Kaiser Permanente Research Human Papillomavirus Vaccine Safety and

























- Slides: 28

Kaiser Permanente Research Human Papillomavirus Vaccine Safety and Effectiveness Research in an Integrated Healthcare Delivery System Allison Naleway, Ph. D Center for Health Research Kaiser Permanente Northwest Oregon HPV Summit – June 11, 2019 © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Presentation Overview • Kaiser Permanente • Center for Health Research • Vaccine Safety Research • Vaccine Effectiveness Research • Questions/Discussion © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Kaiser Permanente • Founded by Henry Kaiser and Dr. Sidney Garfield • Opened to public enrollment July 1945 • Currently provides care in 8 regions • Northern California, Southern California, Northwest, Colorado, Mid-Atlantic, Georgia, Hawaii, Washington (formerly Group Health Cooperative) • ~14 million members • All share same EMR system, Health. Connect • All regions have research capabilities © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Kaiser Permanente Northwest • ~610, 000 members in southwest Washington/northwest Oregon • Portland metropolitan area • I-5 corridor from Eugene to Longview-Kelso • ~287, 000 members also have KP dental coverage • Own 2 hospitals, network of clinics, pharmacies, clinical laboratory • Shared electronic medical record © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Center for Health. Research Kaiser Permanente § Established in 1964 § Research centers in Portland § NW has 250 employees, including 40+ investigators § $45 million budget in 2018 § 85% of funds from external grants, most from federal government © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Vaccine Research at CHR Clinical Trials Safety Effectiveness/Impact Surveillance Interventions HPV Pregnant women Varicella/Herpes zoster Influenza © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research The Vaccine Safety Datalink (VSD) • Collaborative project between CDC and 8 US health systems (7 data-contributing) • Established in 1990 • Allows for planned, post-licensure vaccine safety studies, as well as: • Rapid safety assessments for routine and emergency vaccination campaigns • Monitoring changes in immunization patterns following schedule changes of the introduction of new vaccines • Informs national immunization policy (e. g. , ACIP presentations and work groups) © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH 7

Kaiser Permanente Research The Vaccine Safety Datalink (VSD) • Combined population of approximately 11 million people (~3% of the US) • Harvard Pilgrim participates as a subject matter site only (non data -contributing) © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH 8

Kaiser Permanente Research Rapid Surveillance for AEs following HPV Vaccination © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Rapid Surveillance for AEs following HPV Vaccination • Monitored VSD data weekly August 2006 - October 2009 • ~600, 600 HPV doses observed in 9 -26 year old females • Evaluated 8 pre-specified possible AEs: • Guillain-Barre syndrome • Stroke • VTE • Appendicitis • Anaphylaxis • Seizures* • Syncope* © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Rapid Surveillance for AEs following HPV Vaccination • Compared incidence of these outcomes to historical background rates using max. SPRT • More common outcomes were compared to unvaccinated females with preventive care visits • No increased risk of any outcome was observed • VTE RR was 1. 98 (not statistically significant) in 9 -17 year olds © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research HPV Vaccination and VTE © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

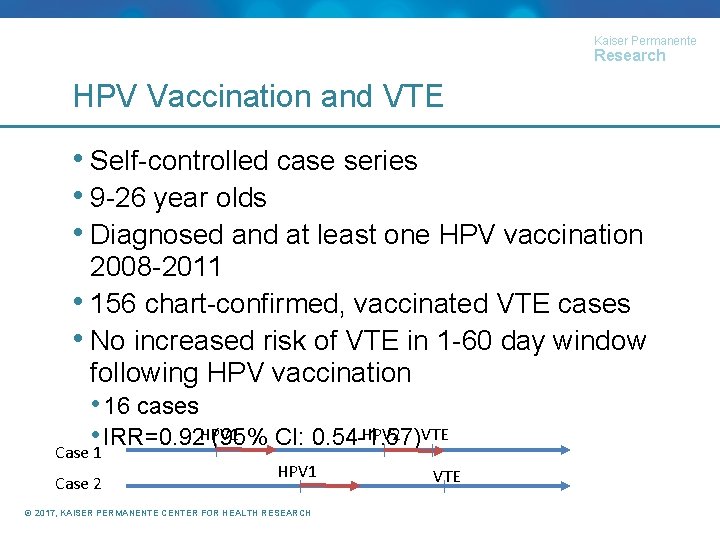
Kaiser Permanente Research HPV Vaccination and VTE • Self-controlled case series • 9 -26 year olds • Diagnosed and at least one HPV vaccination 2008 -2011 • 156 chart-confirmed, vaccinated VTE cases • No increased risk of VTE in 1 -60 day window following HPV vaccination • 16 cases HPV 2 VTE • IRR=0. 92 HPV 1 (95% CI: 0. 54 -1. 57) Case 1 Case 2 HPV 1 © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH VTE

Kaiser Permanente Research VIVE Study © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research VIVE Study • Surveyed 899 young women (11 -26 years of age) in 2008 within a few days of receiving first HPV dose • 78% reported pain at injection site • 17% bruising or discoloration at injection site • 14% swelling at injection site • 15% syncope or pre-syncope • These are commonly reported vaccine reactions • Syncope is especially an issue when © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research POI and Adolescent Vaccination © 2017, KAISER FOR HEALTH RESEARCHPMID 30131438 Naleway AL, et. PERMANENTE al. Pediatrics. CENTER 2018; 142(3): pii: e 20180943. 16

Kaiser Permanente Research POI and Adolescent Vaccination Background • Characterized by dysfunction or depletion of ovarian follicles, menopausal symptoms, and reduced fertility • Typically idiopathic, but may be associated with underlying autoimmune or infectious disease • Recent public concerns of a possible association between POI and HPV vaccine - stemming from published case series, media attention, and social media/internet content © 2017, KAISER FOR HEALTH RESEARCHPMID 30131438 Naleway AL, et. PERMANENTE al. Pediatrics. CENTER 2018; 142(3): pii: e 20180943. 17

Kaiser Permanente Research POI and Adolescent Vaccination Study Objectives • Identify and describe characteristics of idiopathic POI diagnosed in females aged 11 -34 years • Describe prevalence and age-specific incidence of idiopathic POI • Estimate the risk of idiopathic POI in females following HPV vaccination and other adolescent vaccinations © 2017, KAISER FOR HEALTH RESEARCHPMID 30131438 Naleway AL, et. PERMANENTE al. Pediatrics. CENTER 2018; 142(3): pii: e 20180943. 18

Kaiser Permanente Research POI and Adolescent Vaccination © 2017, KAISER FOR HEALTH RESEARCHPMID 30131438 Naleway AL, et. PERMANENTE al. Pediatrics. CENTER 2018; 142(3): pii: e 20180943. 19

Kaiser Permanente Research POI and Adolescent Vaccination Challenges • Time from symptom onset to diagnosis may be variable or long (median = 3 years); onset difficult to ascertain when cases present with primary amenorrhea • Diagnoses of POI may be difficult to accurately identify due to variability of testing practices within health care systems Conclusion • No evidence of increased risk of idiopathic POI was identified following HPV vaccine exposure or other routine adolescent vaccination Naleway AL, et al. Pediatrics 2018; 142(3): pii: e 20180943. PMID 30131438 © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH 20

Kaiser Permanente Research Other HPV Vaccine Safety Studies • In Process • Complex Regional Pain Syndrome (CRPS) • Postural Orthostatic Tachycardia Syndrome (POTS) • Chronic Fatigue Syndrome (CFS)/Myalgic Encephalomyelitis (ME) • Syncope and Syncope-related Injuries • Completed • Rapid Safety Monitoring of 9 v. HPV • Safety of Inadvertent HPV Vaccine Exposure during Pregnancy • HPV Vaccination and Autoimmune Disease Diagnoses © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Measuring HPV Vaccine Effectiveness • 20 -30 years between HPV exposure and cervical cancer diagnosis • Need intermediary outcomes to evaluate effectiveness • HPV infection • Anogenital warts • Cervical cancer precursor lesions © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research HPV Vaccine Impact Study (Hi. TS) © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Hi. TS (paper currently under review) • HPV prevalence in Pap test specimens • Collection periods: 2007, 2012 -13, 2015 -16 • CDC lab performed HPV testing • VT-HPV prevalence (6, 11, 16, 18) decreased • 78% in 20 -24 year old females • 38% in 25 -29 year old females • Decreases observed in vaccinated and unvaccinated suggesting both direct and herd protection © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

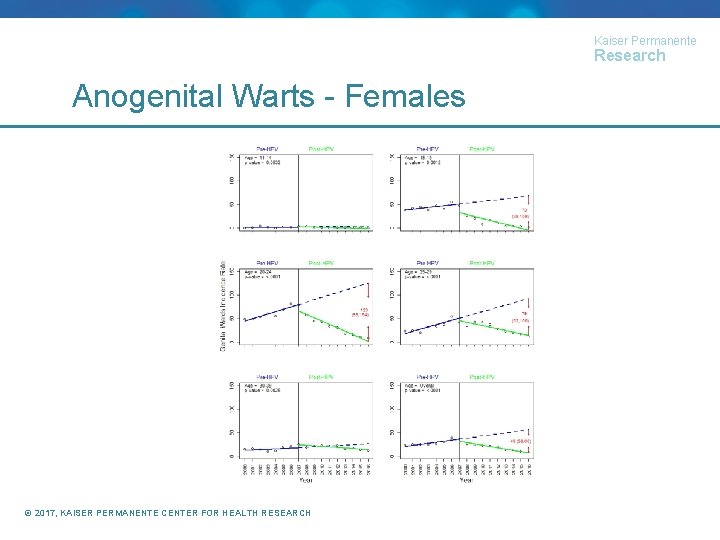
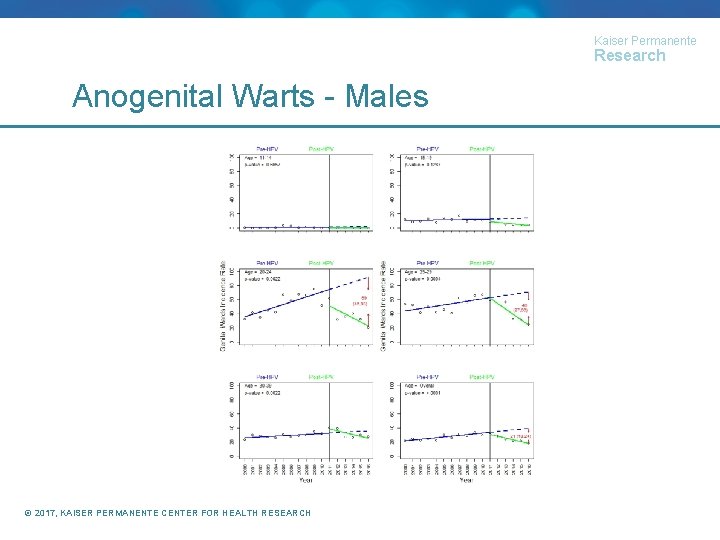
Kaiser Permanente Research Anogenital Wart Incidence (paper under review) • Identified members 11 -39 years of age with at least one calendar year of enrollment • Calculated AGW incidence by age group, calendar year, and gender • Conducted interrupted time series using segmented regression to compare AGW incidence in the periods before and after HPV recommendations • Females: 2000 -2006 vs 2007 -2016 • Males: 2000 -2010 vs 2011 -2016 © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Anogenital Warts - Females © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Anogenital Warts - Males © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH

Kaiser Permanente Research Conclusions • HPV vaccine is safe and effective • The data and resources available to us at KP allow us to address important and wide-ranging topics in immunization safety, effectiveness, and delivery • Thank you! • Questions? Discussion? © 2017, KAISER PERMANENTE CENTER FOR HEALTH RESEARCH