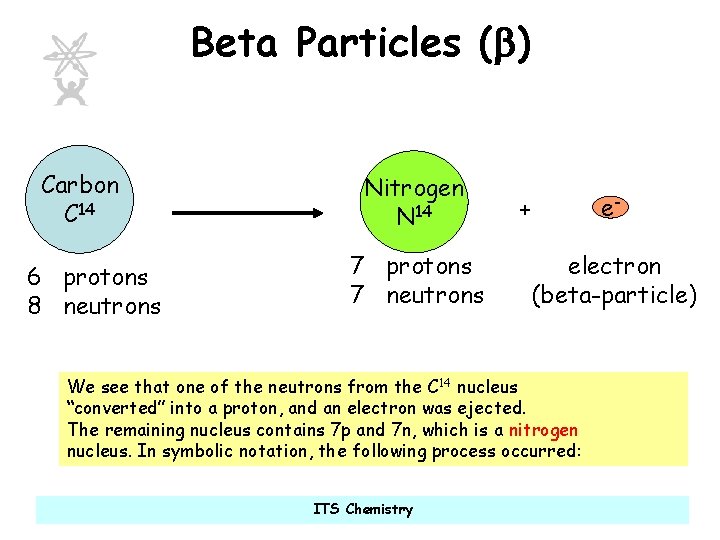
Beta Particles (b) Carbon C 14 6 protons 8 neutrons Nitrogen N 14 7 protons 7 neutrons + electron (beta-particle) We see that one of the neutrons from the C 14 nucleus “converted” into a proton, and an electron was ejected. The remaining nucleus contains 7 p and 7 n, which is a nitrogen nucleus. In symbolic notation, the following process occurred: ITS Chemistry e-
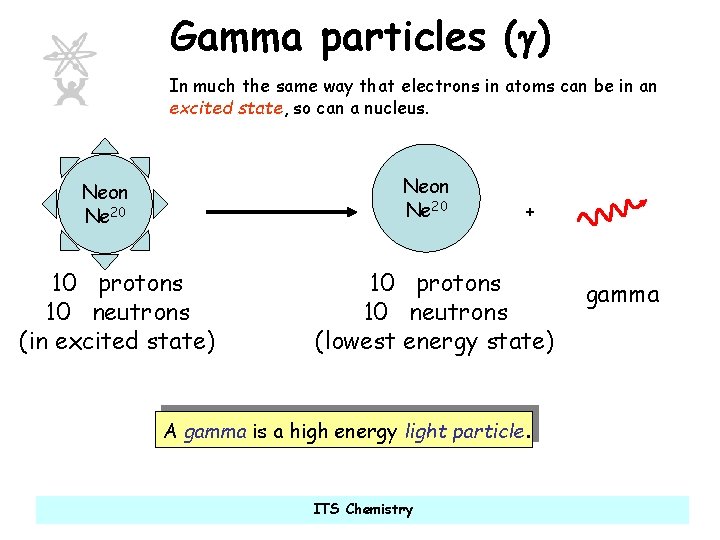
Gamma particles (g) In much the same way that electrons in atoms can be in an excited state, so can a nucleus. Neon Ne 20 10 protons 10 neutrons (in excited state) + 10 protons 10 neutrons (lowest energy state) A gamma is a high energy light particle. ITS Chemistry gamma
- Slides: 26