METALLURGY Level 3 METALLURGY METALLURGY Level 3 OBJECTIVE
METALLURGY Level - 3 METALLURGY

METALLURGY Level - 3 OBJECTIVE QUESTIONS (LEVEL—III)

METALLURGY 1. Column – I (Metals) A) Copper B) Zinc C) Aluminium D) Iron 1) 2) 3) 4) Column – II (Ores) 1) Calamine 2) Cuprite 3) Hematite 4) Bauxite A B C D 1 2 4 2 2 1 3 4 2 3 4 3 1 4 Level - 3 Solution : Ø Cuprite ore contain copper Ø Calamine ore contain Zinc Ø Bauxite ore contain Alminium Ø Hematite ore contain Iron

METALLURGY Level - 3 2. Find the incorrectly matched pair Metals Ores 1) Copper glance 2) Malachite 3) Calamine 4) Siderite Solution : - Copper Magnesium Zinc Iron

METALLURGY Level - 3 PRINCIPLES OF EXTRACTION 3. The principle involved in the washing of the ore with water to separate the ore from impurities in the dressing process is 1) colour 2) different wettings 3) magnetic 4) densities Solution : By gravity separation process

METALLURGY Level - 3 4. The gas that cannot be used for the reduction of the ore is 1) H 2 2) CO 3) CO + H 2 4) O 2 Solution : Remaining are reducing agents

METALLURGY Level - 3 5. Match the following List - I Metallurgical process A) Smelting B) Roasting C) Calcination D) Leaching List - II Main change 1) in the absence of air ore decomposes 2) the ore generally is converted into a soluble compound 3) electrolysis takes place 4) Oxidation and the product in solid state 5) reduction and the product (metal or its sulphide) as liquid

METALLURGY Level - 3 A B C D 1) 5 4 1 2 2) 2 3 4 1 3) 3 1 5 2 4) 5 2 1 3 Solution : All are Definitions

METALLURGY Level - 3 6. Froth floatation process for the concentration of sulphide ores is an illustration of the practical application of 1) adsorption 2) absorption 3) sedimentation 4) coagulation Solution : It depends on adsorption

METALLURGY Level - 3 7. If a metal has low oxygen affinity then the purification of metal may be carried out by 1) liquation 2) distillation 3) zone refining 4) cupellation Solution : In cupellation metal is extracted from metal oxide if it contains less affinity of oxygen

METALLURGY Level - 3 1) (a), (b) only correct 2) (b), (c) only correct 3) (a), (c) only correct 4) (a), (b), (c) are correct Solution : Froth flotation process

METALLURGY Level - 3 9. Choose the correct code regarding Roasting process (I) It is the process of heating ore in air to obtain the oxide (II) It is an exothermic process (III) It is used for hydrated oxide and oxy salt ore (IV) It is used after the concentration of ore 1) I, II and III 2) I, II and IV 3) I, III and IV 4) I, III and IV Solution : In Roasting process oxygen is reacted and the reaction is exothermic, metal is extracted from ore

METALLURGY Level - 3 THERMODYNAMIC APPLICATIONS-METALLURGY 10. Ellingham diagram represents Solution : From the graph

METALLURGY Level - 3 1) (a), (b) only correct 2) (b), (c) only correct 3) (a), (c) only correct 4) (a), (b), (c) correct Solution : From the graph

METALLURGY Level - 3 REFINING & PURIFICATION METHODS 12. Ultrapure elements are obtained in the following method of metal refining 1) Electrolysis 2) Poling 3) Liquation 4) Distillation Solution : Ultra pure metals are obtained by Distillation

METALLURGY Level - 3 13. The solubility of the impurities in the metal in molten and solid states is different. This is the principle involved in the refining of metal by following method 1) Poling 2) Liquation 3) Zone refining 4) electrolysis Solution : In zone refining semiconductors are extracted

METALLURGY Level - 3 14. The method used to refine copper metal is 1) Liquation 2) Distillation 3) Zone refining 4) electrolytic method Solution :

METALLURGY Level - 3 1) Iodide of the metal 2) No reaction takes place 3) Impurities react with iodine 4) Metal doesn’t react Solution : (impure metal) (pure metal)

METALLURGY Level - 3 16. The method of zone refining of metals is based on the principle of 1) Greater mobility of the pure metal than that of impurity 2) Higher melting point of the impurity than that of the pure metal 3) Greater noble character of the solid metal than that of the impurity 4) Greater solubility of the impurity in the molten state than in the solid Solution : Solubility of the impurity: molten state > solid state in zone refining method

METALLURGY Level - 3 17. Match the following List - I Dressing methods List - II Principle involved A) Washing with water 1) Fusibility difference B) Froth floatation 2) density difference C) Hand picking 3) Wetting difference D) Liquation 4) Colour, size etc. of ore particulars difference 5) Magnetic property difference
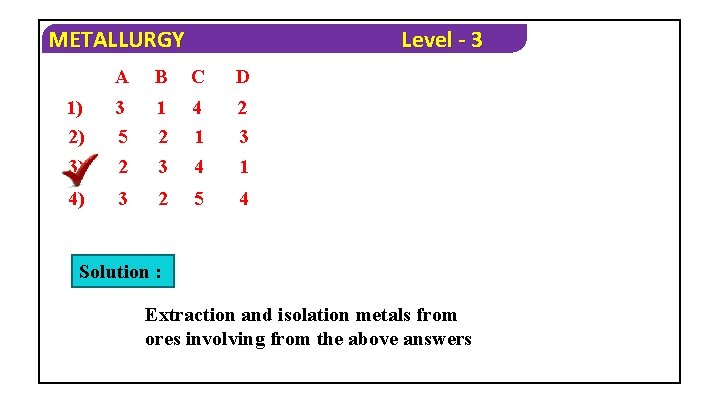
METALLURGY Level - 3 A B C D 1) 2) 3 5 1 2 4 1 2 3 3) 2 3 4 1 4) 3 2 5 4 Solution : Extraction and isolation metals from ores involving from the above answers

METALLURGY Level - 3 18. Which of the following set of elements mostly occur as sulphide ores. 1) Zn, Cu, Na 2) Zn, Cu, Pb 3) Fe, Al, Ti 4) Cu, Ag, Au Solution :

METALLURGY Level - 3 19. The oxide of a metal (R) can be reduced by the metal (P) and metal (R) can reduce the oxide of metal (Q). Then the decreasing order of the reactivity of metal (P), (Q) and (R) with oxygen is 1) P > Q > R 2) P > R > Q 3) R > P > Q 4) Q > P > R Solution : Decreasing of reducing Power from Ellingham diagram

METALLURGY Level - 3 20. The incorrect statements from the following (i) Every mineral is an ore but every ore is not a mineral (ii) Slag is product formed during extraction of metal by combination of flux and impurities. (iii) Highly pure metals can be obtained by zone refining (iv) Cornallite is an ore of magnesium and sodium 1) 1 and 2 2) 1 and 4 3) 1, 2 and 3 4) 1 and 3 Solution : Definition of ore, mineral

METALLURGY Level - 3 ELLINGHAM DIAGRAM Solution : From the Ellingham diagram

METALLURGY Level - 3 22. Consider the following reactions at 1000°C Choose the correct statement at 1000°C 1) Zinc can be oxidised by CO 2) Zinc can be reduced by C 3) Both (1) and (2) are true 4) Both (1) and (2) are false Solution : Zn. O + C→Zn + CO

METALLURGY Level - 3 23. According to Ellingham diagram, the oxidation reaction of carbon and carbon monoxide may be used to reduce which one of the following oxides at the lowest temperature. 1) Al 2 O 3 2) Cu 2 O 3) Mg. O 4) Zn. O Solution : From Ellingham diagram

METALLURGY Level - 3 24. The metal X is prepared by the electrolysis of fused chloride. it reacts with hydrogen to form a colourless solid from which hydrogen gas is released on treatment with water: the metal is 1) Al 2) Ca 3) Cu 4) Zn Solution : ‘Ca’ is obtained by the electrolysis of Ca. H 2 fused Ca. H 2 + 2 H 2 O Ca(OH)2 + 2 H 2
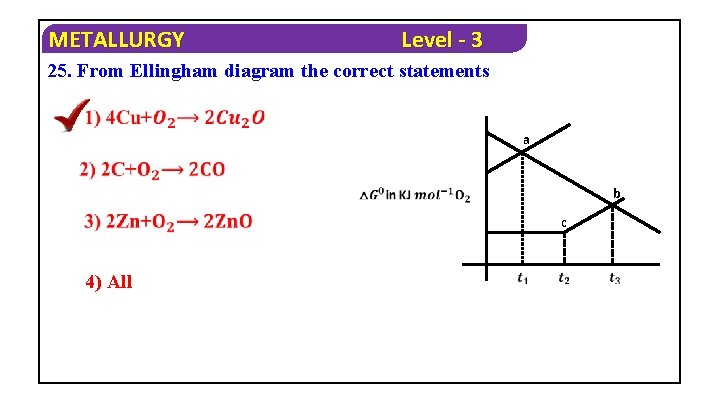
METALLURGY Level - 3 25. From Ellingham diagram the correct statements a b c 4) All

METALLURGY Solution : 4) All Level - 3
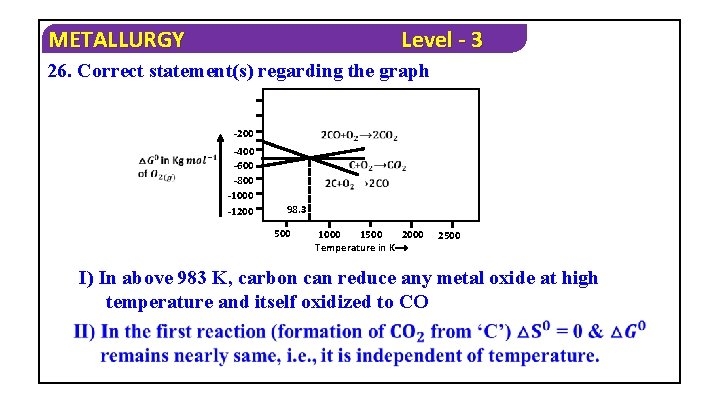
METALLURGY Level - 3 26. Correct statement(s) regarding the graph -200 -400 -600 -800 -1000 -1200 98. 3 500 1000 1500 2000 Temperature in K⟶ 2500 I) In above 983 K, carbon can reduce any metal oxide at high temperature and itself oxidized to CO

METALLURGY Level - 3 1) IV Only 2) I, II Only 3) I, III Only 4) All Solution : From Ellingham diagram

METALLURGY Level - 3 -200 -400 -600 -800 -1000 -1200 98. 3 500 1) Coke 2) CO 4) 1 & 2 Solution : From Ellingham diagram 1000 1500 2000 Temperature in K⟶ 2500
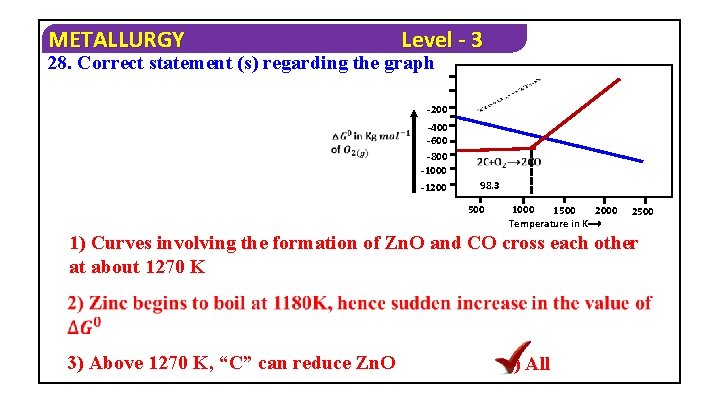
METALLURGY Level - 3 28. Correct statement (s) regarding the graph -200 -400 -600 -800 -1000 -1200 98. 3 500 1000 1500 2000 Temperature in K⟶ 2500 1) Curves involving the formation of Zn. O and CO cross each other at about 1270 K 3) Above 1270 K, “C” can reduce Zn. O 4) All
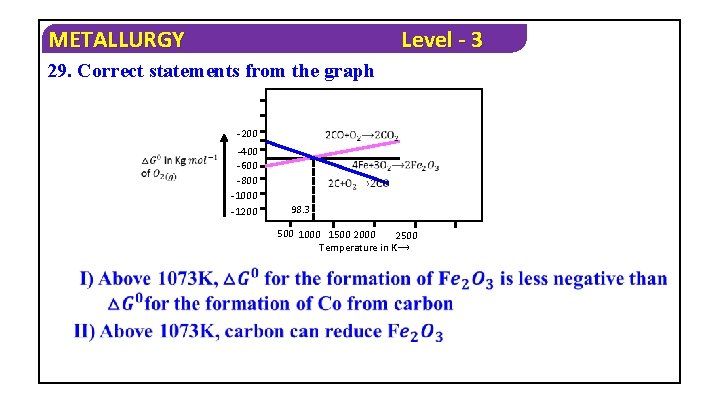
METALLURGY Level - 3 29. Correct statements from the graph -200 -400 -600 -800 -1000 -1200 98. 3 500 1000 1500 2000 2500 Temperature in K⟶

METALLURGY Level - 3 1) Only I 2) I, II Only 3) I, III Only 4) All Solution : From Ellingham diagram

METALLURGY Level - 3 Thank you…
- Slides: 37