Principles of Quantum Mechanics Physics 123 2192021 Lecture






- Slides: 15

Principles of Quantum Mechanics Physics 123 2/19/2021 Lecture XV 1

Concepts • • • De Broigle waves Heisenberg’s uncertainty principle Schrödinger’s equation Particle in a box Boundary conditions Unitarity condition 2/19/2021 Lecture XV 2

Wave – Particle duality • If light exhibits both wave and particle properties then particles (e. g. electrons) must also exhibit wave properties – e. g. interference. • Matter (de Broglie) waves l=h/p p=mv 2/19/2021 Lecture XV 3

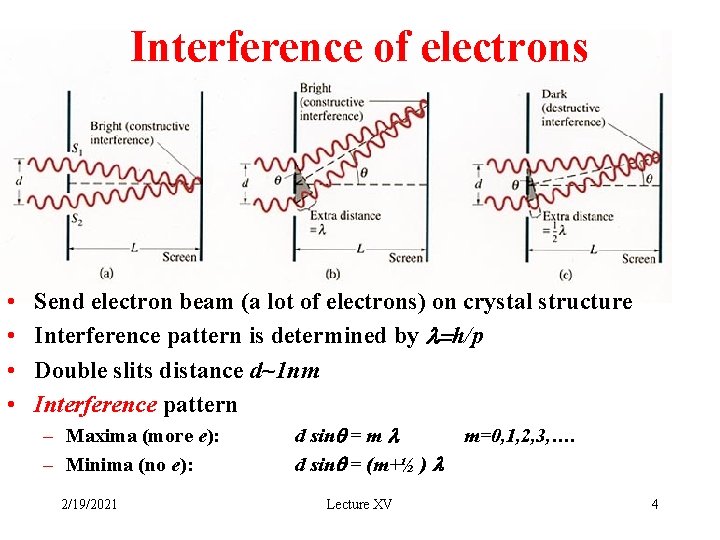
Interference of electrons • • Send electron beam (a lot of electrons) on crystal structure Interference pattern is determined by l=h/p Double slits distance d~1 nm Interference pattern – Maxima (more e): – Minima (no e): 2/19/2021 d sinq = m l d sinq = (m+½ ) l Lecture XV m=0, 1, 2, 3, …. 4

Matter waves • Particle position in space cannot be predicted with infinite precision • Heisenberg uncertainty principle • (Wave function Y of matter wave)2 d. V=probability to find particle in volume d. V. • Laws of quantum mechanics predict Y for a given system • Given Y one can estimate probability for certain outcomes of experiment 2/19/2021 Lecture XV 5

Schrödinger’s equation • Equivalent of energy conservation equation in classical mechanics. • Predicts the shape of the wave function. • System is defined by potential energy, boundary conditions 2/19/2021 Lecture XV 6

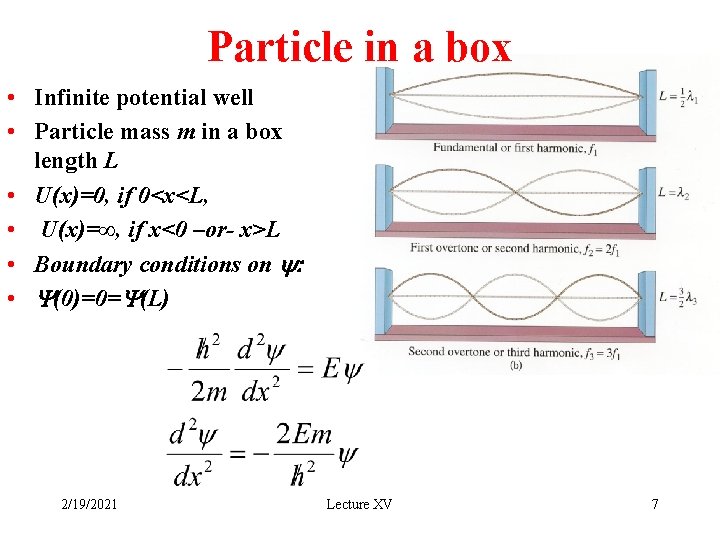
Particle in a box • Infinite potential well • Particle mass m in a box length L • U(x)=0, if 0<x<L, • U(x)=∞, if x<0 –or- x>L • Boundary conditions on y: • Y(0)=0=Y(L) 2/19/2021 Lecture XV 7

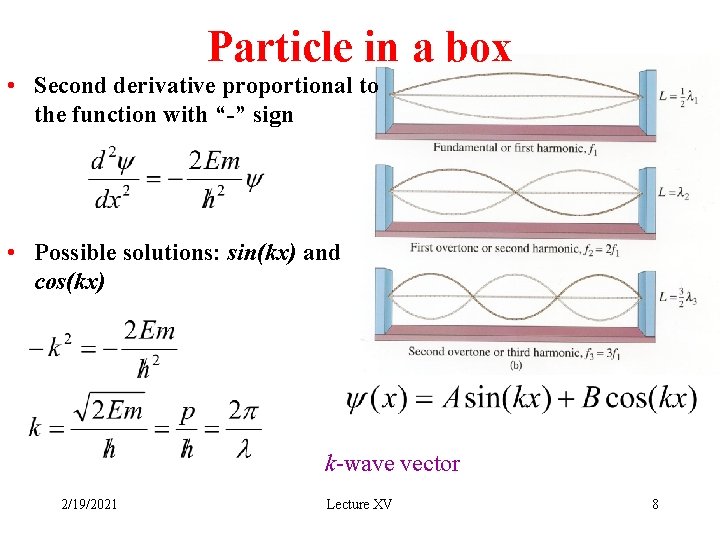
Particle in a box • Second derivative proportional to the function with “-” sign • Possible solutions: sin(kx) and cos(kx) k-wave vector 2/19/2021 Lecture XV 8

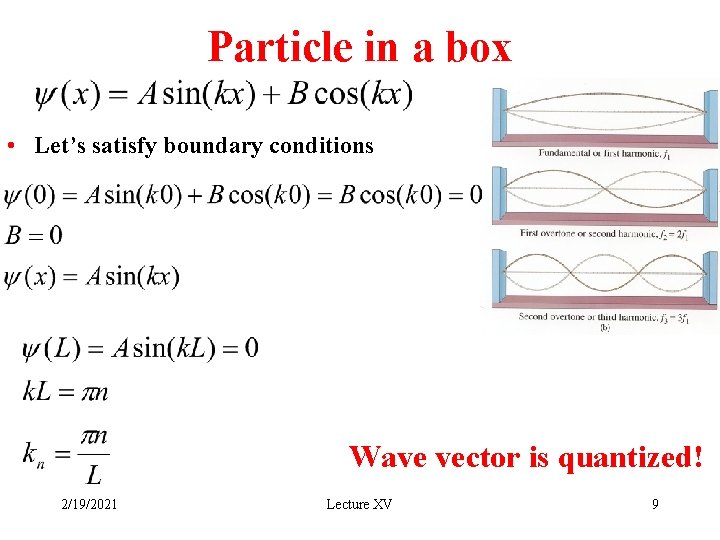
Particle in a box • Let’s satisfy boundary conditions Wave vector is quantized! 2/19/2021 Lecture XV 9

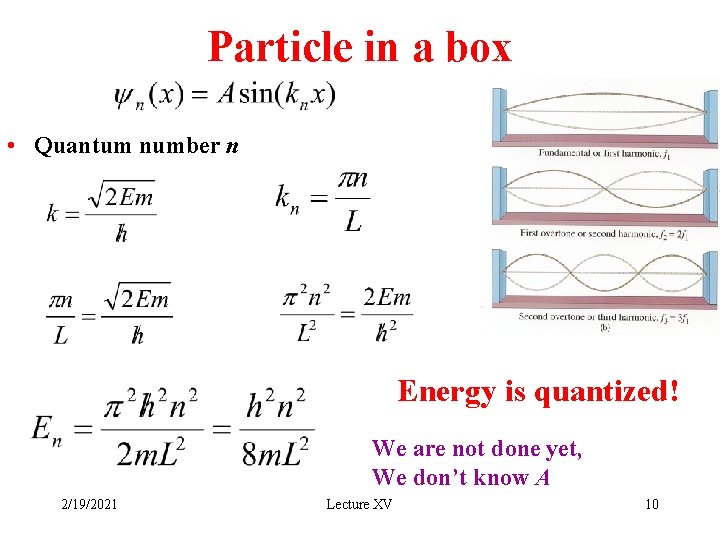
Particle in a box • Quantum number n Energy is quantized! We are not done yet, We don’t know A 2/19/2021 Lecture XV 10

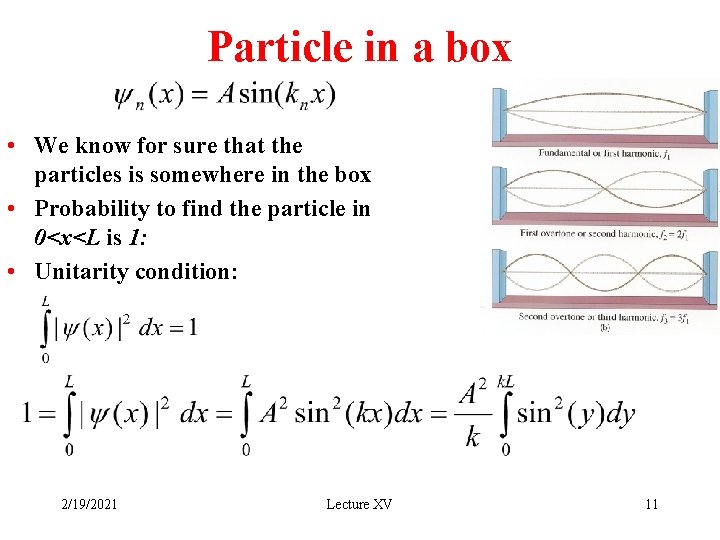
Particle in a box • We know for sure that the particles is somewhere in the box • Probability to find the particle in 0<x<L is 1: • Unitarity condition: 2/19/2021 Lecture XV 11

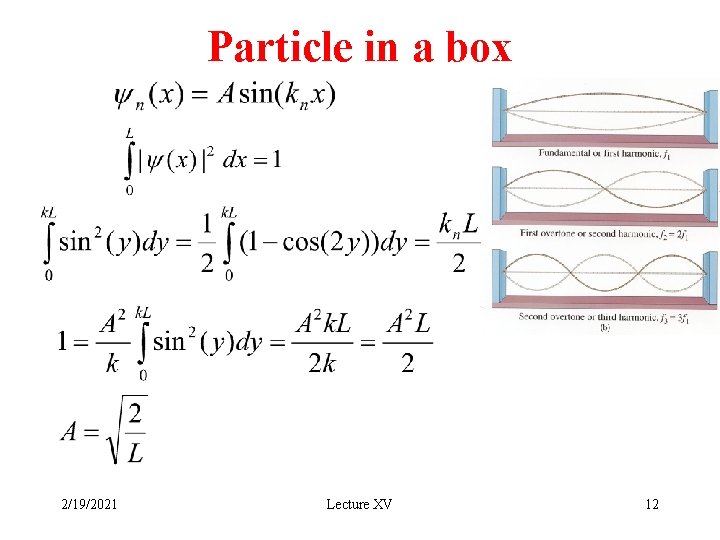
Particle in a box 2/19/2021 Lecture XV 12

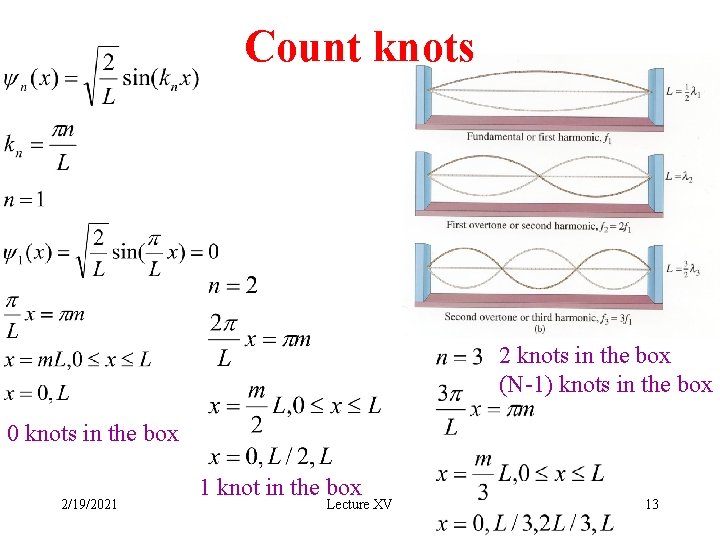
Count knots 2 knots in the box (N-1) knots in the box 0 knots in the box 2/19/2021 1 knot in the box Lecture XV 13

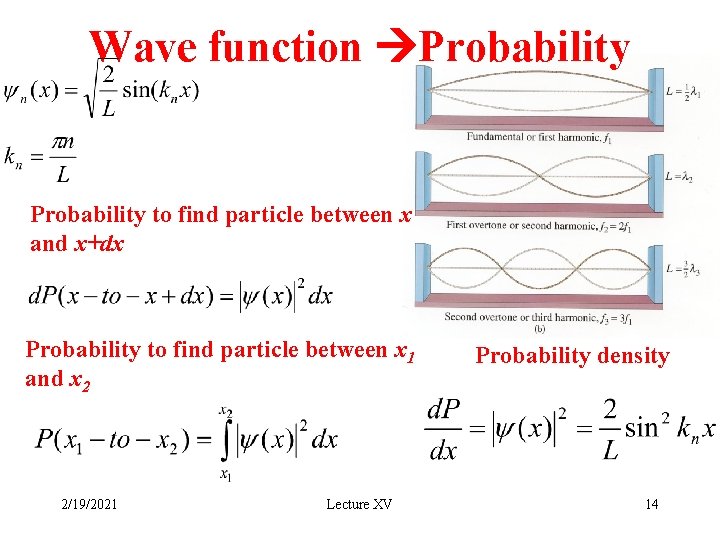
Wave function Probability to find particle between x and x+dx Probability to find particle between x 1 and x 2 2/19/2021 Lecture XV Probability density 14

Symmetry considerations The observable: probability density must respect the symmetry of the system System is symmetric around x=L/2 This means that the probability density function does not change if I replace x with (L-x): Wave function is either symmetric or Odd harmonics have even number of knots and are antisymmetric Even harmonics have odd number of knots and are 2/19/2021 Lecture XV 15 antisymmetric