Exam 3 Review Session Review of Practice Exam








- Slides: 38

Exam 3 Review Session Review of Practice Exam and Concepts

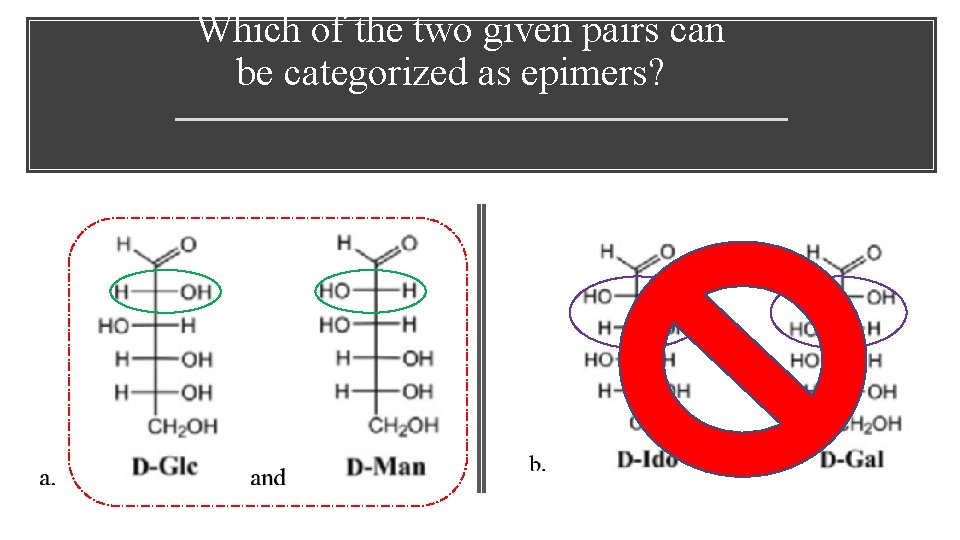
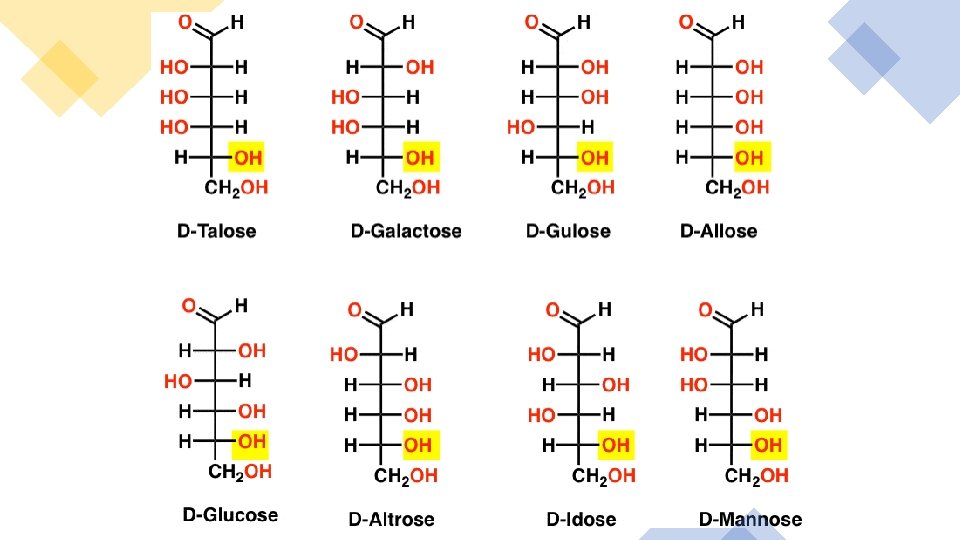
Which of the two given pairs can be categorized as epimers?

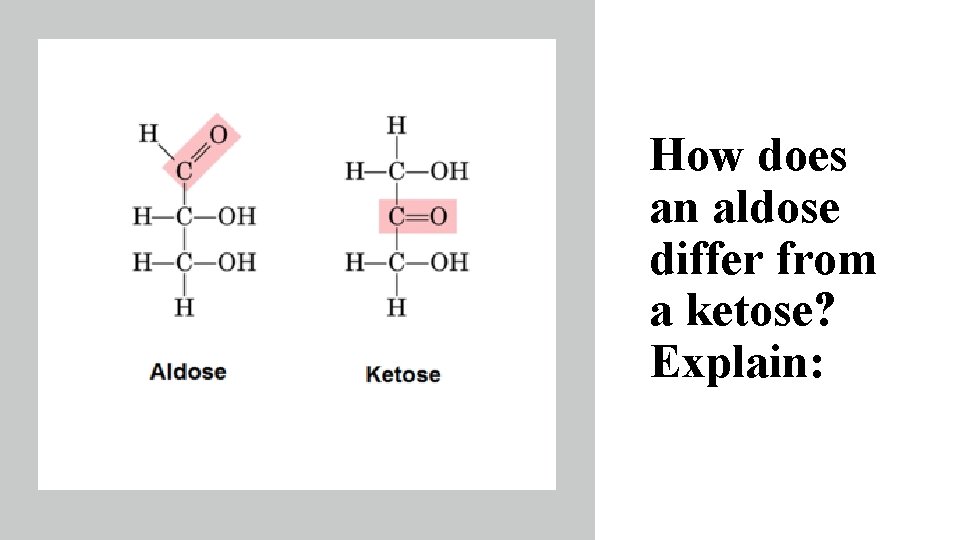
How does an aldose differ from a ketose? Explain:

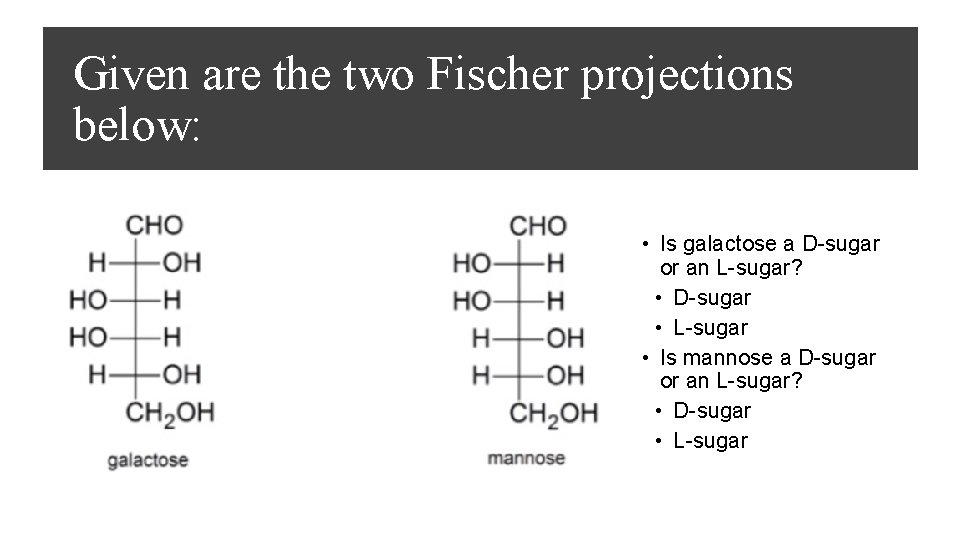
Given are the two Fischer projections below: • Is galactose a D-sugar or an L-sugar? • D-sugar • L-sugar • Is mannose a D-sugar or an L-sugar? • D-sugar • L-sugar

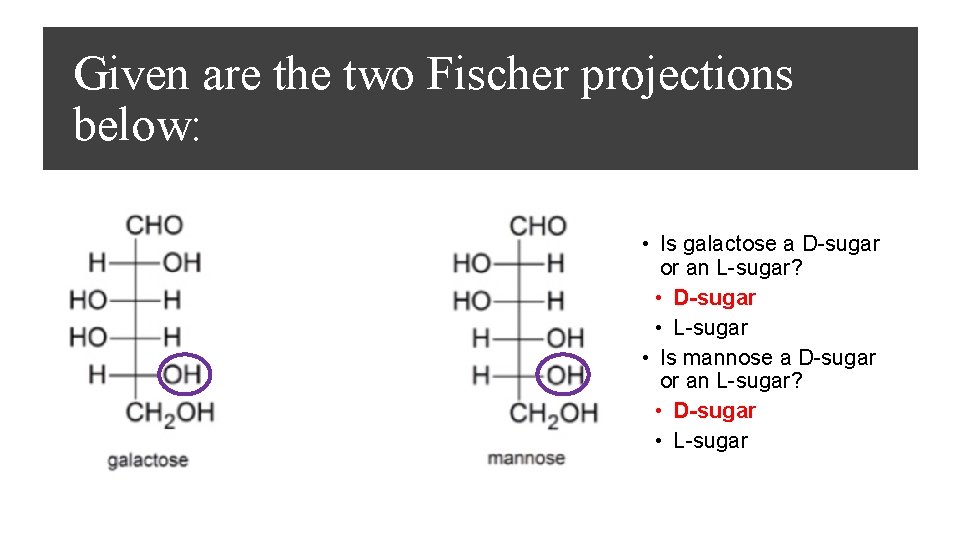
Given are the two Fischer projections below: • Is galactose a D-sugar or an L-sugar? • D-sugar • L-sugar • Is mannose a D-sugar or an L-sugar? • D-sugar • L-sugar


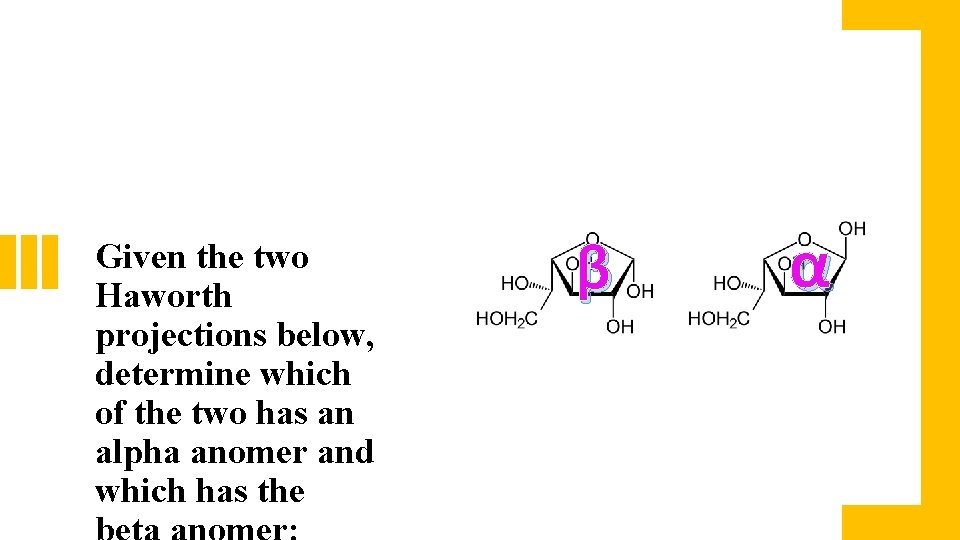
Given the two Haworth projections below, determine which of the two has an alpha anomer and which has the β α

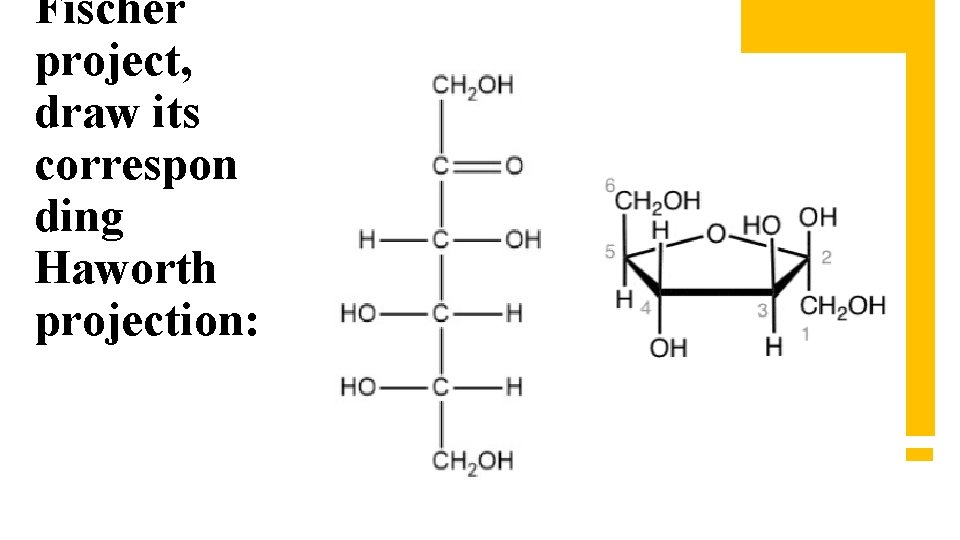
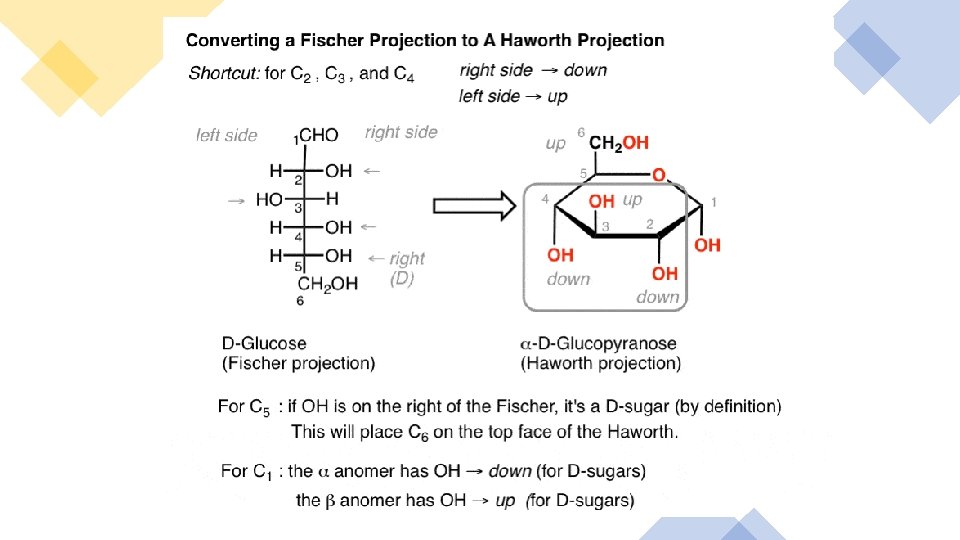
Fischer project, draw its correspon ding Haworth projection:


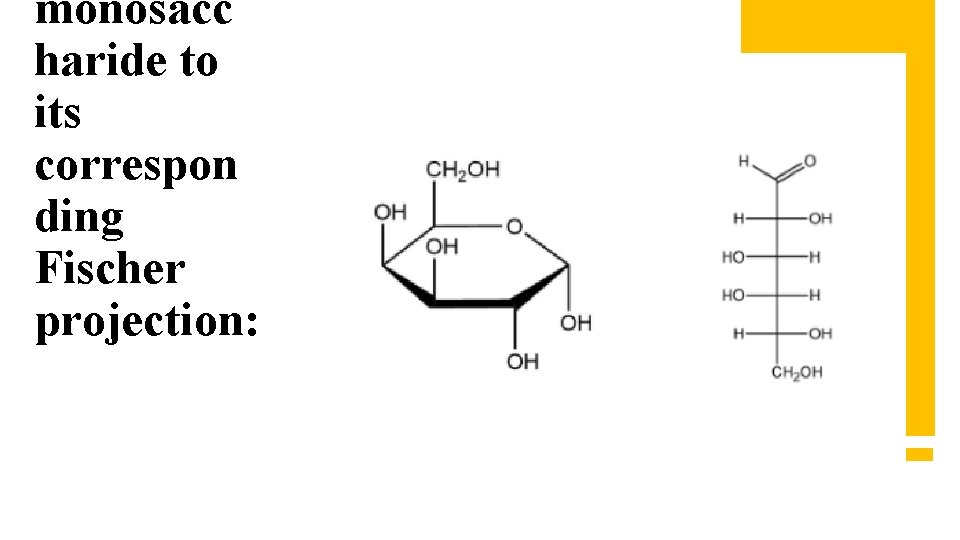
monosacc haride to its correspon ding Fischer projection:

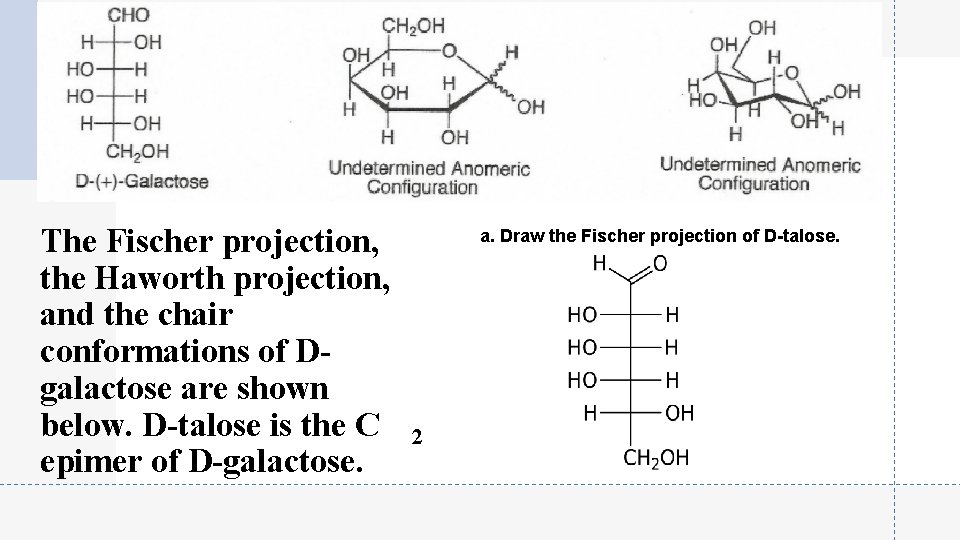
The Fischer projection, the Haworth projection, and the chair conformations of Dgalactose are shown below. D-talose is the C epimer of D-galactose. a. Draw the Fischer projection of D-talose. 2

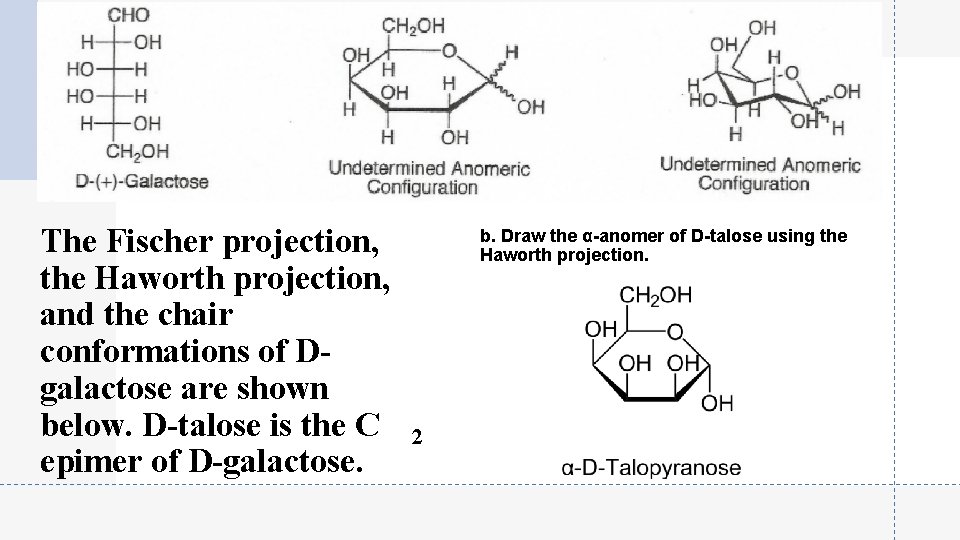
The Fischer projection, the Haworth projection, and the chair conformations of Dgalactose are shown below. D-talose is the C epimer of D-galactose. b. Draw the α-anomer of D-talose using the Haworth projection. 2

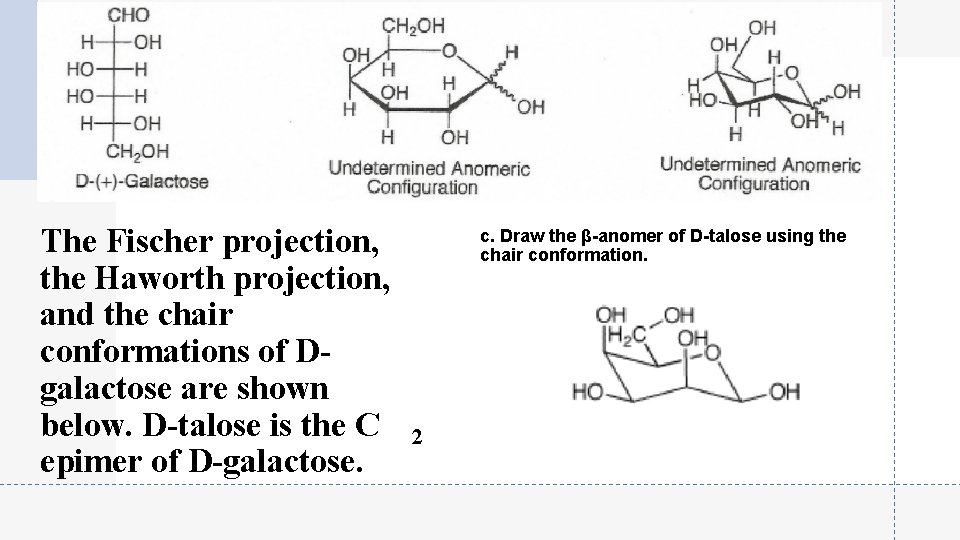
The Fischer projection, the Haworth projection, and the chair conformations of Dgalactose are shown below. D-talose is the C epimer of D-galactose. c. Draw the β-anomer of D-talose using the chair conformation. 2

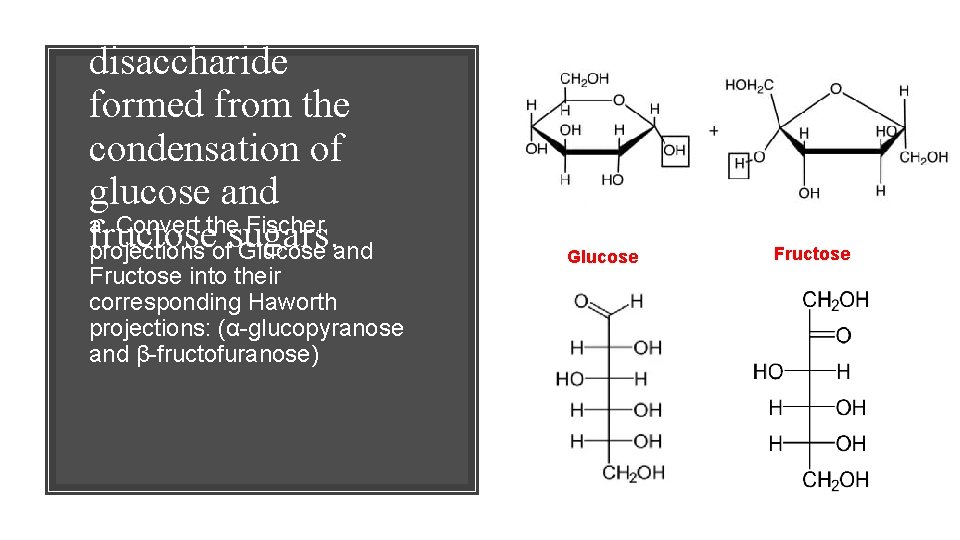
Sucrose is a disaccharide formed from the condensation of glucose and a. Convert the Fischer fructose projections ofsugars. Glucose and Fructose into their corresponding Haworth projections: (α-glucopyranose and β-fructofuranose) Glucose Fructose

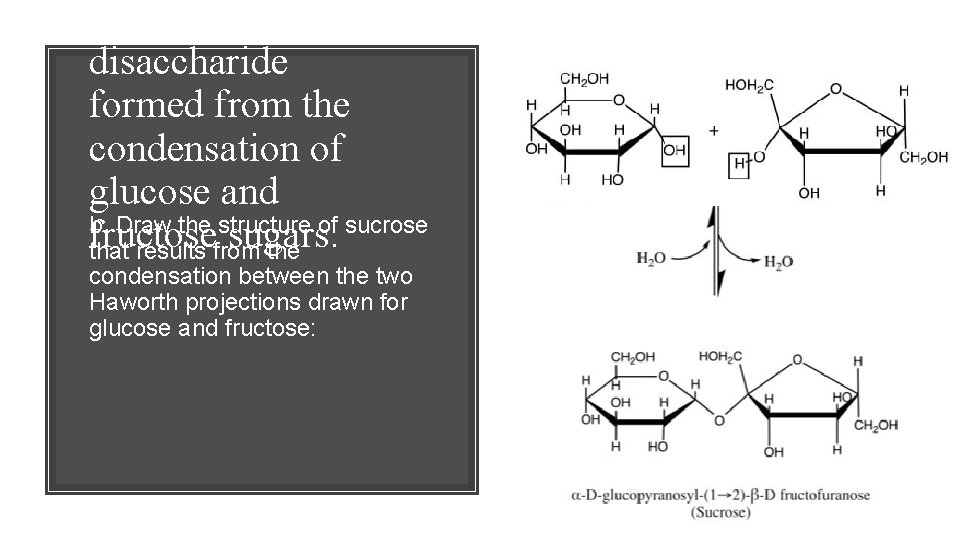
Sucrose is a disaccharide formed from the condensation of glucose and b. Draw the structure of sucrose fructose sugars. that results from the condensation between the two Haworth projections drawn for glucose and fructose:

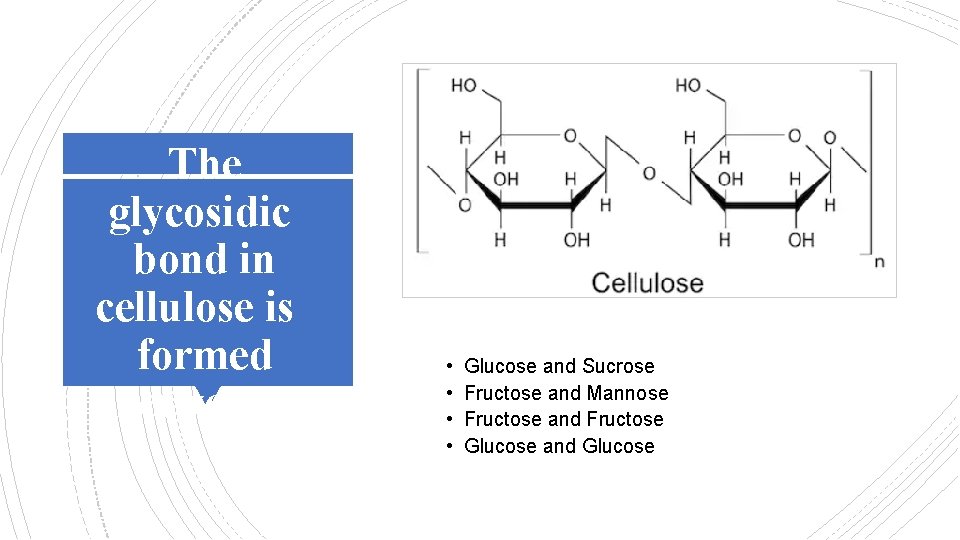
The glycosidic bond in cellulose is formed between: • • Glucose and Sucrose Fructose and Mannose Fructose and Fructose Glucose and Glucose

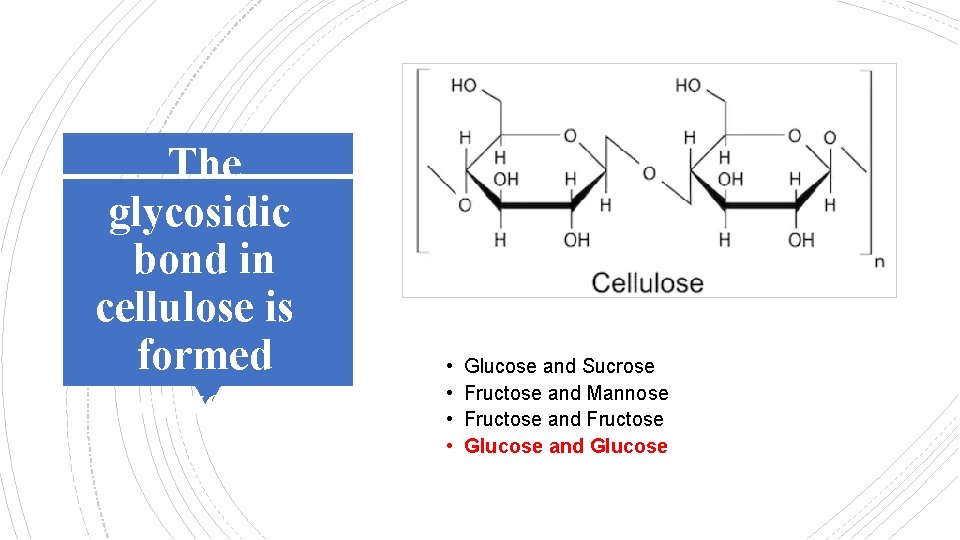
The glycosidic bond in cellulose is formed between: • • Glucose and Sucrose Fructose and Mannose Fructose and Fructose Glucose and Glucose

Cellulose and amylose Starch is a mixture composed of: Amylopectin and amylose Cellulose and chitin Amylopectin and keratin

Cellulose and amylose Starch is a mixture composed of: Amylopectin and amylose Cellulose and chitin Amylopectin and keratin

Oxidation of an aldose converts its aldehyde group to a carboxylic acid group. True False

Oxidation of an aldose converts its aldehyde group to a carboxylic acid group. True False

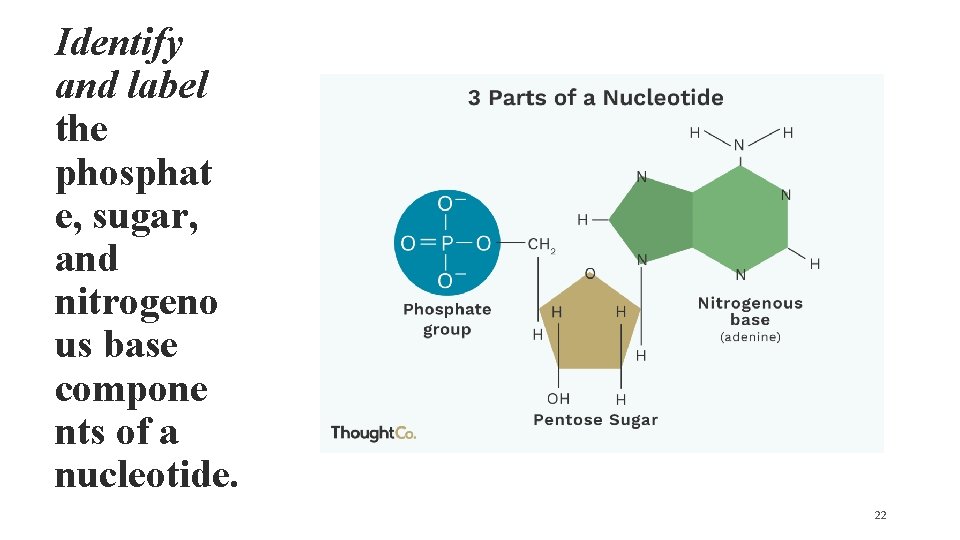
Identify and label the phosphat e, sugar, and nitrogeno us base compone nts of a nucleotide. 22

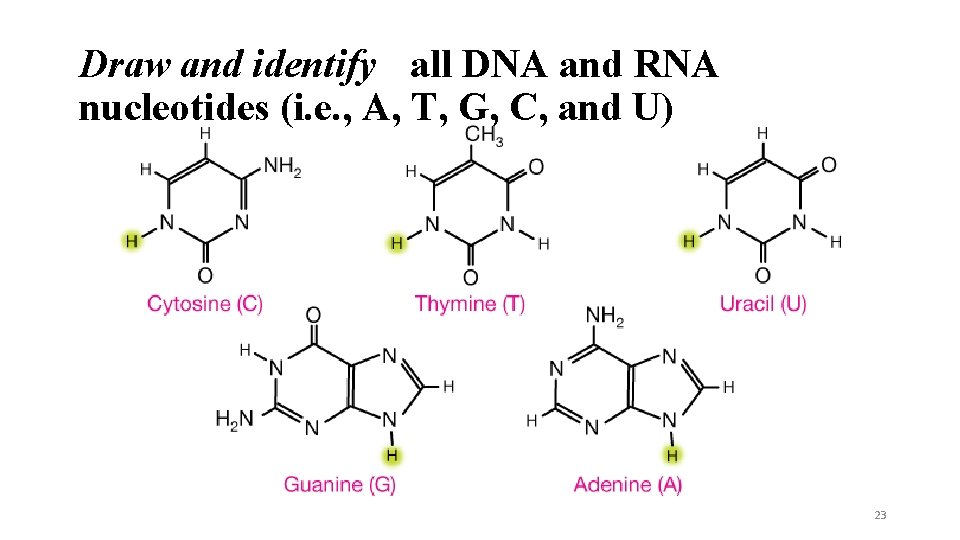
Draw and identify all DNA and RNA nucleotides (i. e. , A, T, G, C, and U) 23

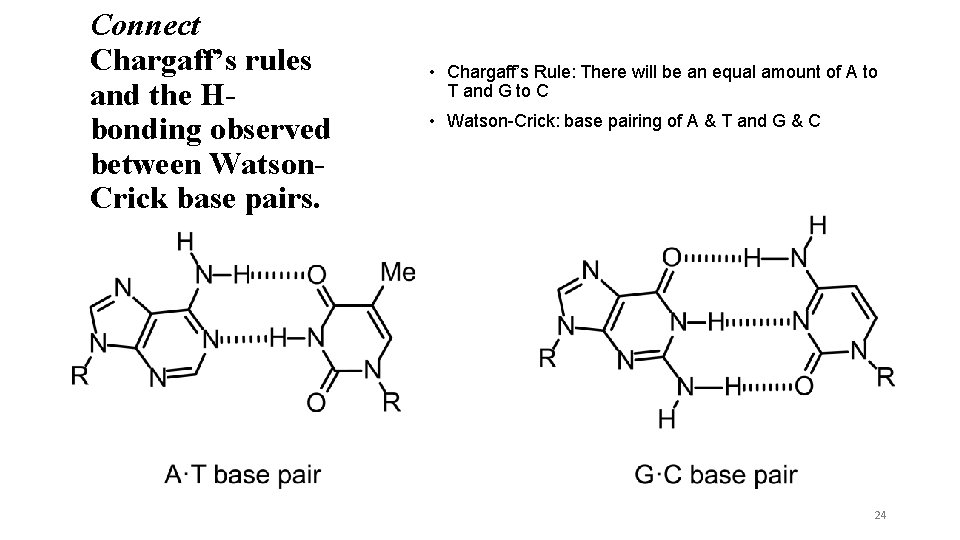
Connect Chargaff’s rules and the Hbonding observed between Watson. Crick base pairs. • Chargaff’s Rule: There will be an equal amount of A to T and G to C • Watson-Crick: base pairing of A & T and G & C 24

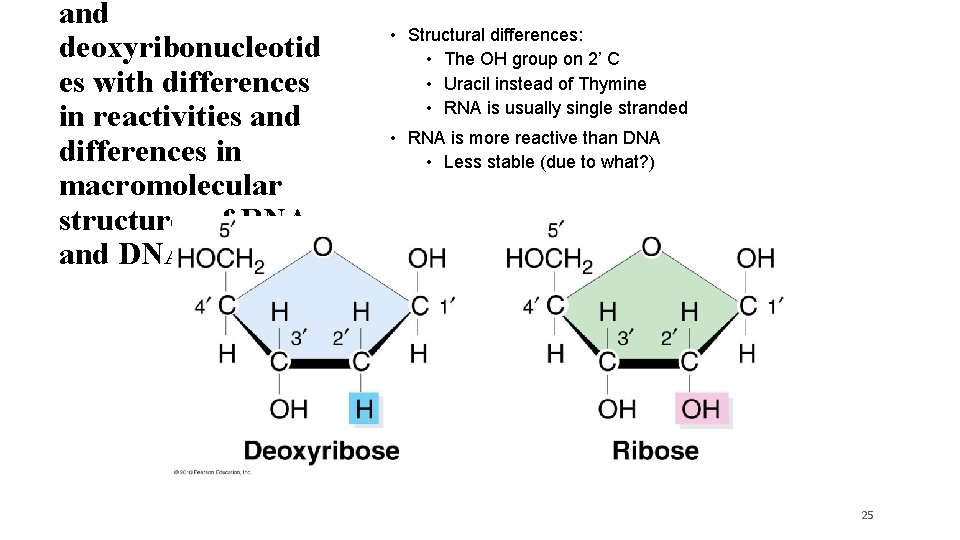
and deoxyribonucleotid es with differences in reactivities and differences in macromolecular structures of RNA and DNA. • Structural differences: • The OH group on 2’ C • Uracil instead of Thymine • RNA is usually single stranded • RNA is more reactive than DNA • Less stable (due to what? ) 25

Interpret DNA electrophoresis data used for DNA sequencing (Sanger method). 26

Design pri mers for DNA amplificatio n by PCR. • The oligonucleotide: (5’-GACTACCACG CTTCCATTAA GCTTACC) was cleaved usingthe restriction enzyme Hind. III, which has a cleavage site at A//AGCTT. a. Cleavage results in two fragments; write the sequence of the shortest fragment. b. Design a 3 nt primer 27

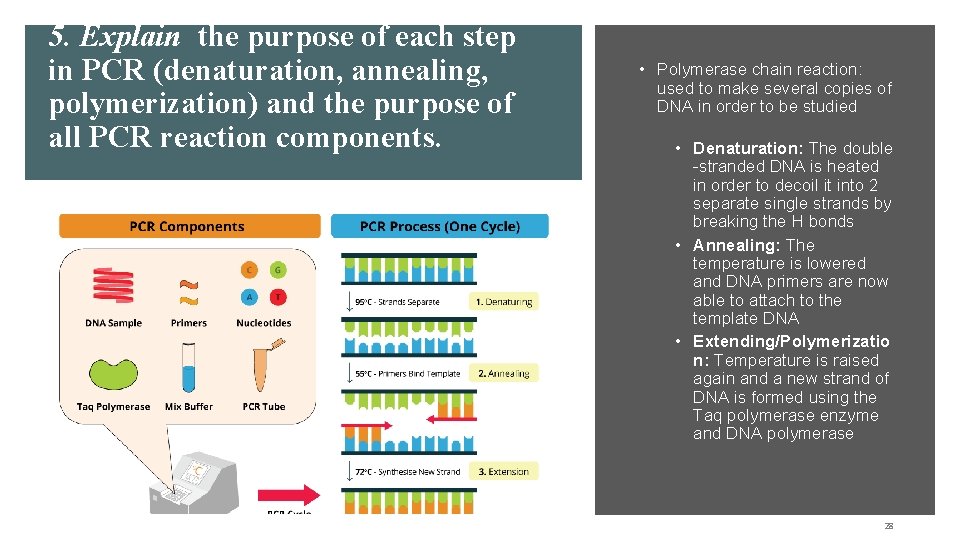
5. Explain the purpose of each step in PCR (denaturation, annealing, polymerization) and the purpose of all PCR reaction components. • Polymerase chain reaction: used to make several copies of DNA in order to be studied • Denaturation: The double -stranded DNA is heated in order to decoil it into 2 separate single strands by breaking the H bonds • Annealing: The temperature is lowered and DNA primers are now able to attach to the template DNA • Extending/Polymerizatio n: Temperature is raised again and a new strand of DNA is formed using the Taq polymerase enzyme and DNA polymerase 28

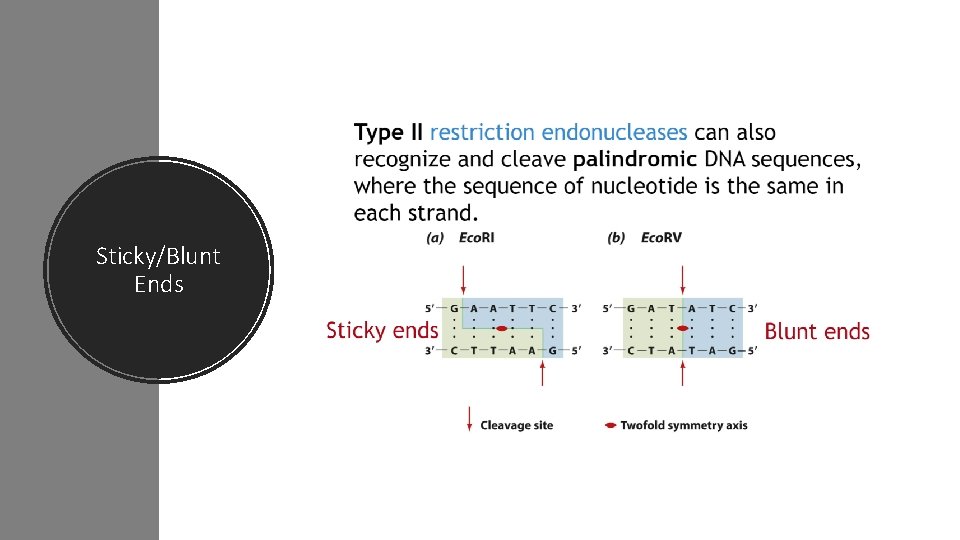
Sticky/Blunt Ends

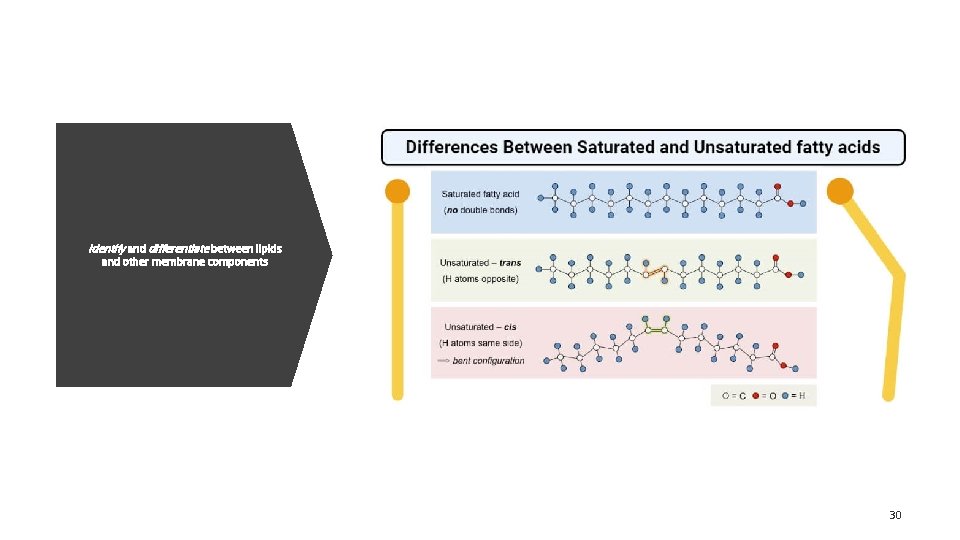
Identify and differentiate between lipids and other membrane components 30

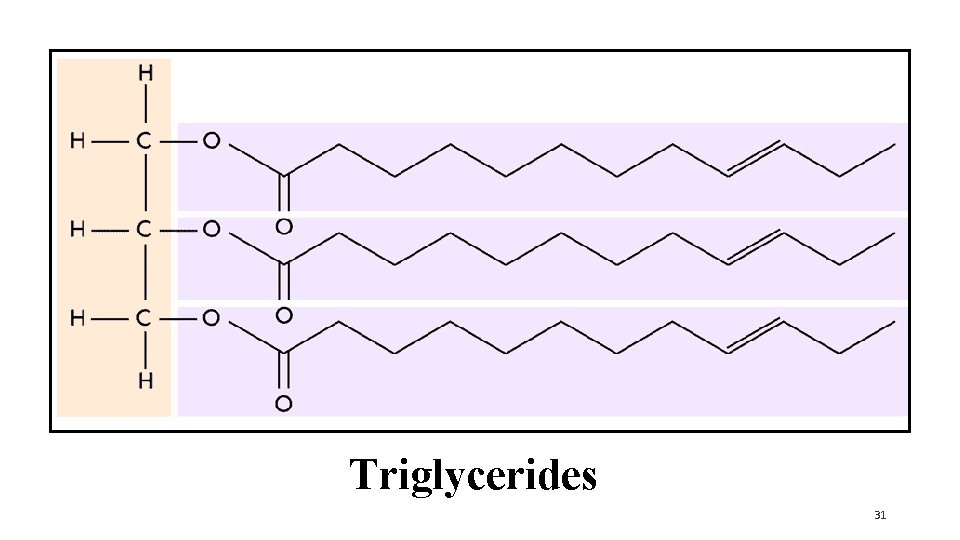
Triglycerides 31

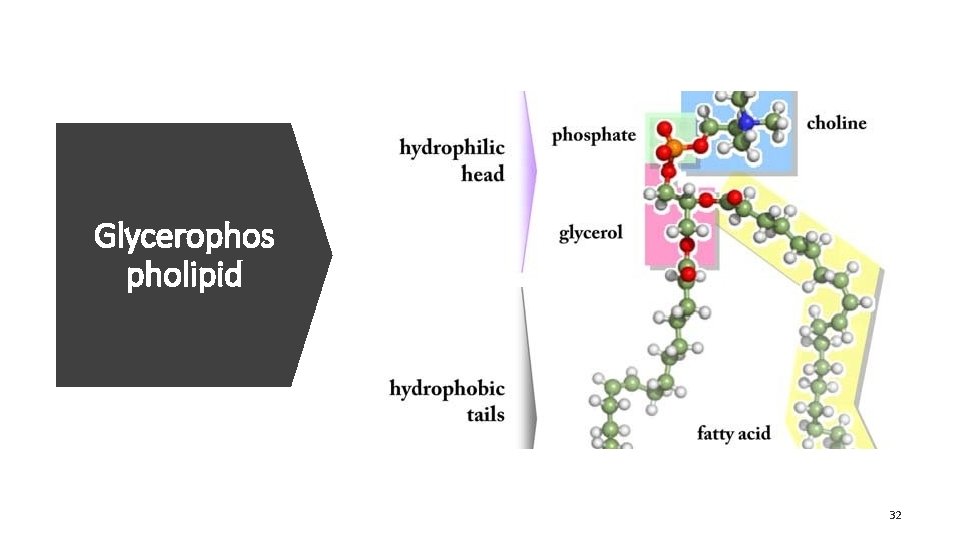
Glycerophos pholipid 32

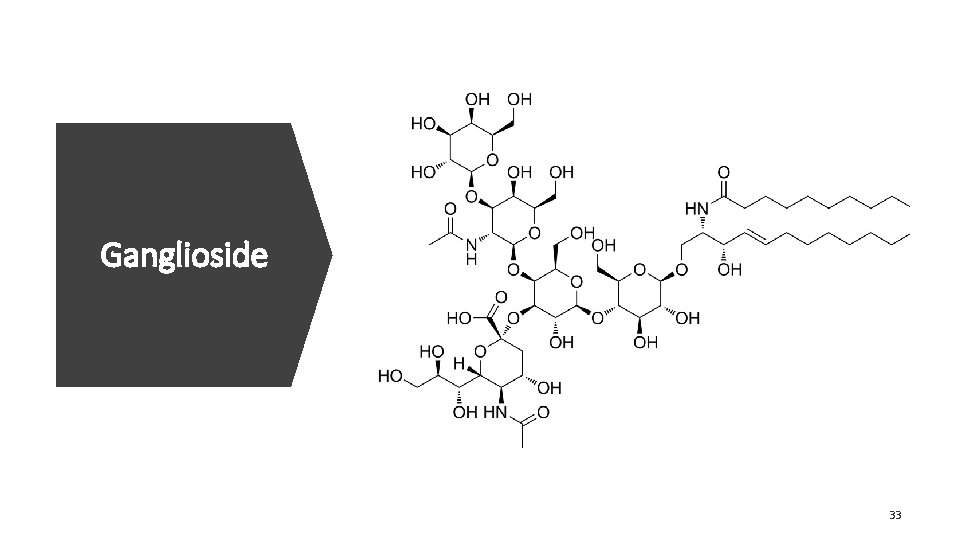
Ganglioside 33

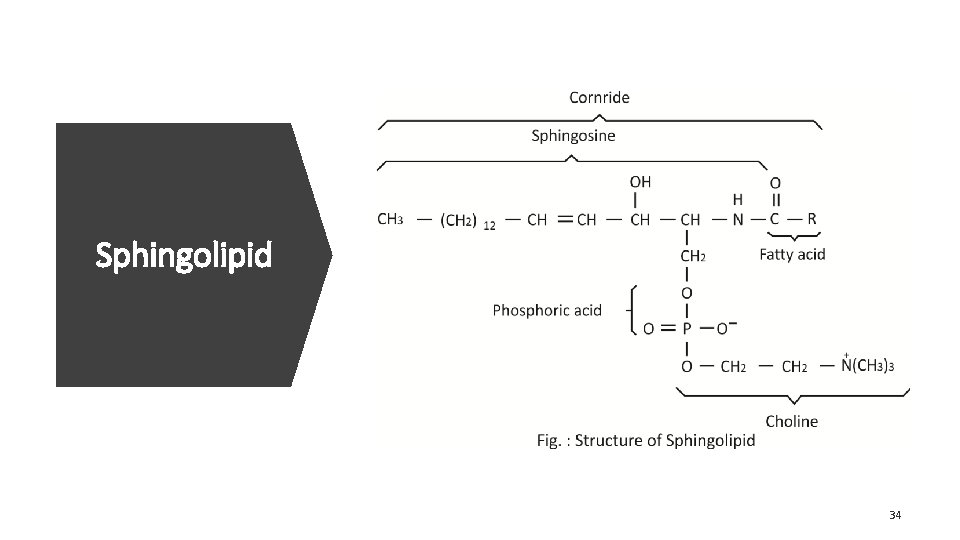
Sphingolipid 34

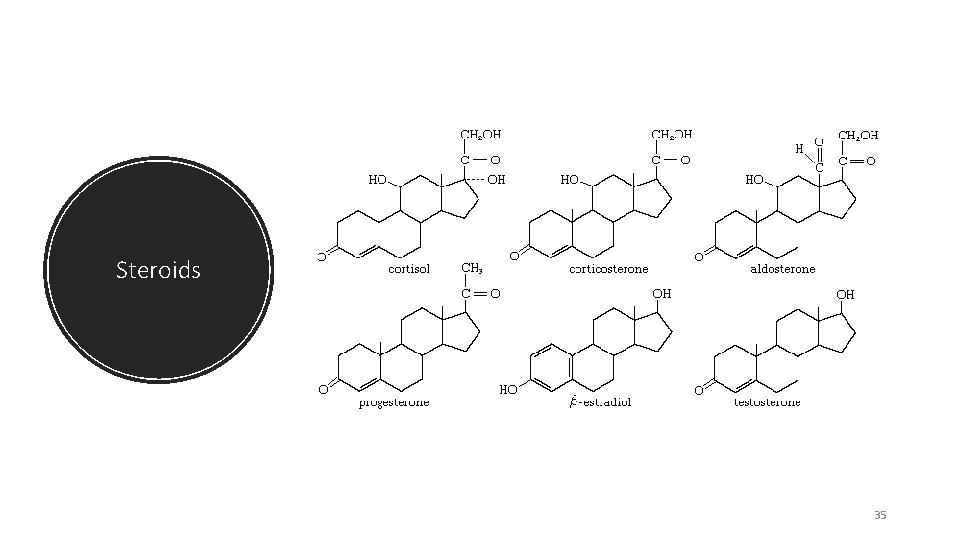
Steroids 35

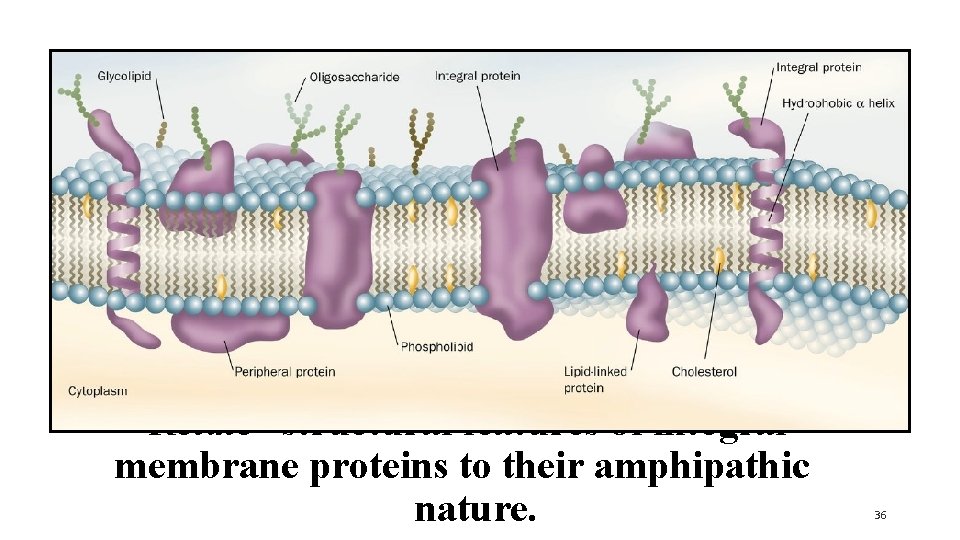
Relate structural features of integral membrane proteins to their amphipathic nature. 36

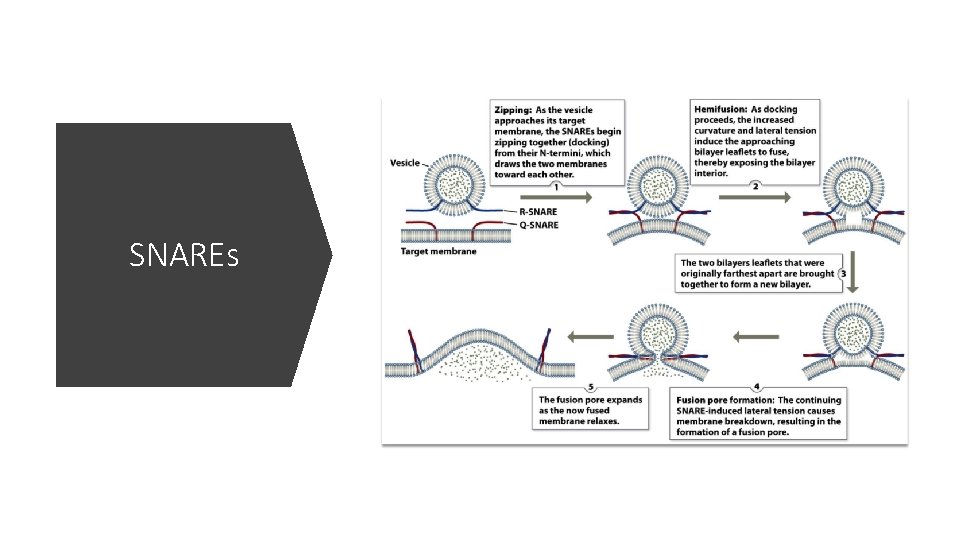
SNAREs

Thank you!