OCEANESS 410 Class 19 Paleoceanography William Wilcock Learning







- Slides: 20

OCEAN/ESS 410 Class 19. Paleoceanography William Wilcock

Learning Goals • Understand how δ 18 O is defined • Understand why δ 18 O decreases with decreasing temperature in ice sheets • Understand what causes δ 18 O in foraminifera to vary and how it can be used to infer past climate.

Paleoclimate • Ice cores 123, 000 years Greenland, 800, 000 years Antarctica Temperature & air bubbles Sub annual resolution • Tree Rings Continuous for a few thousand years (older with radiocarbon dating) • Corals Continuous for a few hundred years (older with dating) • Sediments >100 Million years but not in 1 core and preservation of fossils effectively limits it to significantly less.

Time resolution of sediment record • Typical deep sea sedimentation rates – 0. 1 to 3 cm / 103 yr • Bioturbation in most settings – 3 -10 cm • Resolution – 103 to 105 years – Changes over shorter term cannot be resolved in a sediment core

Dating Sediments • Absolute – Radiometric (14 -C, 230 -Th/U, K-Ar) • Relative from cross-correlation – Paleomagnetic – Fossil record – Lithology • Time on a rubber band

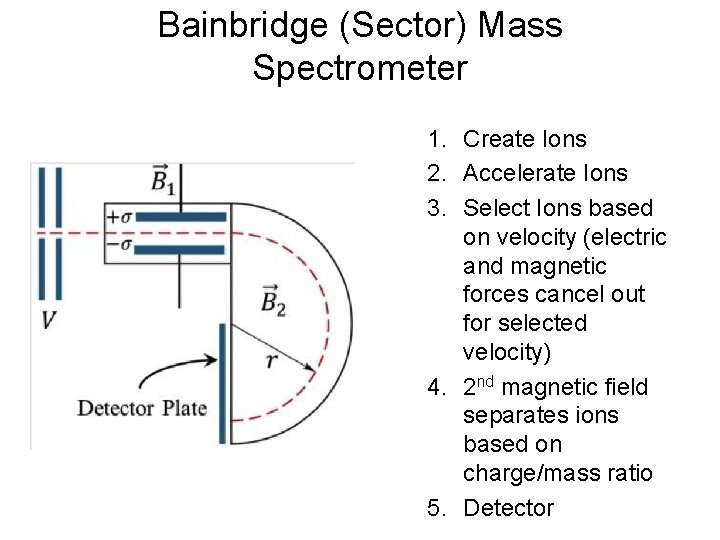
Bainbridge (Sector) Mass Spectrometer 1. Create Ions 2. Accelerate Ions 3. Select Ions based on velocity (electric and magnetic forces cancel out for selected velocity) 4. 2 nd magnetic field separates ions based on charge/mass ratio 5. Detector

Equations for Mass Spectrometer • Velocity selection stage – Electrostatic force • FE = q. E Where q is charge and E is electric field – Magnetic force • FB 1 = qv. B • Where v is velocity and B 1 is magnetic field – Selection (no bending) when FE = FB 1 or v=E/B 1 • Charge to mass ratio separation – Acceleration from magnetic field • FB 2 = ma = qv. B 2 or a=qv. B 2/m – Centripetal force • a = v 2/r = qv. B 2/m or r = mv/(q. B 2) • r increase with mass of ion

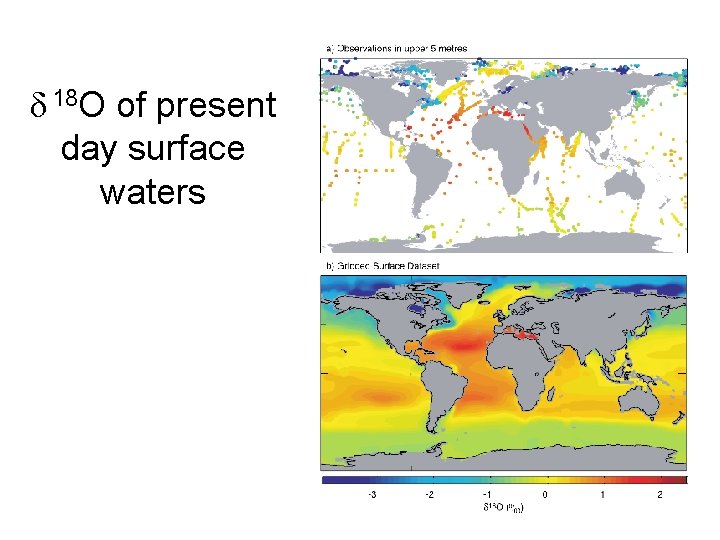
Oxygen Isotopes Stable Isotopes 99. 759% 16 O 0. 037% 17 O 0. 204% 18 O The lighter isotopes is preferentially incorporated into vapor, slightly more so at lower temperatures, and the heavier isotope is preferentially incorporated into rain. Standard = SMOW (Standard Mean Ocean Water) Water vapor in equilibrium with SMOW had δ 18 O = -9 to -11‰

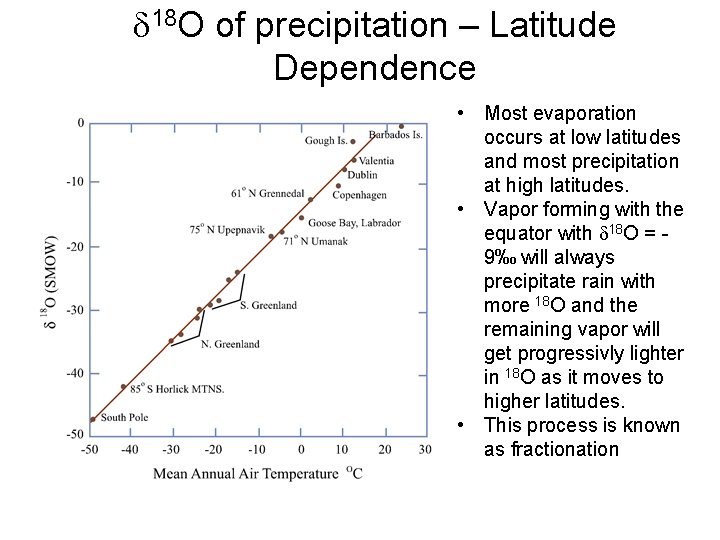
d 18 O of precipitation – Latitude Dependence • Most evaporation occurs at low latitudes and most precipitation at high latitudes. • Vapor forming with the equator with d 18 O = 9‰ will always precipitate rain with more 18 O and the remaining vapor will get progressivly lighter in 18 O as it moves to higher latitudes. • This process is known as fractionation

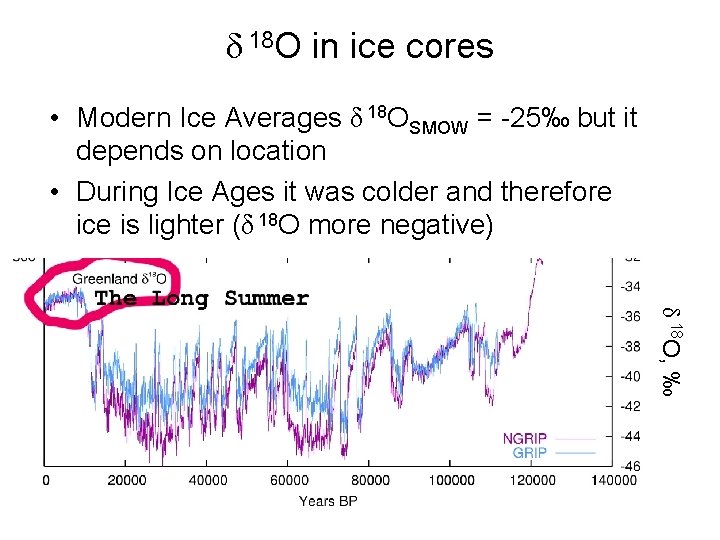
δ 18 O in ice cores • Modern Ice Averages δ 18 OSMOW = -25‰ but it depends on location • During Ice Ages it was colder and therefore ice is lighter (δ 18 O more negative) δ 18 O, ‰

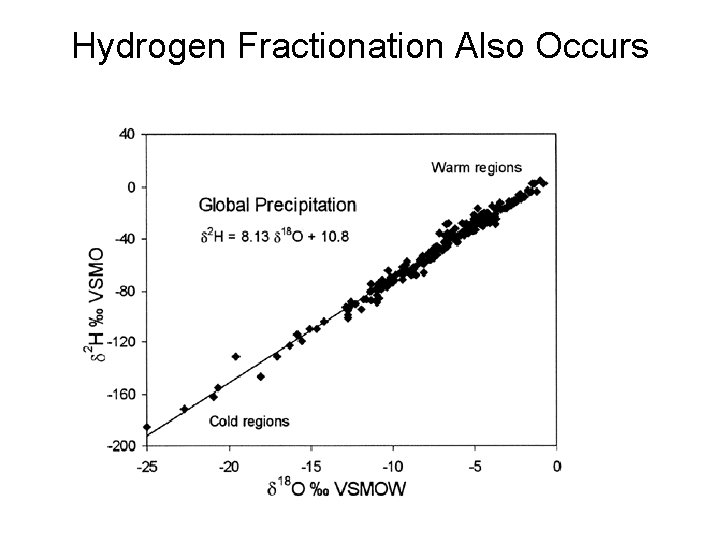
Hydrogen Fractionation Also Occurs

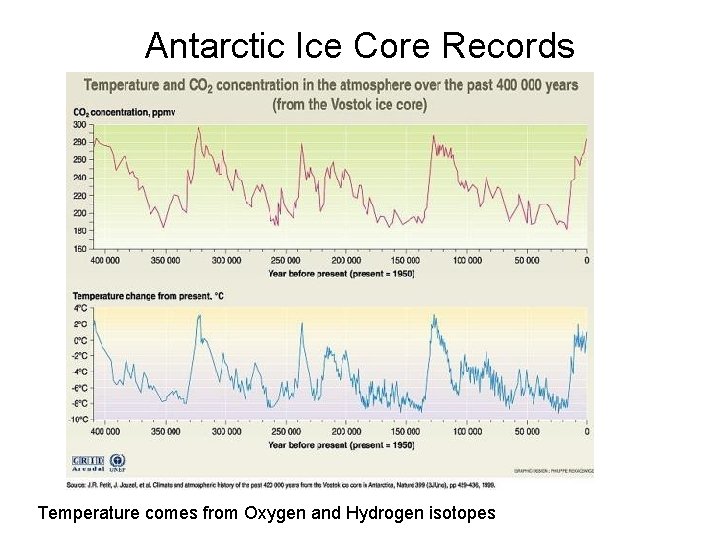
Antarctic Ice Core Records Temperature comes from Oxygen and Hydrogen isotopes

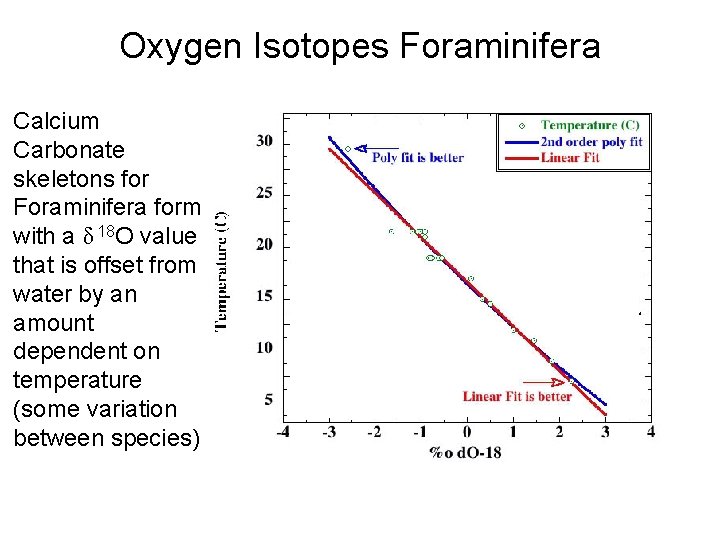
Oxygen Isotopes Foraminifera Calcium Carbonate skeletons for Foraminifera form with a δ 18 O value that is offset from water by an amount dependent on temperature (some variation between species)

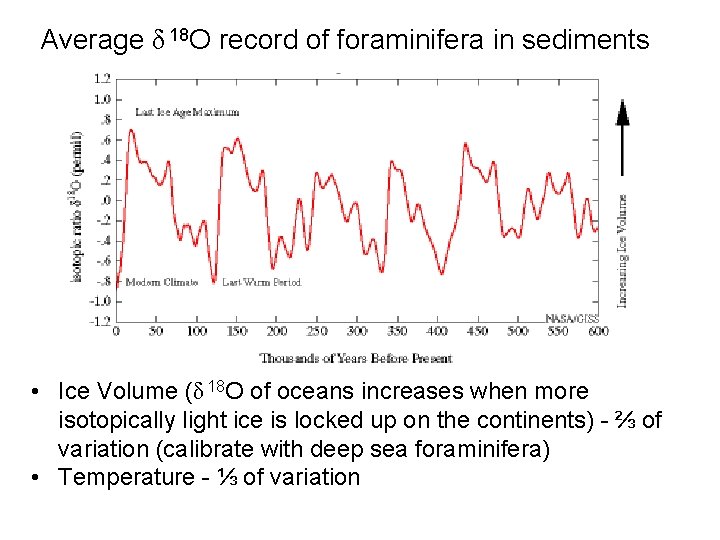
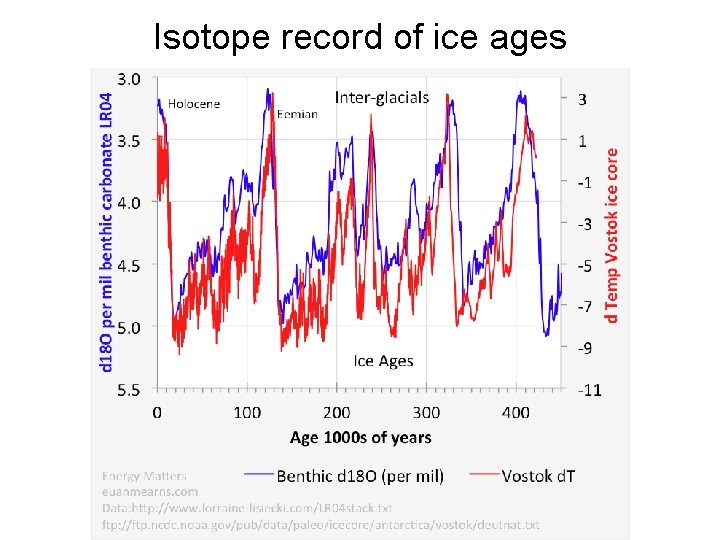
Average δ 18 O record of foraminifera in sediments • Ice Volume (δ 18 O of oceans increases when more isotopically light ice is locked up on the continents) - ⅔ of variation (calibrate with deep sea foraminifera) • Temperature - ⅓ of variation

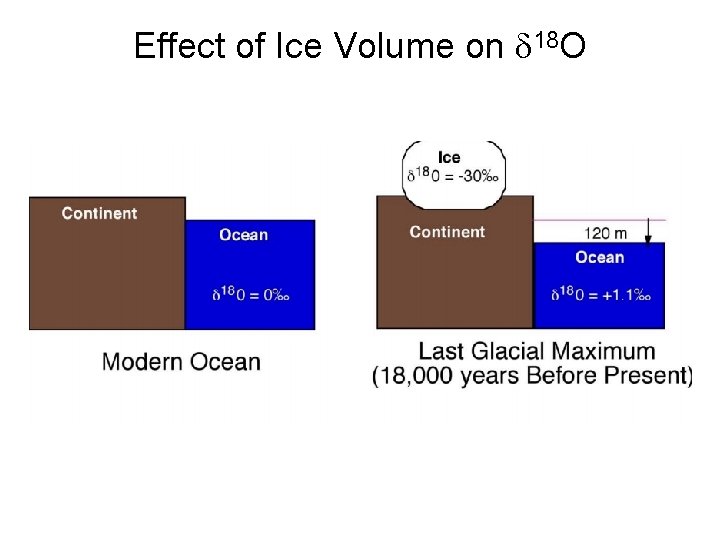
Effect of Ice Volume on d 18 O

δ 18 O of present day surface waters

Isotope record of ice ages

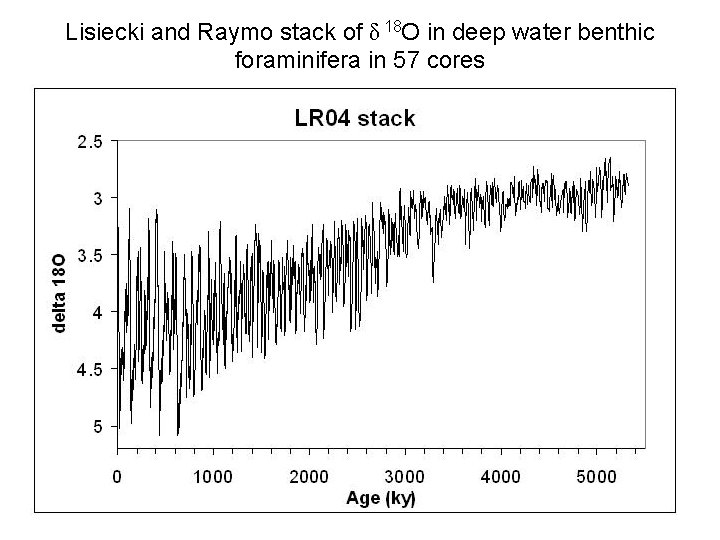
Lisiecki and Raymo stack of δ 18 O in deep water benthic foraminifera in 57 cores

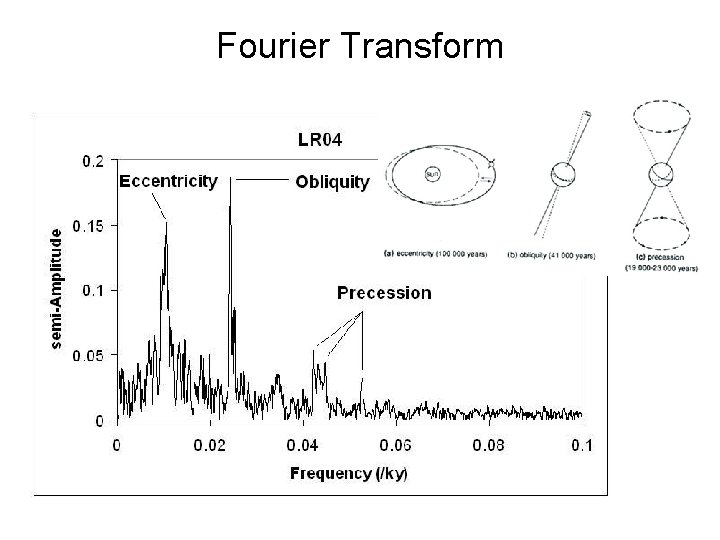
Fourier Transform

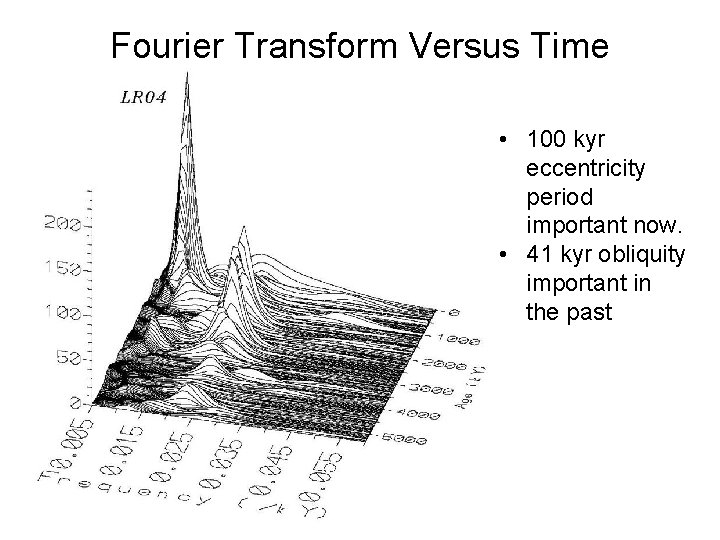
Fourier Transform Versus Time • 100 kyr eccentricity period important now. • 41 kyr obliquity important in the past