Math Review Units Scientific Notation Significant Figures and



































- Slides: 42

Math Review • Units, Scientific Notation, Significant Figures, and Dimensional analysis • Algebra – – Per Cent Change Solving simultaneous equations Cramers Rule Quadratic equation • Trigonometry and geometry – sin, cos, and tan, Pythagorean Theorem, Coversion to radians • Vectors – – – Unit vectors Adding, subtracting, finding components Dot product Cross product Examples • Derivatives – Rules – Examples • Integrals – Examples

The system of units we will use is the Standard International (SI) system; the units of the fundamental quantities are: • Length – meter • Mass – kilogram • Time – second • Charge - Coulomb

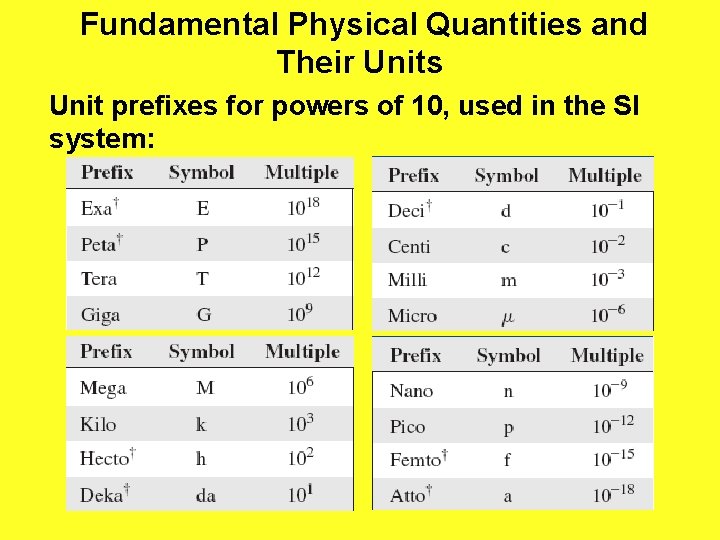
Fundamental Physical Quantities and Their Units Unit prefixes for powers of 10, used in the SI system:

Scientific Notation Scientific notation: use powers of 10 for numbers that are not between 1 and 10 (or, often, between 0. 1 and 100); exponents add if multiplying and subtract if dividing:

Accuracy and Significant Figures If numbers are written in scientific notation, it is clear how many significant figures there are: 6 × 1024 has one 6. 1 × 1024 has two 6. 14 × 1024 has three …and so on. Calculators typically show many more digits than are significant. It is important to know which are accurate and which are meaningless.

Other systems of units: cgs, which uses the centimeter, gram, and second as basic units British, which uses the foot for length, the second for time, and the pound force or weight – all of these units are now defined relative to the SI system.

Accuracy and Significant Figures The number of significant figures represents the accuracy with which a number is known. Terminal zeroes after a decimal point are significant figures: 2. 0 is between 1. 95 and 2. 05, whereas 2. 00 is between 1. 995 and 2. 005.

The number of significant figures represents the accuracy with which a number is known. Trailing zeroes with no decimal point are not significant. This has only 2 significant figures. 1200 is between 1150 and 1250, whereas 1200. is between 1199. 5 and 1200. 5.

Dimensional Analysis The dimension of a quantity is the particular combination that characterizes it (the brackets indicate that we are talking about dimensions): [v] = [L]/[T] Note that we are not specifying units here – velocity could be measured in meters per second, miles per hour, inches per year, or whatever.

Problems Involving Percent Change A cart is traveling along a track. As it passes through a photogate its speed is measured to be 3. 40 m/s. Later, at a second photogate, the speed of the cart is measured to be 3. 52 m/s. Find the percent change in the speed of the cart.

Simultaneous Equations FIND X AND Y

Cramer’s Rule

Quadratic Formula EQUATION: SOLVE FOR X: SEE EXAMPLE NEXT PAGE

Example

Derivation Complete the Square

Arc Length and Radians S is measured in radians

Small Angle Approximation Small-angle approximation is a useful simplification of the laws of trigonometry which is only approximately true for finite angles. FOR EXAMPLE

Scalars and Vectors

Vectors and Unit Vectors • Representation of a vector : has magnitude and direction – i and j unit vectors – angle and magnitude – x and y components • Example of vectors • Addition and subtraction • Scalar or dot product

Vectors Red arrows are the i and j unit vectors. Magnitude = Angle between A and x axis =

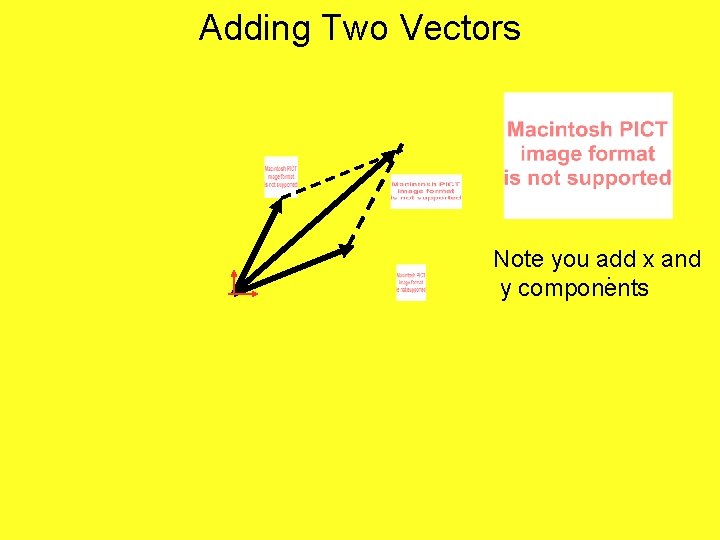
Adding Two Vectors Create a Parallelogram with The two vectors You wish you add.

Adding Two Vectors Note you add x and. y components

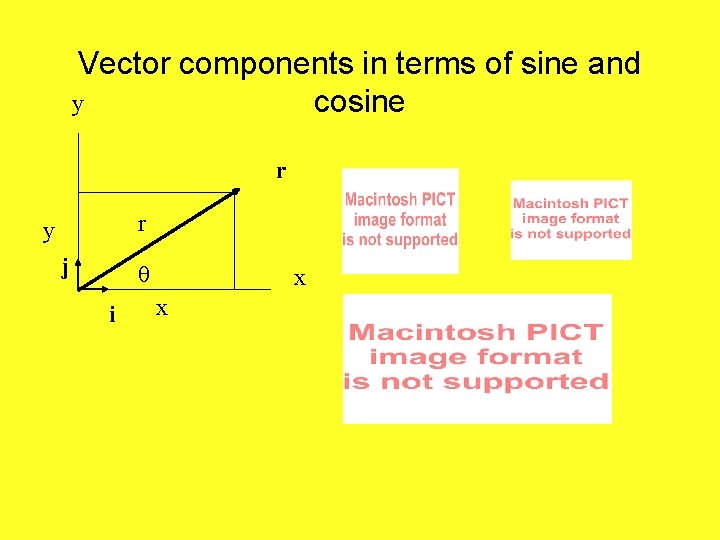
Vector components in terms of sine and y cosine r r y j i x x

Scalar product = A AB Also B

AB is the perpendicular projection of A on B. Important later. A B AB Also 90 deg.

Vectors in 3 Dimensions

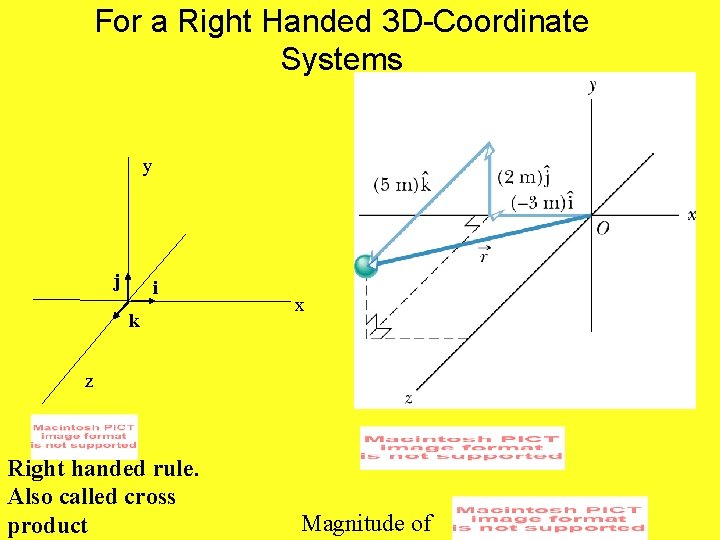
For a Right Handed 3 D-Coordinate Systems y j i k x z Right handed rule. Also called cross product Magnitude of

Suppose we have two vectors in 3 D and we want to add them y 2 j 5 i x k r 1 7 r 2 z 1

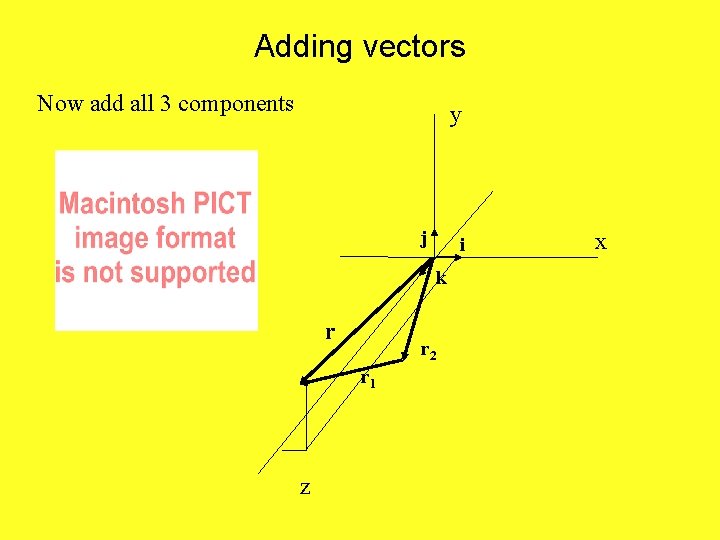
Adding vectors Now add all 3 components y j i k r r 2 r 1 z x

Scalar product = Cross Product See your textbook Chapter 3 for more information on vectors When we get to rotations we will need to talk about cross products. Also in E/M.

Differential Calculus

Definition of Velocity when it is smoothly changing Define the instantaneous velocity Recall (average) as Dt Example 0 = dx/dt (instantaneous)

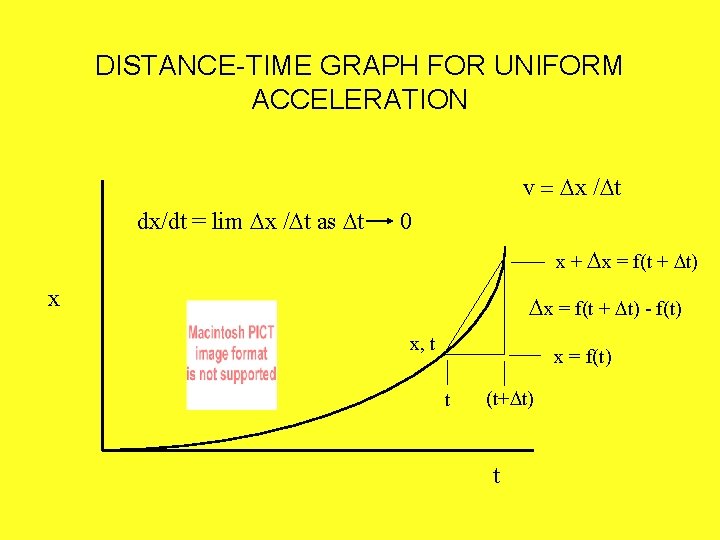
DISTANCE-TIME GRAPH FOR UNIFORM ACCELERATION v = Dx /Dt dx/dt = lim Dx /Dt as Dt 0 x + Dx = f(t + Dt) x Dx = f(t + Dt) - f(t) x, t . t x = f(t) (t+Dt) t

Differential Calculus: an example of a derivative dx/dt = lim Dx /Dt as Dt 0 velocity in the x direction

Three Important Rules of Differentiation Power Rule Product Rule Chain Rule

Problem 4 -7 The position of an electron is given by the following displacement vector , where t is in s and r is in m. What is the electron’s velocity v(t)? What is the electron’s velocity at t= 2 s? +vy 3 +vx f -16 What is the magnitude of the velocity or speed? What is the angle relative to the positive direction of the x axis?

Integral Calculus

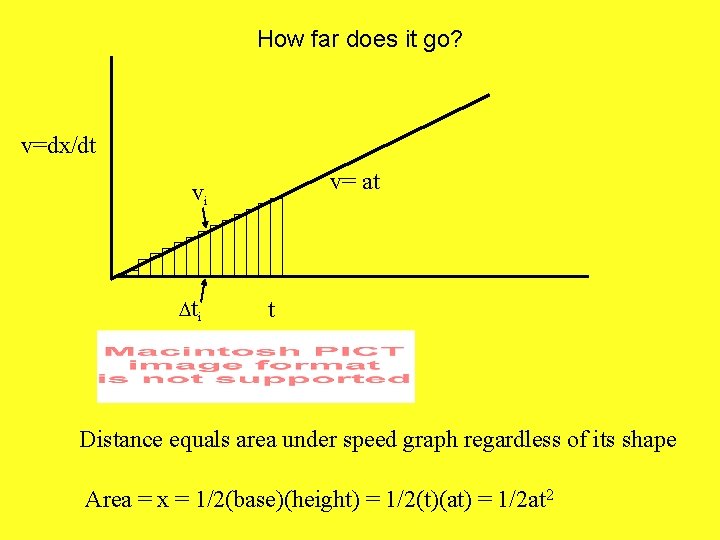
How far does it go? v=dx/dt v= at vi Dti t Distance equals area under speed graph regardless of its shape Area = x = 1/2(base)(height) = 1/2(t)(at) = 1/2 at 2

Integration: anti-derivative


Differentiation Practice QUESTION: Differentiate the following values with respect to x, t, or z. And let a and b be constants.

Integration Practice