Science Joke of the Day Two atoms were














- Slides: 20

Science Joke of the Day Two atoms were walking on the street. One atom lost an electron. The second atom asks: Are you sure you want to continue walking? Yes, I’m positive.

Review from last day: Write the names of the following compounds. 1. 2. 3. 4. 5. Ca. F 2 KBr Be. O Li. Cl Rb 3 P

Review from last day: Write the names of the following compounds. 1) Ca. F 2 Calcium fluoride 2) KBr Potassium bromide 3) Be. O Beryllium oxide 4) Li. Cl Lithium chloride 5) Rb 3 P Rubidium phosphide

Formulas of Ionic Compounds

Zn 3 N 2 Subscripts “sub” means below and “script” means to write

Subscript: the tiny number located on the bottom right of each element in a compound • Reminder: No subscript means: 1! • Subscripts show the RATIO (relative amounts) of each ion in an ionic lattice Ex. Zn 3 N 2 MEANS: There are 3 Zn 2+ ions for every 2 N 3 - ions in the lattice

SUBSCR IPTS = ?

Ionic compounds are electrically neutral so that means…. the positive charges of the cations in the lattice balance out the negative charges of the anions in the lattice! (the sum of the ion charges equals zero!!!) ttice Some Ionic La Types ALL ARE NEUTRAL (charge =0) !!!

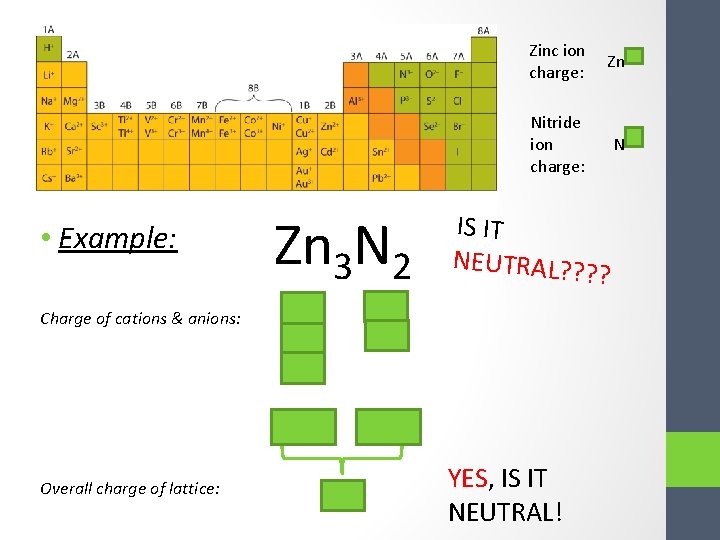
Zinc ion Zn 2+ charge: Nitride ion charge: • Example: Charge of cations & anions: Zn 3 N 2 Zn 2+ N 3 - IS IT NEUTRAL? ? ? ? Zn 2+ Overall charge of lattice: ___ 6+ 60!! N 3 - YES, IS IT NEUTRAL!

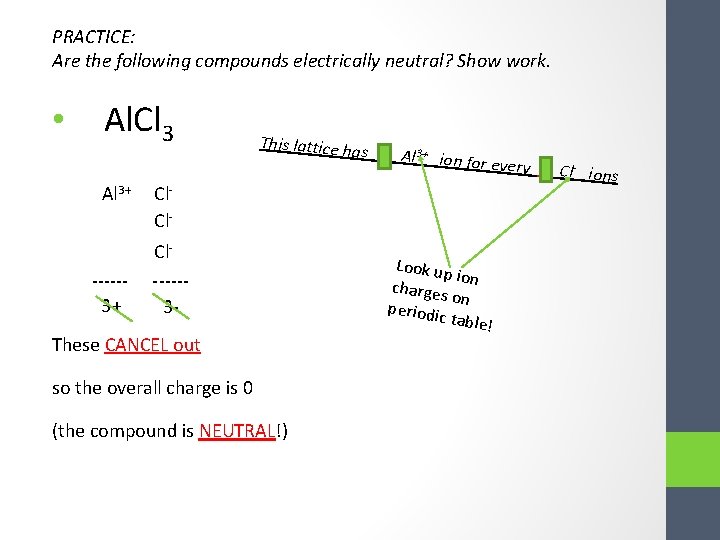
PRACTICE: Are the following compounds electrically neutral? Show work. • Al. Cl 3 Al 3+ -----3+ This lattice ha Cl. Cl-----3 - These CANCEL out so the overall charge is 0 (the compound is NEUTRAL!) s 1 Al 3+ ion for every 3 C l ions Look up io charges n o periodi n c table !

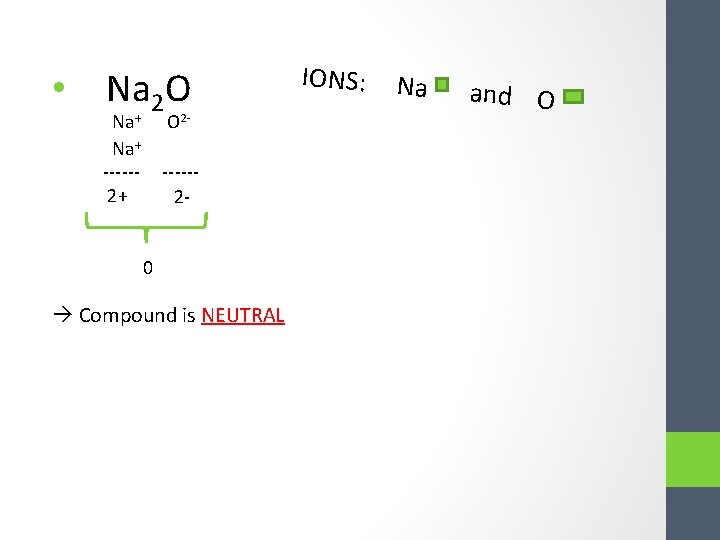
• Na 2 O Na+ -----2+ O 2 - -----2 - 0 Compound is NEUTRAL IONS: Na + and O 2 -

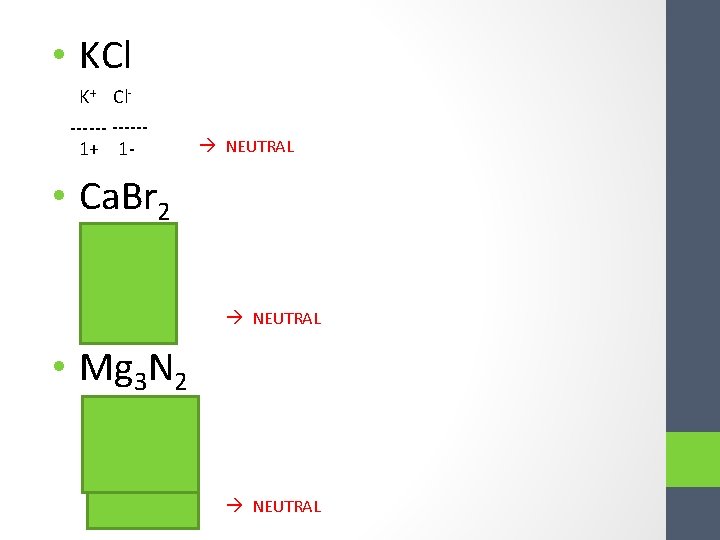
• KCl K+ Cl------1+ 1 - NEUTRAL • Ca. Br 2 Ca 2+ Br ----- 2+ 2 - NEUTRAL • Mg 3 N 2 Mg 2+ N 3 - Mg 2+ ----- 6+ 6 - NEUTRAL

Can you work backwards and figure out what the subscripts of the formula are? 1. magnesium oxide 2. Zirconium sulfide 3. Aluminum fluoride

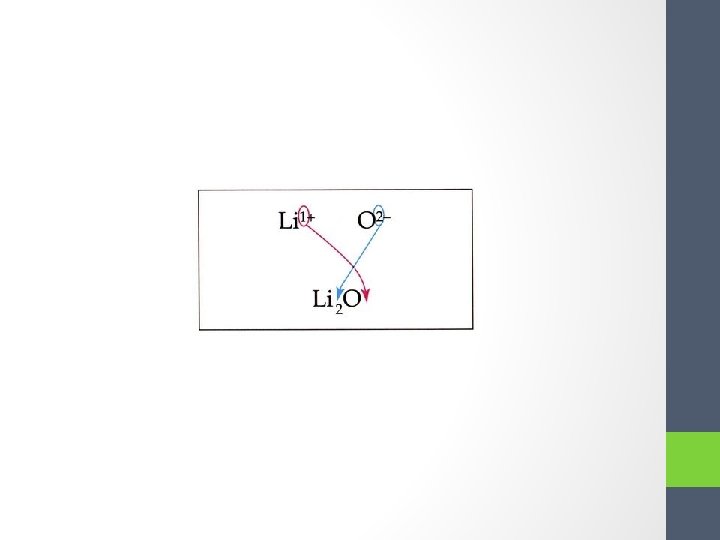
Short Cut • “Swap and Drop” • https: //www. youtube. com/watch? v=Zem. NCgv. M 3 Kw

Rules for writing formulas of ionic compounds: 1. Identify each ion and its charge 2. Swap and Drop the “charges” into “subscripts” 3. Double check to make sure that the final formula has the smallest whole number ratio

Examples: Aluminum oxide • 1. Identify each ion and its charge • Cation = Al 3+ • Anion = O 22. Cross the charges • Use subscripts to write the formula • •

3. Double check to make sure that the final formula has the smallest whole number ratio • Al 2 O 3

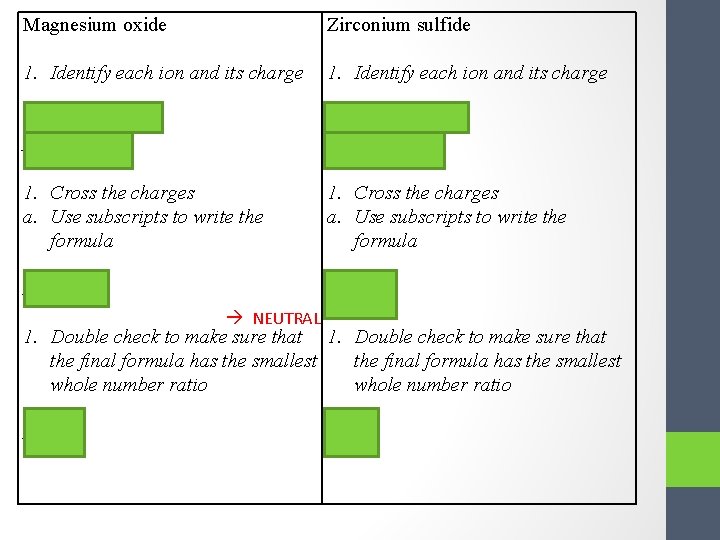
Magnesium oxide Zirconium sulfide 1. Identify each ion and its charge Cation = Mg 2+ Cation = Zr 4+ Anion = O 2 - Anion = S 2 - 1. Cross the charges a. Use subscripts to write the formula Mg 2 O 2 Zr 2 S 4 NEUTRAL 1. Double check to make sure that the final formula has the smallest whole number ratio Mg. O Zr. S 2


Try practice problems on pg. 87 of your text (Make sure the ones on pg. 86 are done!)