HAHS Avogadro The Mole and Grams The Mole











- Slides: 14

HAHS Avogadro, The Mole, and Grams

The Mole The amount of substance that contains as many particles as there atoms in exactly 12 g of carbon. We can think of the mole is a counting unit for chemists. Examples: 1 dozen =12 1 ream of paper = 500 sheets

Avogadro’s Number Is the number of particles in exactly one mole of a pure substance. 6. 02 x 1023 is called “Avogadro’s Number” in honor of the Italian chemist Amadeo Avogadro (1776 -1855). I didn’t discover it. Its just named after me!

Avogadro’s Number (con’t) There is 60200000000000 atoms in 1 mole of a substance THAT’S A GIGANTIC NUMBER!!!!!

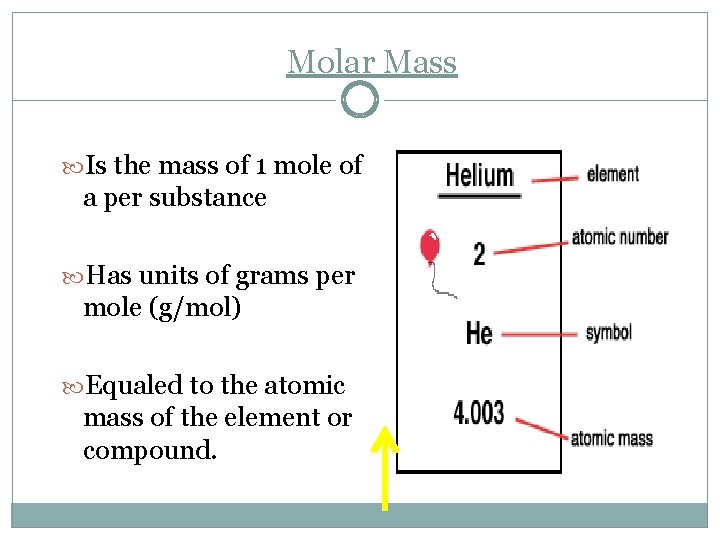
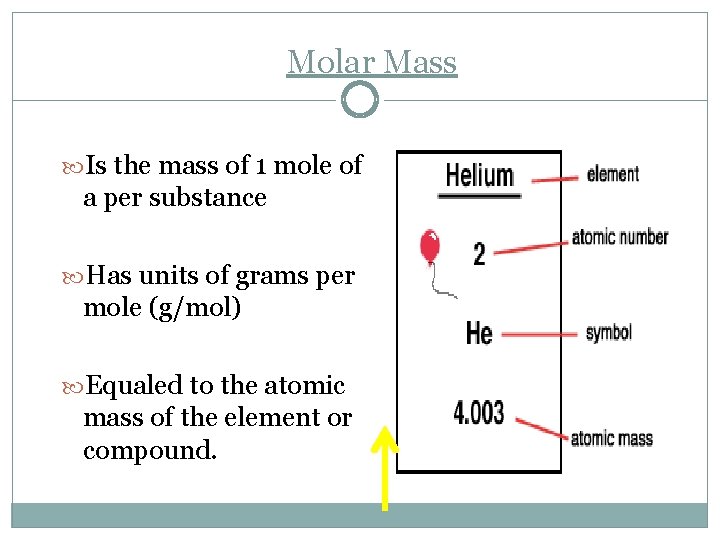
Molar Mass Is the mass of 1 mole of a per substance Has units of grams per mole (g/mol) Equaled to the atomic mass of the element or compound.

Molar Mass of a Compound Add up all the masses of each type of atom in the compound. Example: Molar Mass of Ca(OH)2 Ca: 1 x 40 = 40 O: 2 x 16 = 32 H: 2 x 1 = 2 The molar mass is the sum (40 + 32 + 2) which is equaled to 74 g/mol

Molar Mass Is the mass of 1 mole of a per substance Has units of grams per mole (g/mol) Equaled to the atomic mass of the element or compound.

Calculations: Converting moles to grams Given # of mole X g (look at periodic table)= 1 mole g of How many grams of lithium are in 3. 50 moles of lithium? 3. 50 mole X 6. 941 g = 24. 29 g Li 1 mol

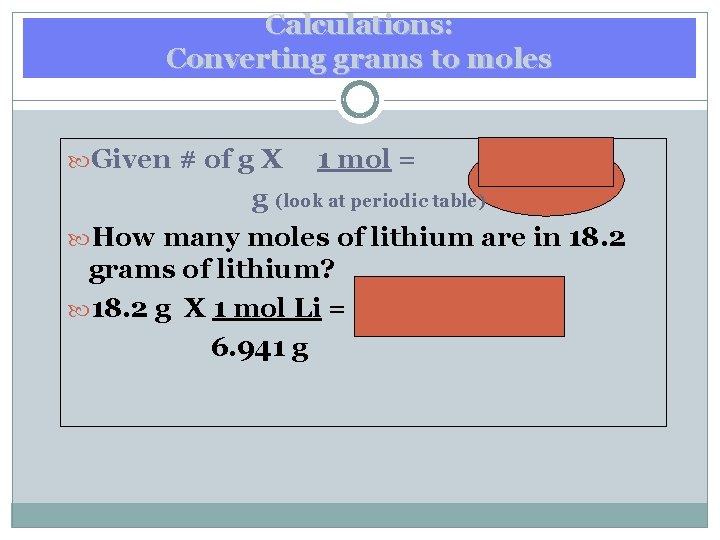
Calculations: Converting grams to moles Given # of g X 1 mol = mol of g (look at periodic table) How many moles of lithium are in 18. 2 grams of lithium? 18. 2 g X 1 mol Li = 2. 622 mol Li 6. 941 g

Calculations: Converting Moles to Particles Given # of mol x 6. 02 x 1023 part= atoms 1 mol How many atoms/particles/molecules of lithium are in 3. 50 moles of lithium? 3. 50 mole X 6. 02 x 1023 = 2. 11 x 1024 atoms of Li 1 mol

Calculations: Converting Particles to Moles Given # of particles x 1 mole = mol 6. 02 x 1023 How many moles of lithium are there in 1. 2044 x 1024 particles of Li? 1. 2044 x 1024 part x 1 mole = 2. 0 mol Li 6. 02 x 1023 part

Calculations: Converting grams to particles Given # of grams x 1 mol x 6. 022 x 1023= particles ? g 1 mol How many atoms/particles/molecules of lithium are in 18. 2 g of lithium? 18. 2 g x 1 mol x 6. 022 x 1023 = 1. 58 x 1024 particle Li 6. 941 g 1 mol

Calculations: Converting particles to grams Given # of particles x 1 mol x ? g= g 6. 022 x 1023 1 mol 25 particles How many grams are there in 8. 02 x 10 How many grams of lithium are in 3. 50 moles of lithium?

How many grams of lithium are in 3. 50 moles of lithium? How many moles are in 9. 8 grams of Na. Cl? How many ions/atoms/particles are in 5. 0 moles of H 2 O? How many moles are in 7. 8 x 10 particles of Cu? 27