Glargine Lantus 1562007 Dr HK Pang Endocrinologist Endocrine









- Slides: 15

Glargine (Lantus®) 15/6/2007. Dr. HK Pang. Endocrinologist, Endocrine team. Department of medicine. PYNEH

What is insulin Glargine (Lantus®) • A type of modified insulin. 1. Glargine (Lantus®) 2. Detemir ( Levemir®) – with property of the “Ideal” Long-Acting Insulin.

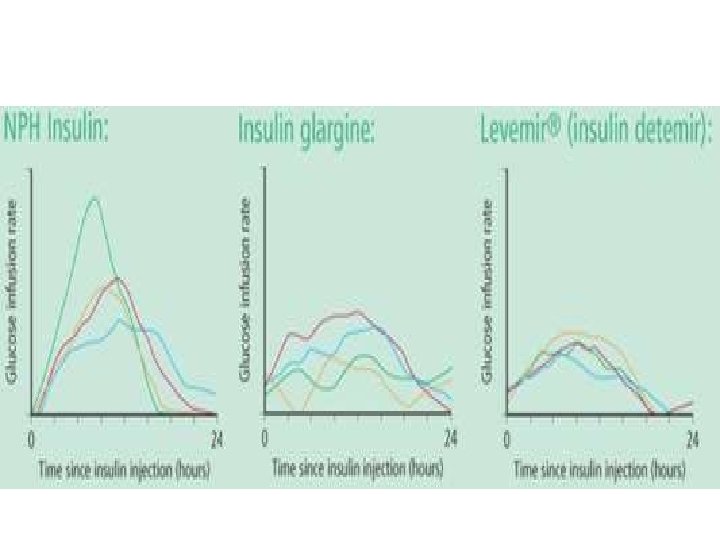
What is the “Ideal” Long-Acting Insulin? • Should have the property of – 1 injection daily 24 hours basal insulin coverage. – No peaks (peakless) provide an ideal, constant level of basal insulin – mimicks the physiological basal insulin level.

“Older” generation of insulin with peak, and cannot last for 24 hrs……

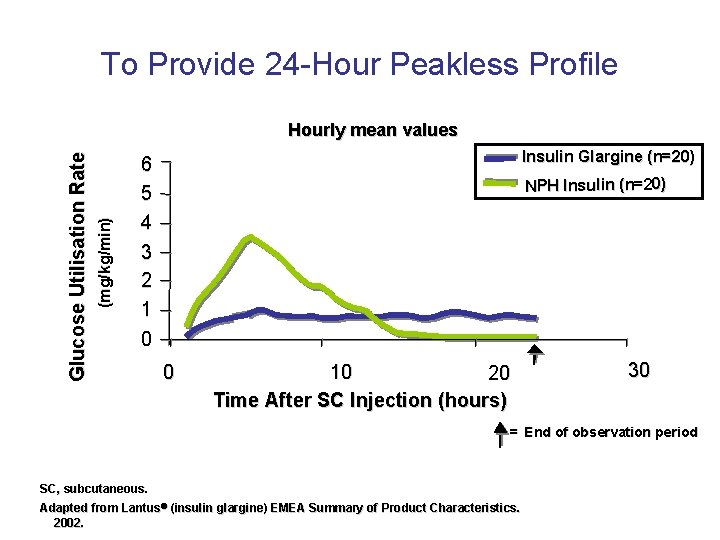
To Provide 24 -Hour Peakless Profile (mg/kg/min) Glucose Utilisation Rate Hourly mean values Insulin Glargine (n=20) 6 5 4 3 2 1 0 NPH Insulin (n=20) 0 10 20 Time After SC Injection (hours) 30 = End of observation period SC, subcutaneous. Adapted from Lantus (insulin glargine) EMEA Summary of Product Characteristics. 2002.

1. Insulin Glargine (Lantus®)

Glargine (Lantus®) • Has the property of an “ideal” basal insulin – A recombinant human insulin analogue 1 • A basal (long-acting) insulin 1 • Relatively constant peakless profile over 24 hours 1, 2 • Once-daily SC administration 1 1. Lantus® (insulin glargine) EMEA Summary of Product Characteristics. 2002. ® 2. Lantus receives European approval for pediatric use. Aventis Pharma Web site. Available at: http: //www. aventis. no/nyheter/nyheter_lantus_eu_approval_pediatric. shtml. Accessed March 19, 2003.

Glargine (Lantus®) • Modification of the insulin molecule makes it acquires the property of the “ ideal”. …

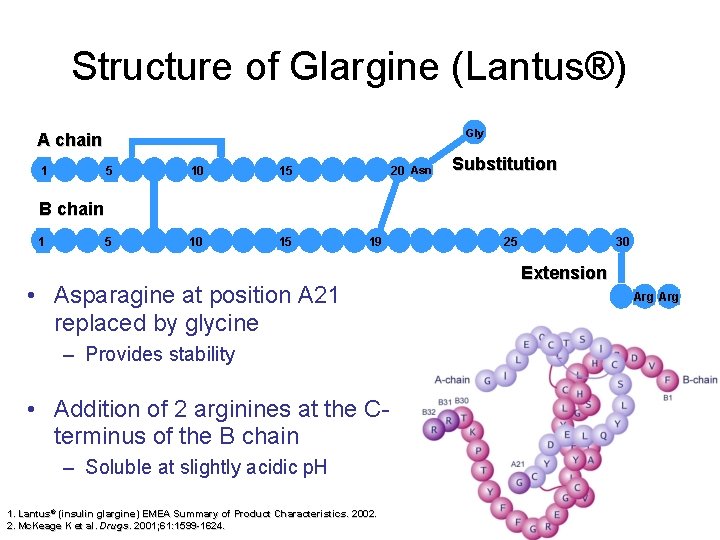
Structure of Glargine (Lantus®) Gly A chain 1 5 10 15 5 10 10 15 15 20 Asn Substitution B chain 1 19 • Asparagine at position A 21 replaced by glycine – Provides stability • Addition of 2 arginines at the Cterminus of the B chain – Soluble at slightly acidic p. H 1. Lantus® (insulin glargine) EMEA Summary of Product Characteristics. 2002. 2. Mc. Keage K et al. Drugs. 2001; 61: 1599 -1624. 25 30 Extension Arg

Mechanism of Action - Glargine (Lantus®) The mechanics of sustained release 1, 2 Injection of an acidic solution (p. H 4. 0)3 Microprecipitation of insulin glargine in subcutaneous tissue (p. H 7. 4)3 Slow dissolution of free insulin glargine hexamers from microprecipitates (stabilised aggregates)3 Protracted action 3 Balance state at day 2 with 1 injection/day and without accumulation 1. Lantus® (insulin glargine) EMEA Summary of Product Characteristics. 2002. 2. Mc. Keage K et al. Drugs. 2001; 61: 1599 -1624. 3. Kramer W. Exp Clin Endocrinol Diabetes. 1999; 107(suppl 2): S 52 -S 61.

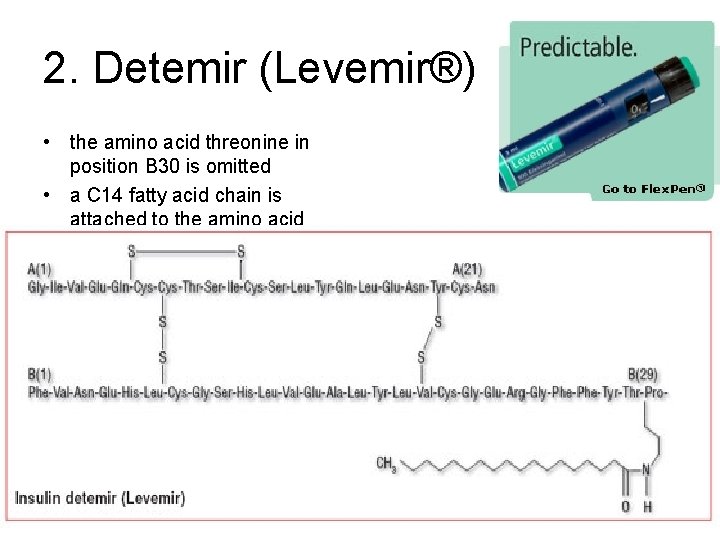
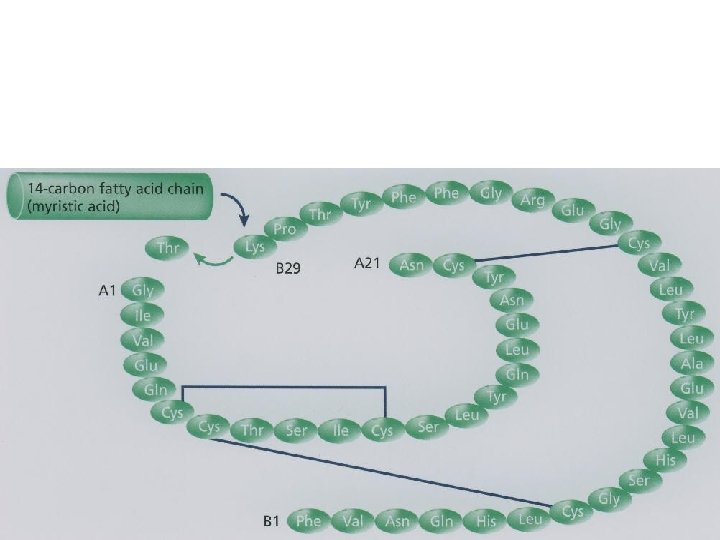
2. Detemir (Levemir®) • the amino acid threonine in position B 30 is omitted • a C 14 fatty acid chain is attached to the amino acid B 29, imparting its long-acting properties


2. Detemir (Levemir®) 1. Form depot - slow systemic absorption from the injection site 2. detemir is highly bound to albumin - slow distribution.


END Thanks