University of Jeddah Department of Chemical Engineering Jeddah







![Materials Solvent Trioctylphosphine oxide (TOPO) ( [CH 3(CH 2)7]3 PO, mol wt. 386. 64 Materials Solvent Trioctylphosphine oxide (TOPO) ( [CH 3(CH 2)7]3 PO, mol wt. 386. 64](https://slidetodoc.com/presentation_image_h2/f862a6910e63ca21119a68a35a1558a3/image-13.jpg)






![Extraction % of Mixed Acids Fermented Using CYANEX 923 Extraction % [w/w] 110, 0 Extraction % of Mixed Acids Fermented Using CYANEX 923 Extraction % [w/w] 110, 0](https://slidetodoc.com/presentation_image_h2/f862a6910e63ca21119a68a35a1558a3/image-21.jpg)






- Slides: 33

University of Jeddah, Department of Chemical Engineering, Jeddah, Saudi Arabia University of Maine, Department of Chemical and Biological Engineering, Orono, Maine 04469 USA Dr. Aymn Abdulrahman September, 2016

Outline Introduction Motivation Objectives Material and Method Procedure Results and Conclusion Future Works Acknowledgment

Introduction Maine is a Forest state ( covers 90 % of the state) Major wood components Cellulose Hemicelluloses Lignin Biomass-derived fuels such as ethanol, renewable, widespread, cheap and sustainable 10% to 50% of transportation fuels could replaced between 2010 and 2050 in USA (B. Han, 2006) High selling price of acids such as acetic acid is double that of ethanol

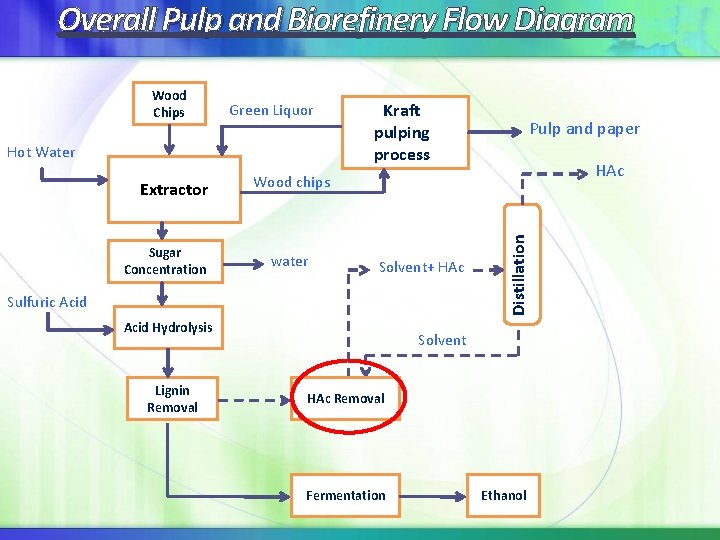
Overall Pulp and Biorefinery Flow Diagram Green Liquor Hot Water Extractor Sugar Concentration Kraft pulping process HAc Wood chips water Solvent+ HAc Sulfuric Acid Hydrolysis Lignin Removal Pulp and paper Distillation Wood Chips Solvent HAc Removal Fermentation Ethanol

Objectives � Liquid-liquid extraction to recover mixed acids (C 2 -C 7 aliphatic acids) from two systems: � Clean system ( mixed acid in DI water ) � Mixed acids from fermentation broth � Using CYANEX 923 as a solvent and compare to TOPO and TOA � Determining best extraction conditions � Develop an analytical method for organic phase � Solvent recyclability � Washing by 1 M Na. OH solution

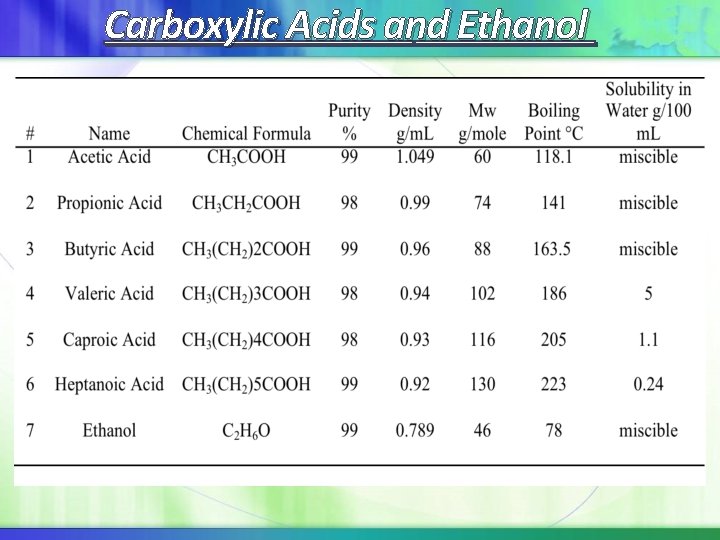
Carboxylic Acids and Ethanol

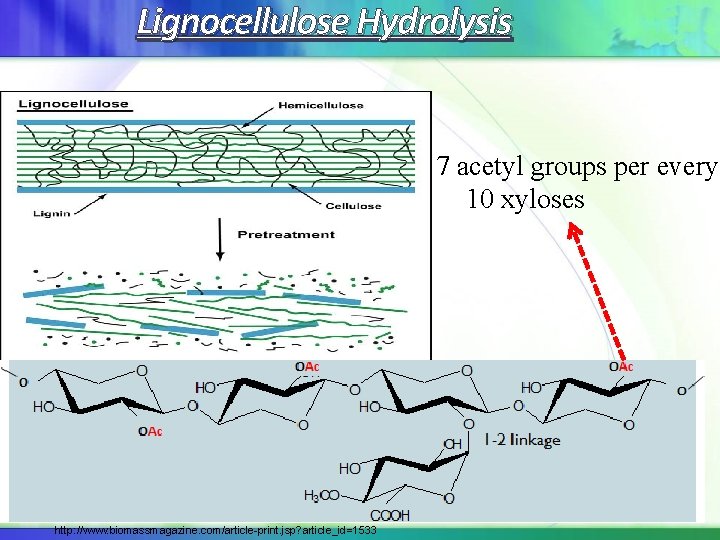
Lignocellulose Hydrolysis 7 acetyl groups per every 10 xyloses USDA Agricultural Research Service http: //www. biomassmagazine. com/article-print. jsp? article_id=1533

Pretreatment Processes Solubilizes polymer sugars in the wood Makes them accessible to enzymatic hydrolysis Fermentable by the mixed culture of microbes

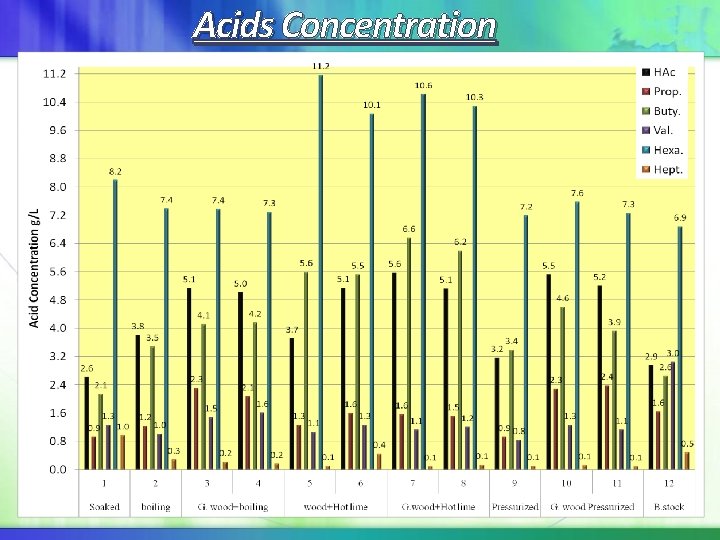
Acids Concentration

Lignin Removal Solid Lignin Filtration Centrifuging Soluble Lignin Lower p. H Centrifuging

L-L Extraction Methods Liquid-Liquid Extraction Clean System (acids in DI water) Mixed acids from fermentation broth Separations Centrifuging Distillation Recycling

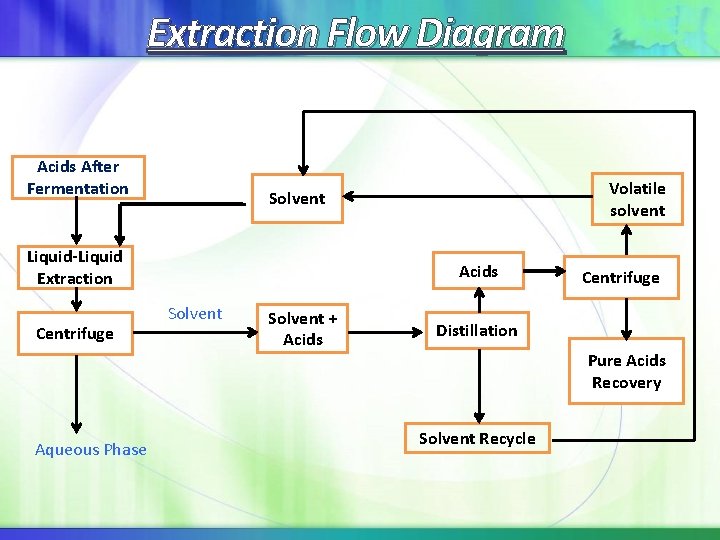
Extraction Flow Diagram Acids After Fermentation Liquid-Liquid Extraction Centrifuge Aqueous Phase Volatile solvent Solvent Acids Solvent + Acids Centrifuge Distillation Pure Acids Recovery Solvent Recycle
![Materials Solvent Trioctylphosphine oxide TOPO CH 3CH 273 PO mol wt 386 64 Materials Solvent Trioctylphosphine oxide (TOPO) ( [CH 3(CH 2)7]3 PO, mol wt. 386. 64](https://slidetodoc.com/presentation_image_h2/f862a6910e63ca21119a68a35a1558a3/image-13.jpg)
Materials Solvent Trioctylphosphine oxide (TOPO) ( [CH 3(CH 2)7]3 PO, mol wt. 386. 64 ) Undecane ( C 11 H 24, mol. wt. 156. 3 ) Trioctylamine (TOA) ( [CH 3(CH 2)7]3 N, mol wt. 353. 67 ) Octanol ( CH 3(CH 2)7 OH, mol wt. 130. 23 ) CYANEX 923 a mixture of liquid phosphine oxide (mol wt. 348 ) C 2 -C 7 aliphatic acids Sulfuric acid ( H 2 SO 4 ), Phosphoric acids or HCL

CYANEX 923 Advantages Completely miscible with all common hydrocarbon Easy Handle it at low room temperatures Direct use it (no dilution needed)

L-L Extraction Procedure Aq. + Cyanex Analysis Shaker for 30 min, 230 speed Centrifuge (3000 rpm, 30 minutes)

Two Phases Separation Note: Phase separation after TOPO-undecane extraction at 70 C and p. H=1

Distillation � Fractional distillation column � Separate mixed acid from solvent

Analysis Aqueous phase High Performance Liquid Chromatography (HPLC) (Bio-Rad Aminex HPX-87 H Column) Gas Chromatography (GC) with He as carrier gas and FID Organic phase Gas Chromatography (GC) with He as carrier gas and FID p. H meter

Extraction Conditions Effect of concentrations of solvent in the organic solution Effect of ratios of organic to aqueous phase Effect of p. H Effect of temperature Effect of agitation on extraction % (using CYANEX 923) Effect of centrifuging on extraction % (using CYANEX 923)

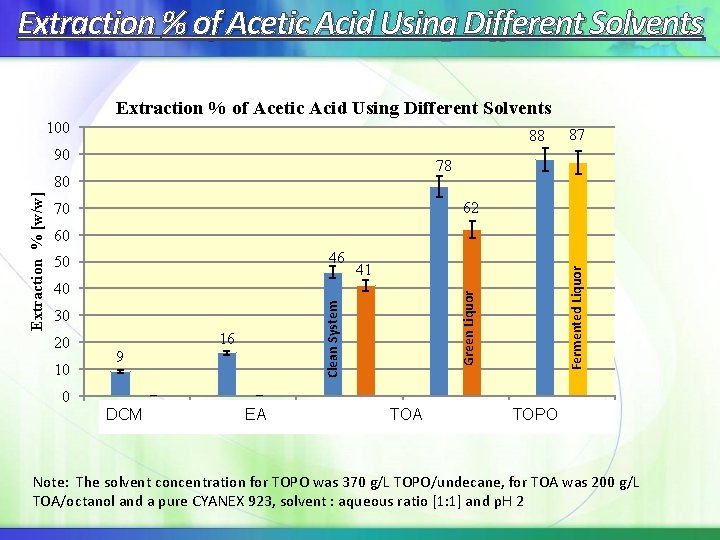
Extraction % of Acetic Acid Using Different Solvents 100 88 90 78 80 62 70 41 40 30 20 10 16 9 Green Liquor 46 50 Fermented Liquor 60 Clean System Extraction % [w/w] 87 0 DCM EA EA TOA TOPO CYANEX Note: The solvent concentration for TOPO was 370 g/L TOPO/undecane, for TOA was 200 g/L TOA/octanol and a pure CYANEX 923, solvent : aqueous ratio [1: 1] and p. H 2
![Extraction of Mixed Acids Fermented Using CYANEX 923 Extraction ww 110 0 Extraction % of Mixed Acids Fermented Using CYANEX 923 Extraction % [w/w] 110, 0](https://slidetodoc.com/presentation_image_h2/f862a6910e63ca21119a68a35a1558a3/image-21.jpg)
Extraction % of Mixed Acids Fermented Using CYANEX 923 Extraction % [w/w] 110, 0 100, 0 HAc 90, 0 Prop. 80, 0 Buty. 70, 0 60, 0 50, 0 40, 0 95 89, 7 87, 7 90, 4 96 96, 7 91, 2 79 Val Hex. 45, 6 Hept. 30, 0 20, 0 10, 0 0, 5 3, 4 6, 7 p. H 2 Note: Using pure CYANEX 923, solvent : aqueous ratio [1: 1], initial concentration of acids as following: 5. 6 g/L of acetic acid, 1. 6 g/L of propionic acid, 6. 6 g/L of butyric acid, 1. 1 g/L of valeric acid, 10. 6 g/L of hexanoic acid and 0. 1 g/L of heptanoic acid

Acetic Acid Recovered by Distillation Solvent HAc before Distillation g/L HAc after Distillation g/L HAc Recovering [w/w] % TOA 3. 6 0 TOPO 5. 6 0. 2 89 5. 1 0. 3 84 CYANEX 923 Note: Green liquor extracts are used for TOA and TOPO, fermented broth is used for CYANEX 923, volume ratio was 1, p. H is 2, 370 g/L TOPO/undecane, 200 g/L TOA/octanol and pure CYANEX 923

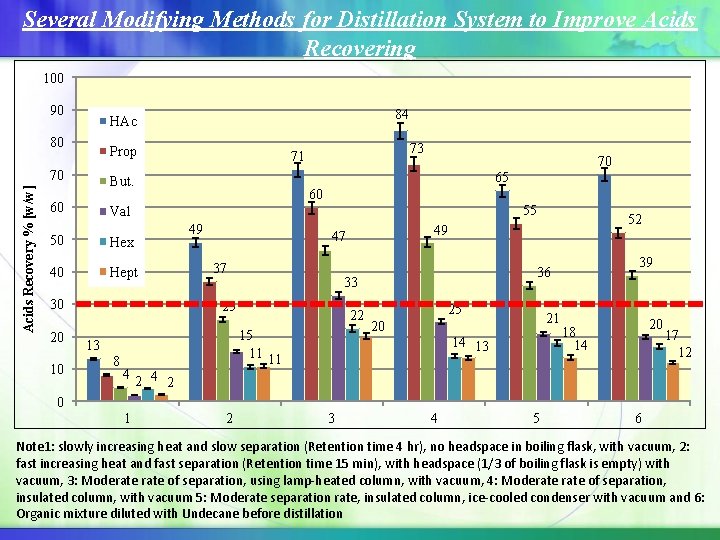
Several Modifying Methods for Distillation System to Improve Acids Recovering 100 90 80 Prop Acids Recovery % [w/w] 70 70 65 60 55 Val 49 50 Hex 40 Hept 30 10 73 71 But. 60 20 84 HAc 47 37 15 11 11 13 8 4 22 39 36 33 25 52 49 25 21 20 14 13 20 18 14 17 12 2 4 2 0 1 2 3 4 5 6 Note 1: slowly increasing heat and slow separation (Retention time 4 hr), no headspace in boiling flask, with vacuum, 2: fast increasing heat and fast separation (Retention time 15 min), with headspace (1/3 of boiling flask is empty) with vacuum, 3: Moderate of separation, using lamp-heated column, with vacuum, 4: Moderate of separation, insulated column, with vacuum 5: Moderate separation rate, insulated column, ice-cooled condenser with vacuum and 6: Organic mixture diluted with Undecane before distillation

Effect of High Boiling Temperature on Solvent’s Color Note: Boiling temperature is the temperature at the bottom of the distillation column

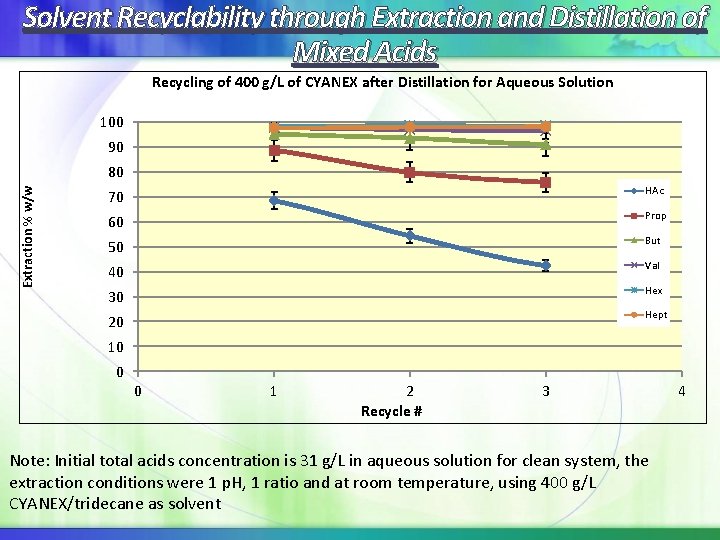
Solvent Recyclability through Extraction and Distillation of Mixed Acids Recycling of 400 g/L of CYANEX after Distillation for Aqueous Solution 100 90 Extraction % w/w 80 70 HAc 60 Prop 50 But 40 Val 30 Hex 20 Hept 10 0 0 1 2 Recycle # 3 Note: Initial total acids concentration is 31 g/L in aqueous solution for clean system, the extraction conditions were 1 p. H, 1 ratio and at room temperature, using 400 g/L CYANEX/tridecane as solvent 4

Conclusion on Distillation • Not work for long chain acids • Due to an azeotrope formation • Need high temperature • May cause solvent degradation • No significant affect using vacuum system • Reduce solvent recyclability

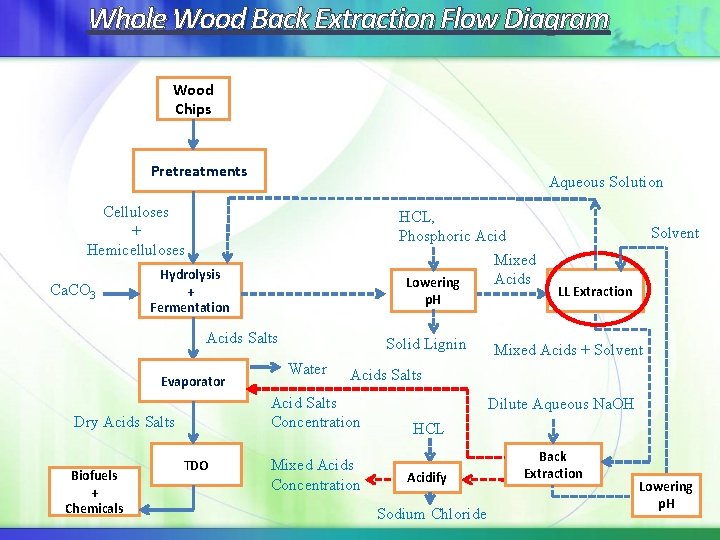
Whole Wood Back Extraction Flow Diagram Wood Chips Pretreatments Aqueous Solution Celluloses + Hemicelluloses Ca. CO 3 HCL, Phosphoric Acid Mixed Acids Lowering Hydrolysis + Fermentation p. H Acids Salts Evaporator Water TDO LL Extraction Mixed Acids + Solvent Acids Salts Acid Salts Concentration Dry Acids Salts Biofuels + Chemicals Solid Lignin Solvent Mixed Acids Concentration Dilute Aqueous Na. OH HCL Acidify Sodium Chloride Back Extraction Lowering p. H

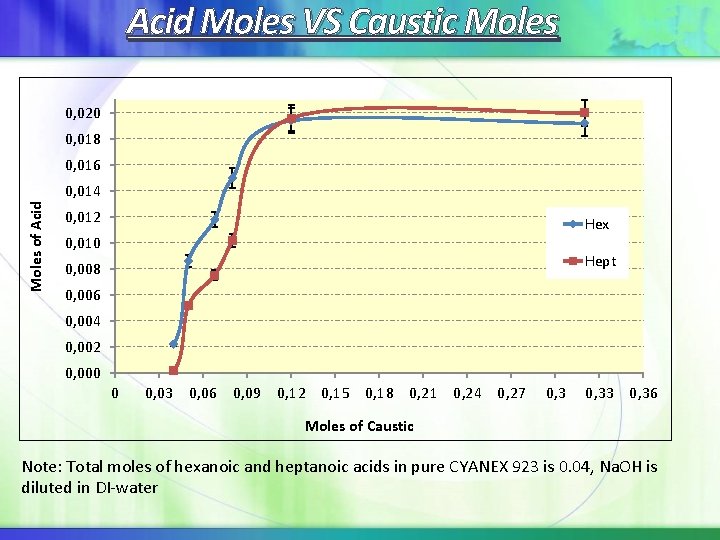
Acid Moles VS Caustic Moles 0, 020 0, 018 0, 016 Moles of Acid 0, 014 0, 012 Hex 0, 010 Hept 0, 008 0, 006 0, 004 0, 002 0, 000 0 0, 03 0, 06 0, 09 0, 12 0, 15 0, 18 0, 21 0, 24 0, 27 0, 33 0, 36 Moles of Caustic Note: Total moles of hexanoic and heptanoic acids in pure CYANEX 923 is 0. 04, Na. OH is diluted in DI-water

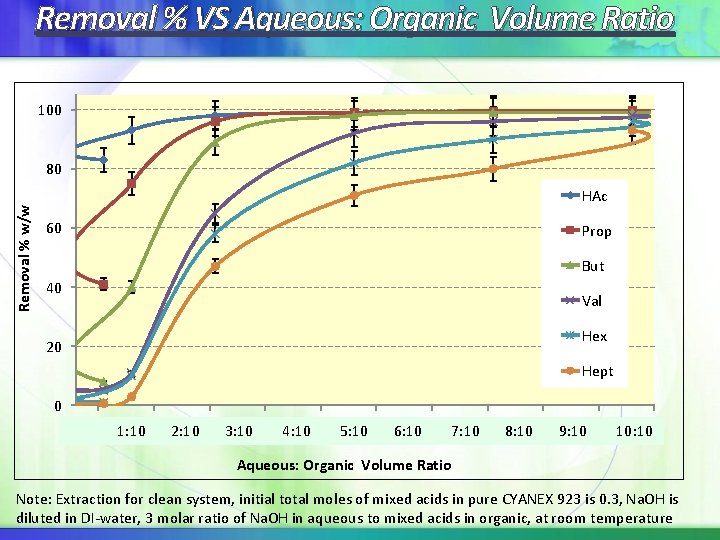
Removal % VS Aqueous: Organic Volume Ratio 100 Removal % w/w 80 HAc 60 Prop But 40 Val Hex 20 Hept 0 0, 00 0, 10 1: 10 0, 20 2: 10 0, 30 3: 10 0, 40 4: 10 0, 50 5: 10 0, 60 6: 10 0, 70 7: 10 0, 80 8: 10 0, 90 9: 10 1, 00 10: 10 Aqueous: Organic Volume Ratio Note: Extraction for clean system, initial total moles of mixed acids in pure CYANEX 923 is 0. 3, Na. OH is diluted in DI-water, 3 molar ratio of Na. OH in aqueous to mixed acids in organic, at room temperature

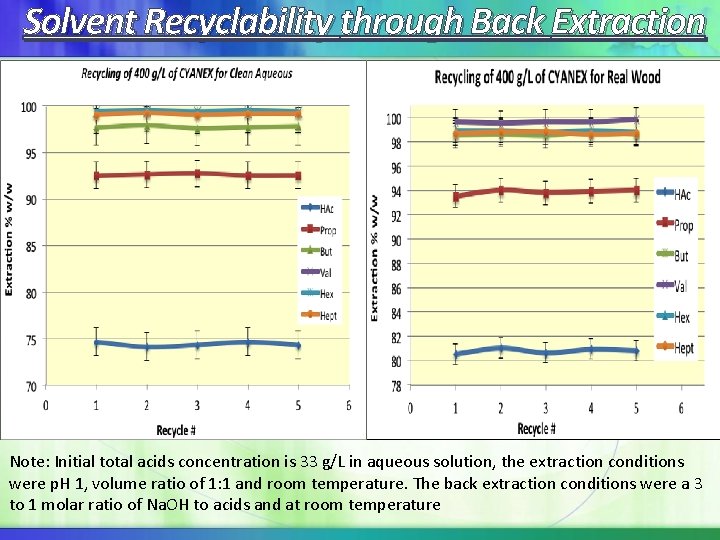
Solvent Recyclability through Back Extraction Note: Initial total acids concentration is 33 g/L in aqueous solution, the extraction conditions were p. H 1, volume ratio of 1: 1 and room temperature. The back extraction conditions were a 3 to 1 molar ratio of Na. OH to acids and at room temperature

Conclusions Liquid-liquid extraction recovered mixed acid from two systems: Clean system ( mixed acid in D-water ) Mixed acids from fermentation broth Best extraction conditions have been determined The distillation not working Not work for long chain acids Due to an azeotrope formation Back extraction by dilute sodium hydroxide Acid salts or mixed acids concentration

Future Works Distillation Well know on an azeotrope (break azeotrope) Using different solvents Back Extraction Using different strong basic solution Acids concentration Scale up for a pilot plant

Acknowledgment University of Jeddah Dr. G. Peter van Walsum Thesis Committee members (Dr. van Heiningen, Dr. Wheeler, Dr. Genco and Dr. Cole) Diane Smith (Analytical Support) Dr. Yang Yu (Mixed Acids Fermentation) Nick Hill (Technical Support)