Welding Metallurgy 2 Welding Metallurgy 2 Lesson Objectives

Welding Metallurgy 2

Welding Metallurgy 2 Lesson Objectives When you finish this lesson you will understand: • The various region of the weld where liquid does not form • Mechanisms of structure and property changes associated with these regions Learning Activities 1. View Slides; 2. Read Notes, 3. Listen to lecture 4. Do on-line workbook 5. Do homework Keywords: Heat affected zone, Base metal, Solutionizing treatment, Aging, welding procedure, heat input, Hydrogen cracking, Carbon equivalent, Lamellar Tearing, Reheat Cracking, Knife-line attack,
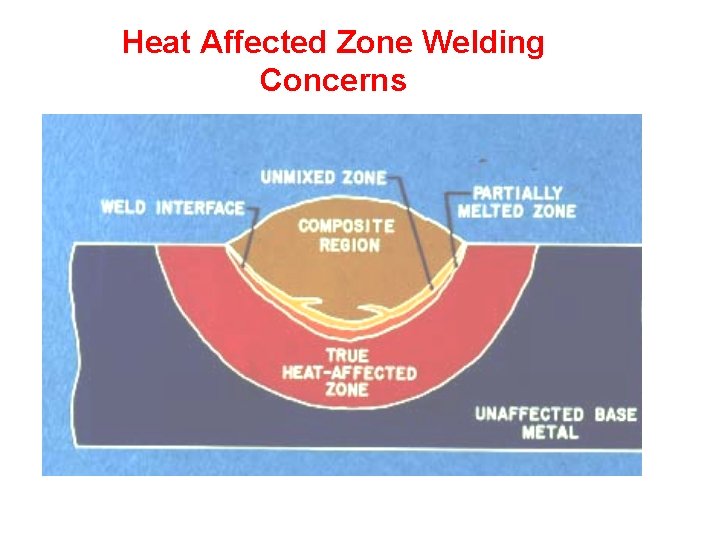
Heat Affected Zone Welding Concerns

Heat Affected Zone Welding Concerns • Changes in Structure Resulting in Changes in Properties • Cold Cracking Due to Hydrogen
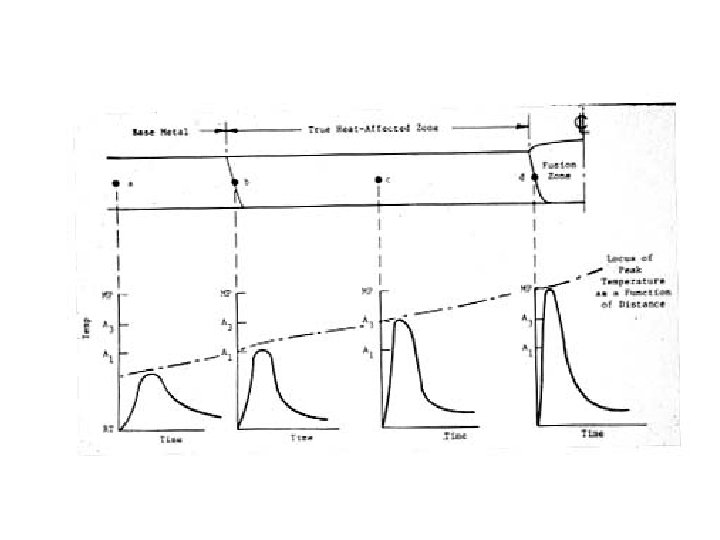

Look At Two Types of Alloy Systems
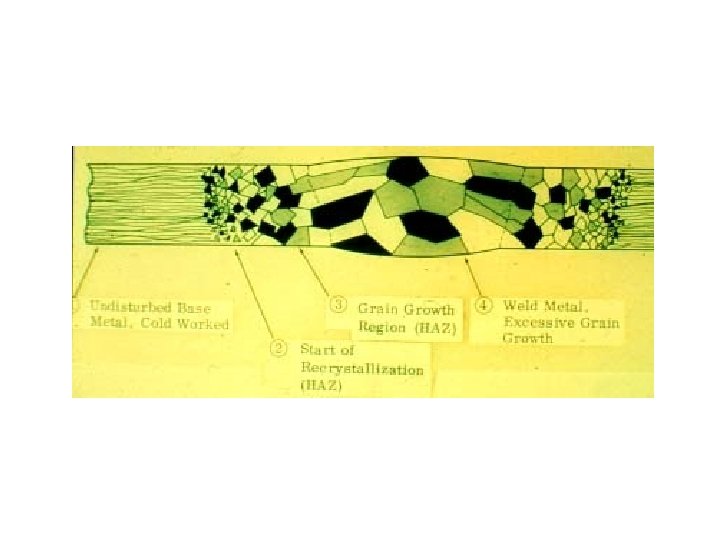
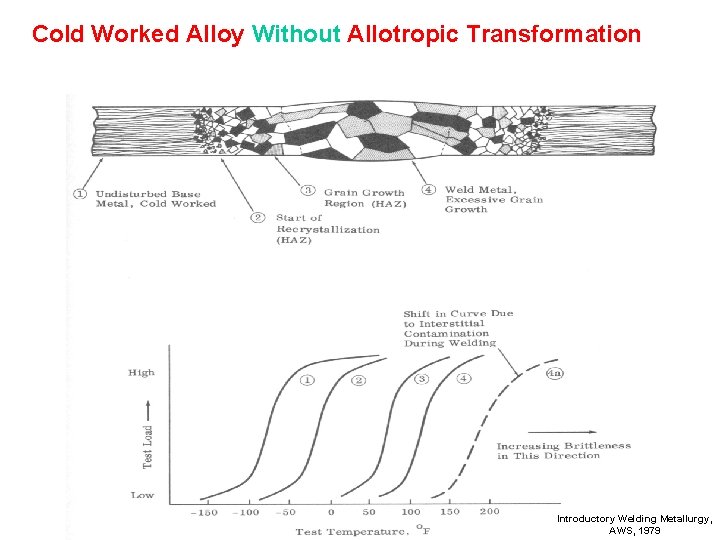
Cold Worked Alloy Without Allotropic Transformation Introductory Welding Metallurgy, AWS, 1979

Welding Precipitation Hardened Alloys Without Allotropic Phase Changes Welded In: • Full Hard Condition • Solution Annealed Condition Introductory Welding Metallurgy, AWS, 1979
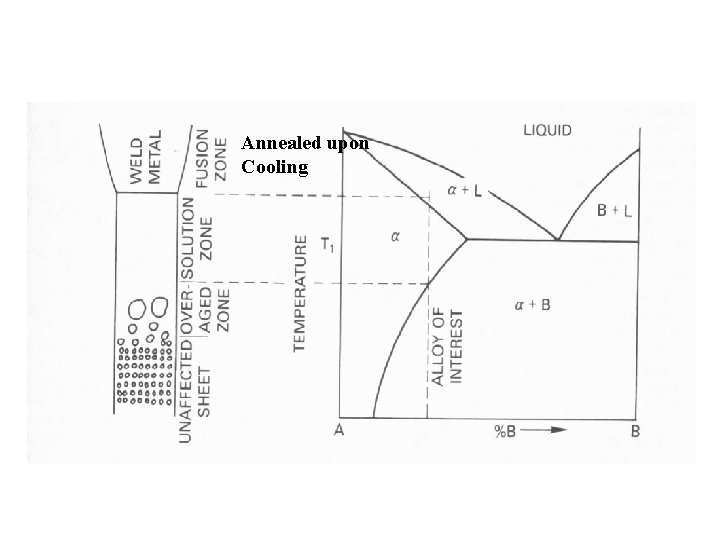
Annealed upon Cooling
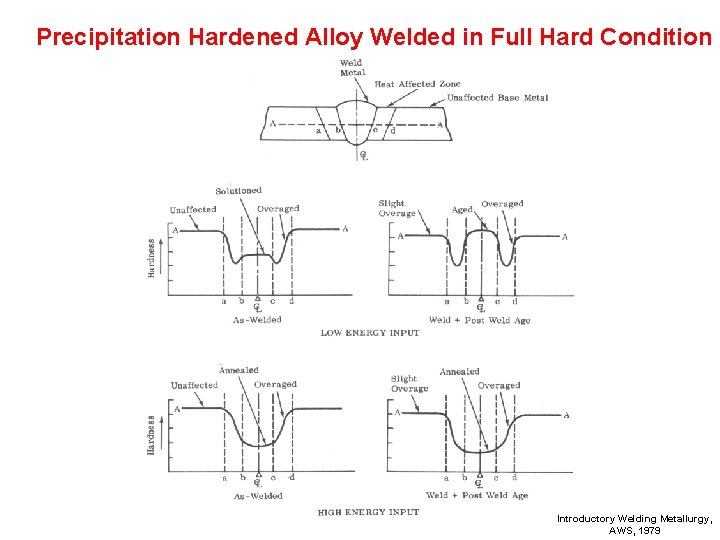
Precipitation Hardened Alloy Welded in Full Hard Condition Introductory Welding Metallurgy, AWS, 1979
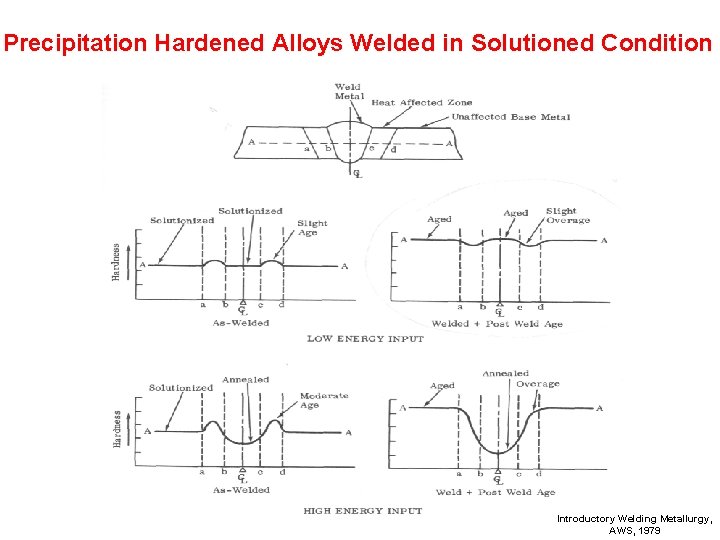
Precipitation Hardened Alloys Welded in Solutioned Condition Introductory Welding Metallurgy, AWS, 1979

Turn to the person sitting next to you and discuss (1 min. ): • Precipitation hardened austenitic stainless steel is used for high strength applications like rocket components etc. Reviewing the various procedures for welding precipitation hardened steels, what procedure would you recommend? Does it make any difference that this is austenitic stainless steel and not just plain carbon steel?
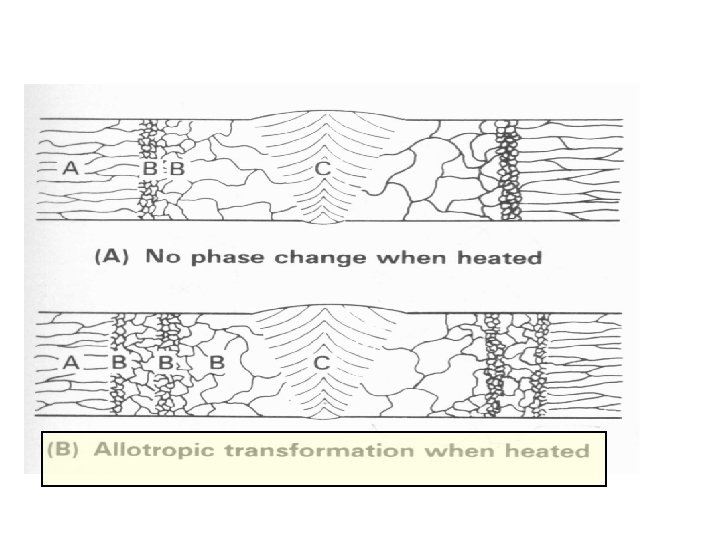
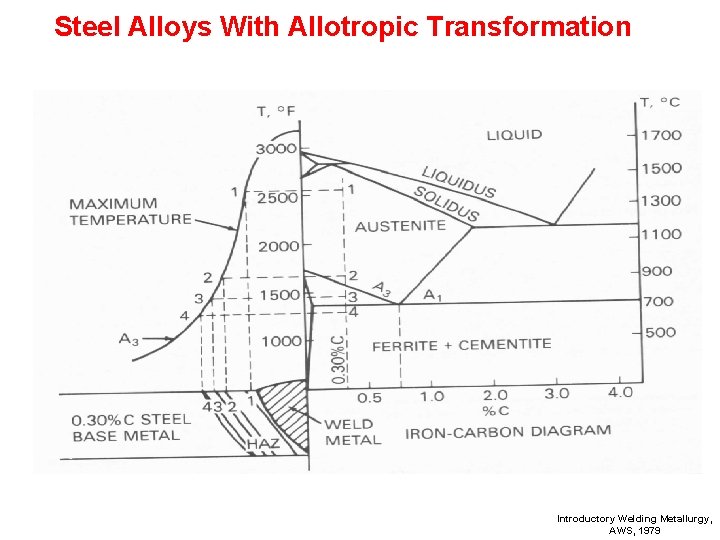
Steel Alloys With Allotropic Transformation Introductory Welding Metallurgy, AWS, 1979


Introductory Welding Metallurgy, AWS, 1979

Turn to the person sitting next to you and discuss (1 min. ): • As we saw, the cooling rate can depend upon the preheat and the heat input. Many codes actually specify the range of heat inputs that can be used to weld certain materials. We had an equation to determine the heat input before. What is it? What processes have the highest Heat Inputs? The lowest?

Cracking in Welds Hydrogen Cracking • Hydrogen cracking, also called cold cracking, requires all three of these factors – Hydrogen – Stress – Susceptible microstructure (high hardness) • Occurs below 300°C • Prevention by – Preheat slows down the cooling rate; this can help avoid martensite formation and supplies heat to diffuse hydrogen out of the material – Low-hydrogen welding procedure 0. 1. 1. 5. 2. T 12. 95. 12
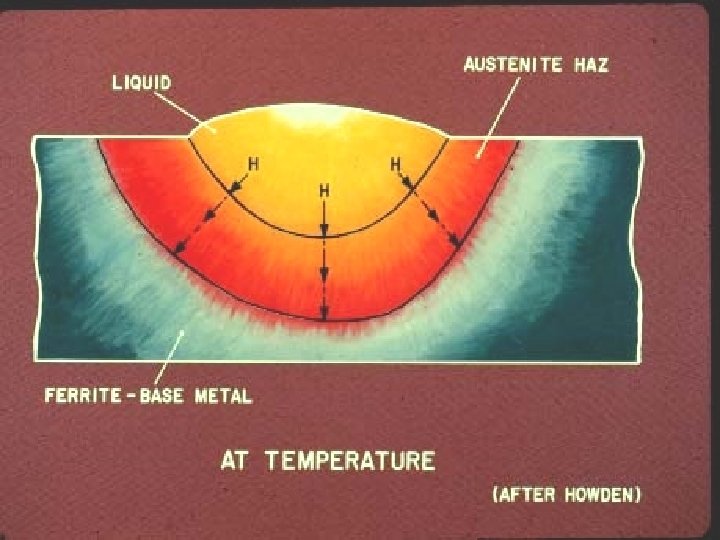

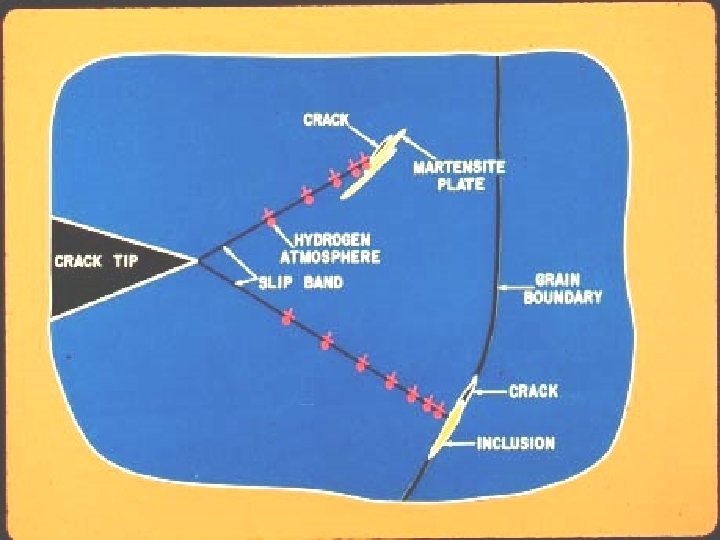

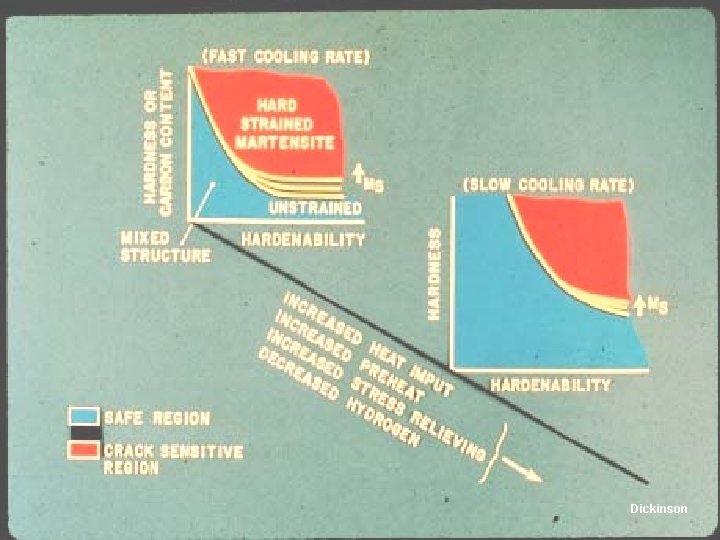
Dickinson

Carbon and Low-Alloy Steels Why Preheat? • Preheat reduces the temperature differential between the weld region and the base metal – Reduces the cooling rate, which reduces the chance of forming martensite in steels – Reduces distortion and shrinkage stress – Reduces the danger of weld cracking – Allows hydrogen to escape 0. 1. 1. 5. 1. T 9. 95. 12
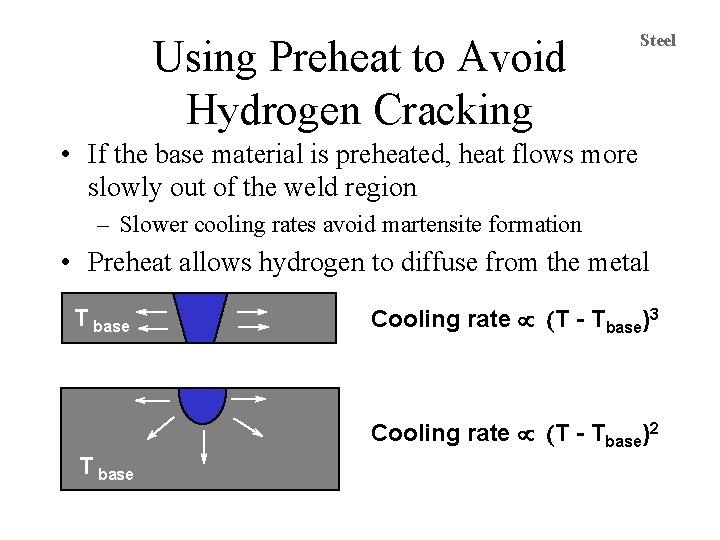
Using Preheat to Avoid Hydrogen Cracking Steel • If the base material is preheated, heat flows more slowly out of the weld region – Slower cooling rates avoid martensite formation • Preheat allows hydrogen to diffuse from the metal T base Cooling rate µ (T - Tbase)3 Cooling rate µ (T - Tbase)2 T base

Interaction of Preheat and Composition Steel CE = %C + %Mn/6 + %(Cr+Mo+V)/5 + %(Si+Ni+Cu)/15 • Carbon equivalent (CE) measures ability to form martensite, which is necessary for hydrogen cracking – CE < 0. 35 treatment – 0. 35 < CE < 0. 55 – 0. 55 < CE treatment no preheat or postweld heat preheat and postweld heat • Preheat temp. as CE and plate thickness

Carbon and Low-Alloy Steels Why Post-Weld Heat Treat? • The fast cooling rates associated with welding often produce martensite • During postweld heat treatment, martensite is tempered (transforms to ferrite and carbides) – – Reduces hardness Reduces strength Increases ductility Increases toughness • Residual stress is also reduced by the postweld heat treatment 0. 1. 1. 5. 1. T 10. 95. 12

Postweld Heat Treatment and Hydrogen Cracking Steel • Postweld heat treatment (~ 1200°F) tempers any martensite that may have formed – Increase in ductility and toughness – Reduction in strength and hardness • Residual stress is decreased by postweld heat treatment • Rule of thumb: hold at temperature for 1 hour per inch of plate thickness; minimum hold of 30 minutes

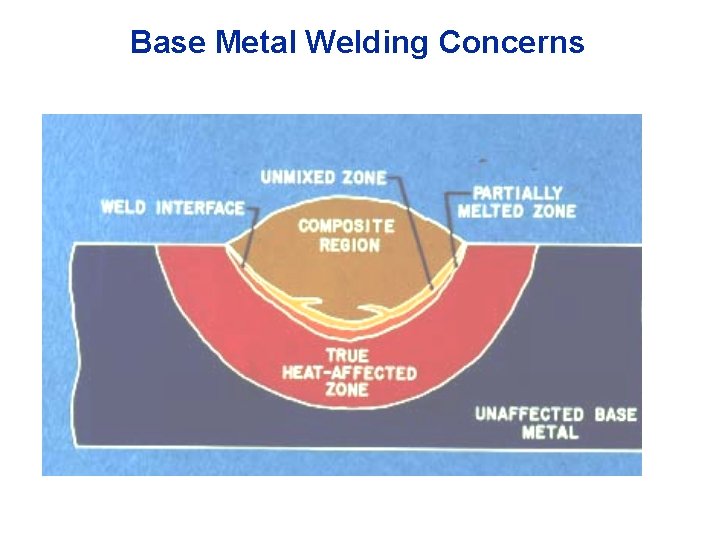
Base Metal Welding Concerns

Cracking in Welds Lamellar Tearing • Occurs in thick plate subjected to high transverse welding stress • Related to elongated non-metallic inclusions, sulfides and silicates, lying parallel to plate surface and producing regions of reduced ductility • Prevention by – Low sulfur steel – Specify minimum ductility levels in transverse direction – Avoid designs with heavy through-thickness direction stress 0. 1. 1. 5. 2. T 14. 95. 12
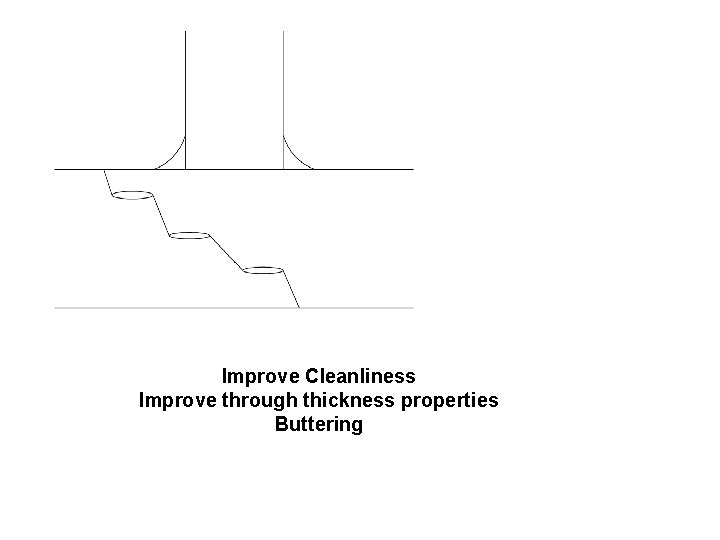
Improve Cleanliness Improve through thickness properties Buttering

Carbon and Low-Alloy Steels Multipass Welds • Heat from subsequent passes affects the structure and properties of previous passes – Tempering – Reheating to form austenite – Transformation from austenite upon cooling • Complex Microstructure 0. 1. 1. 5. 1. T 11. 95. 12
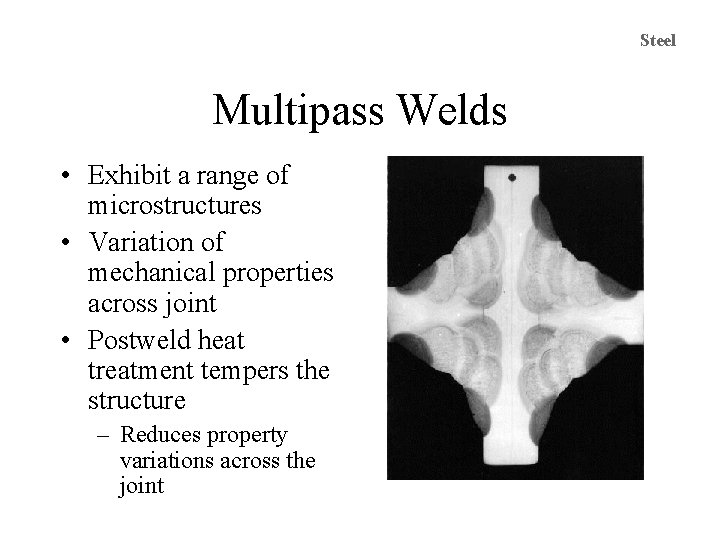
Steel Multipass Welds • Exhibit a range of microstructures • Variation of mechanical properties across joint • Postweld heat treatment tempers the structure – Reduces property variations across the joint

Cracking in Welds Reheat Cracking • Mo-V and Mo-B steels susceptible • Due to high temperature embrittlement of the heataffected zone and the presence of residual stress • Coarse-grained region near fusion line most susceptible • Prevention by – – – Low heat input welding Intermediate stress relief of partially completed welds Design to avoid high restraint Restrict vanadium additions to 0. 1% in steels Dress the weld toe region to remove possible areas of 0. 1. 1. 5. 2. T 15. 95. 12
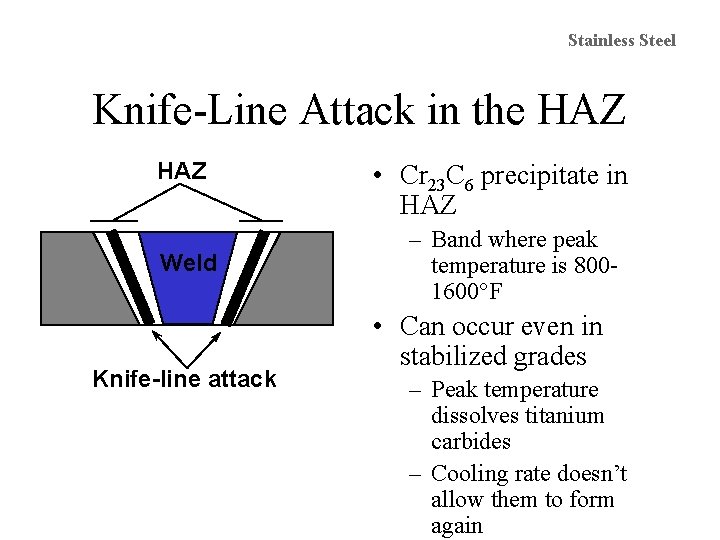
Stainless Steel Knife-Line Attack in the HAZ Weld Knife-line attack • Cr 23 C 6 precipitate in HAZ – Band where peak temperature is 8001600°F • Can occur even in stabilized grades – Peak temperature dissolves titanium carbides – Cooling rate doesn’t allow them to form again
- Slides: 37