2017 AP Chemistry Exam Breakdown Question 1 The

2017 AP Chemistry Exam Breakdown
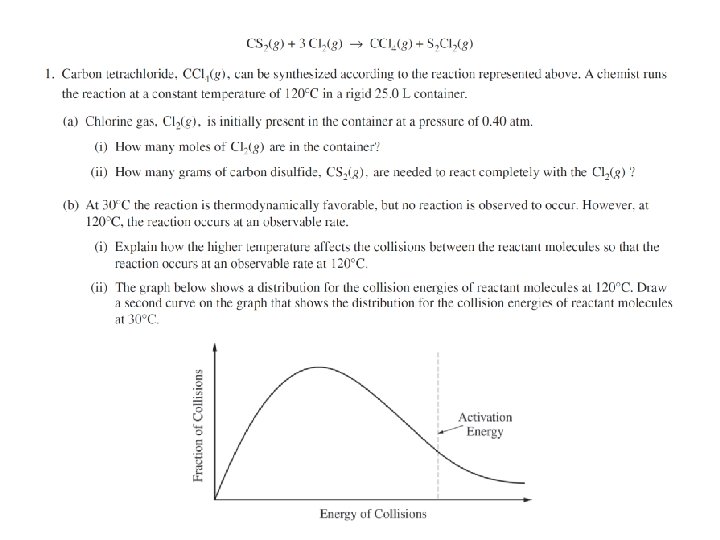


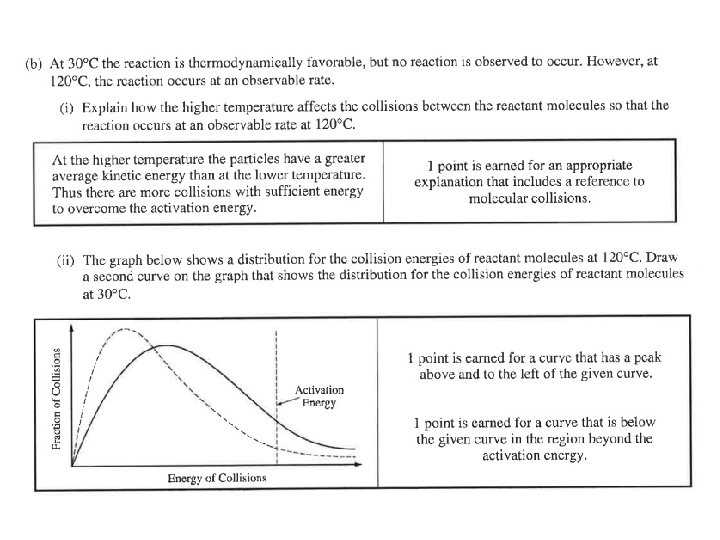



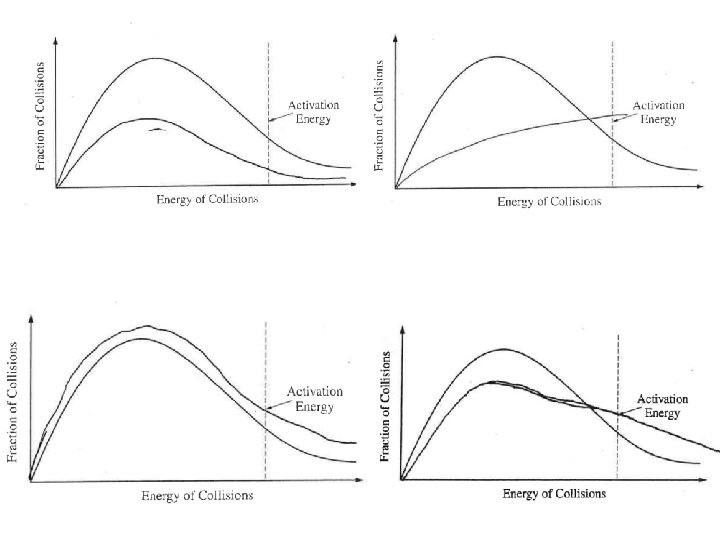
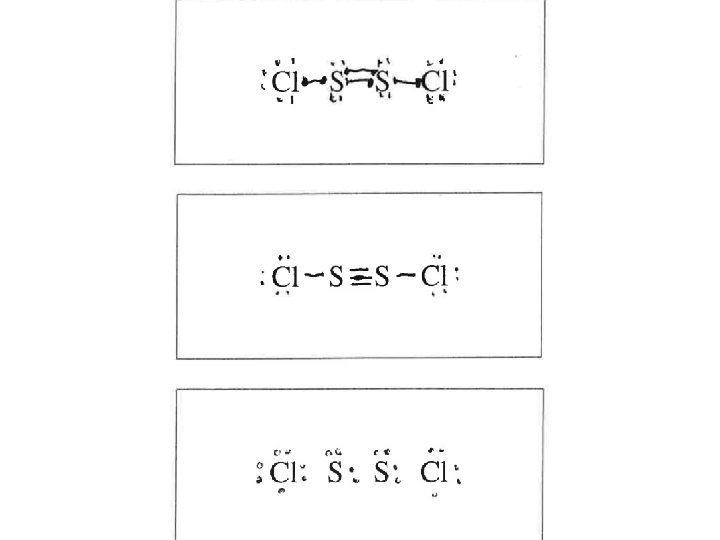
Question 1 The kids made the typical PV=n. RT mistakes. Typical stoichiometry mistakes. Students said heat is a catalyst or lowers the activation energy. Many 180 o and names of the shapes instead of angles. Lots of double bonds and expanded octets in the Lewis structure. Confusion between intramolecular vs intermolecular forces. (Also, H BONDING!)



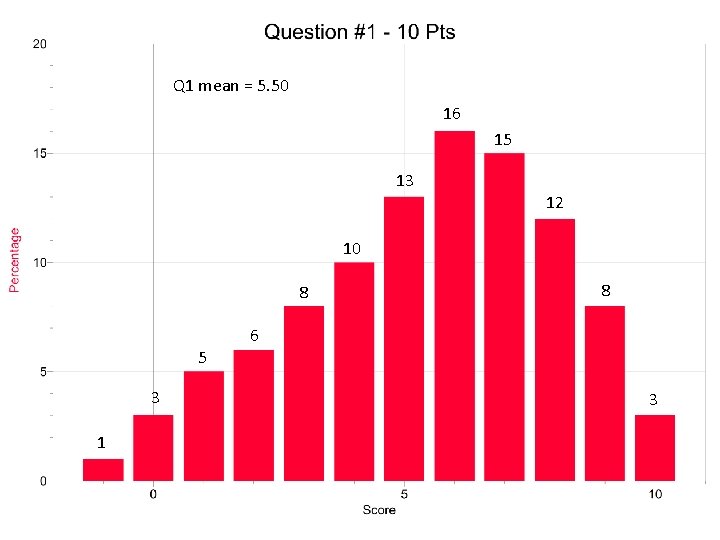
Q 1 mean = 5. 50 16 15 13 12 10 8 5 3 1 8 6 3

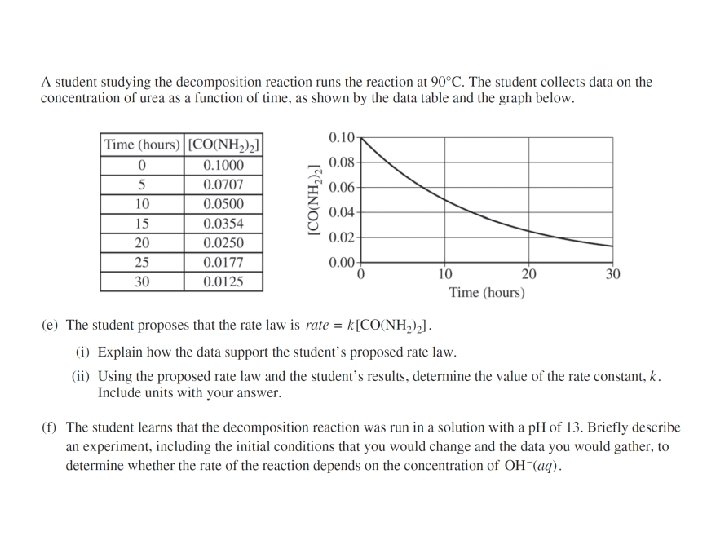

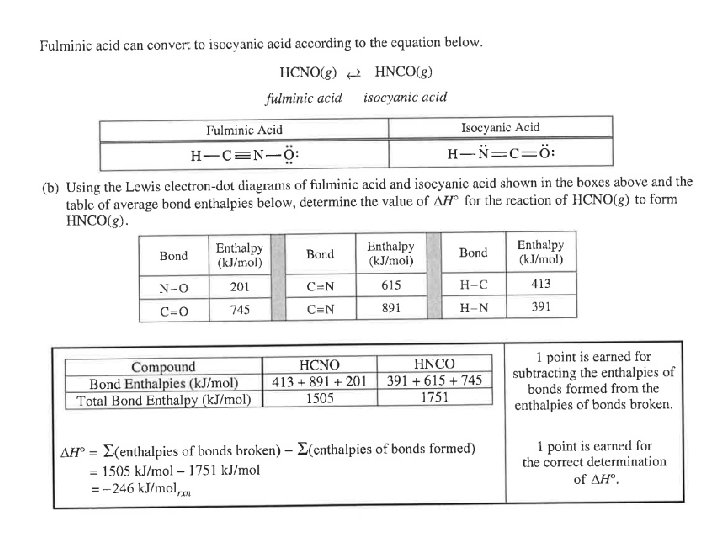



Question 2 Most students did not know how to do Formal Charges. Have the students show the TOTAL bond energy of the reactants and the TOTAL bond energy of products. Many students said because it is exothermic, the reaction is thermodynamically favorable. Students said that the products would be favored over the reactants, but that did not make a statement about the equilibrium constant. Students still can not recognize half-life patterns.



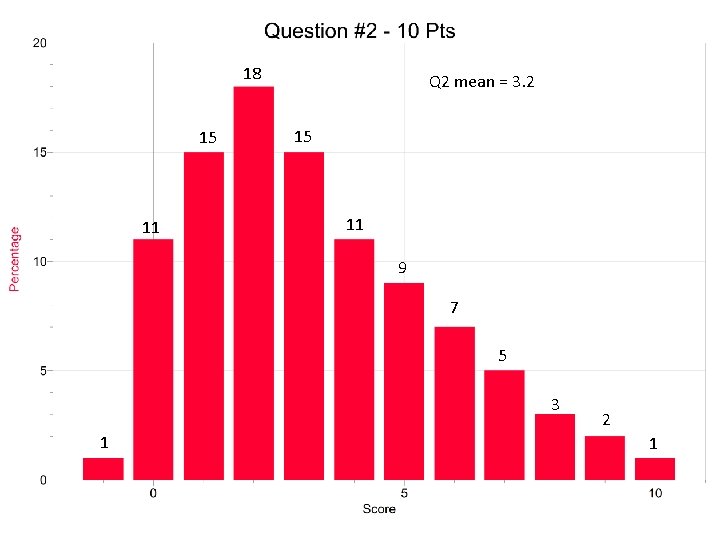
18 15 11 Q 2 mean = 3. 2 15 11 9 7 5 3 1 2 1







Question 3 Students don’t know what a buffer is. PV=n. RT. Math on calculating K. HNO 2 is not a strong acid. Water is not a great buffer! ( ) are not [ ]




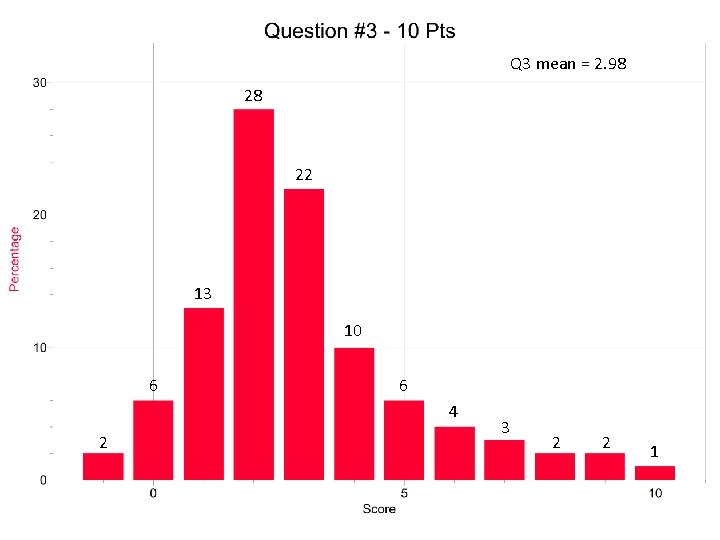
Q 3 mean = 2. 98 28 22 13 10 6 6 4 2 3 2 2 1
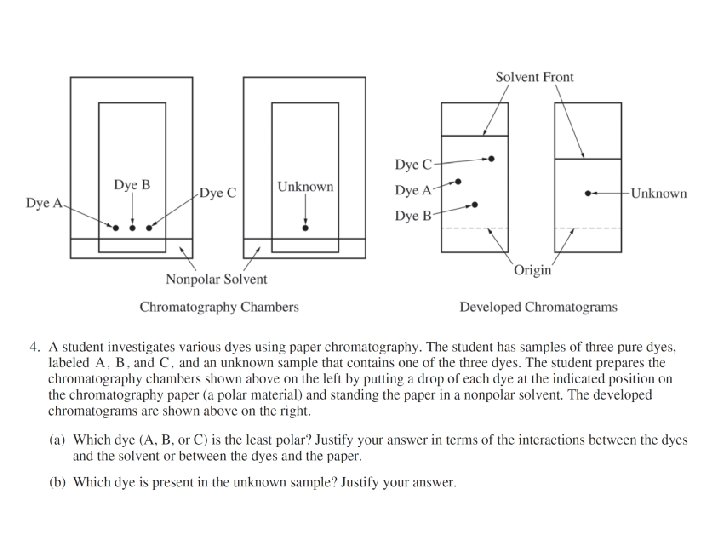


Question 4 Dye B since it is closest to the nonpolar solvent. Dye B because when they did the experiment the lowest one was the most nonpolar. Dye A is the unknown because it is closest to that of the unknown. Ratios and Retention factors were rarely discussed.

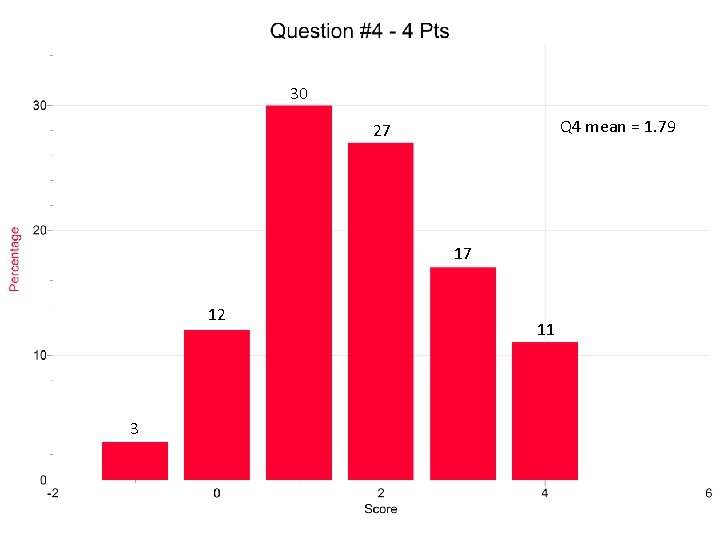
30 Q 4 mean = 1. 79 27 17 12 3 11

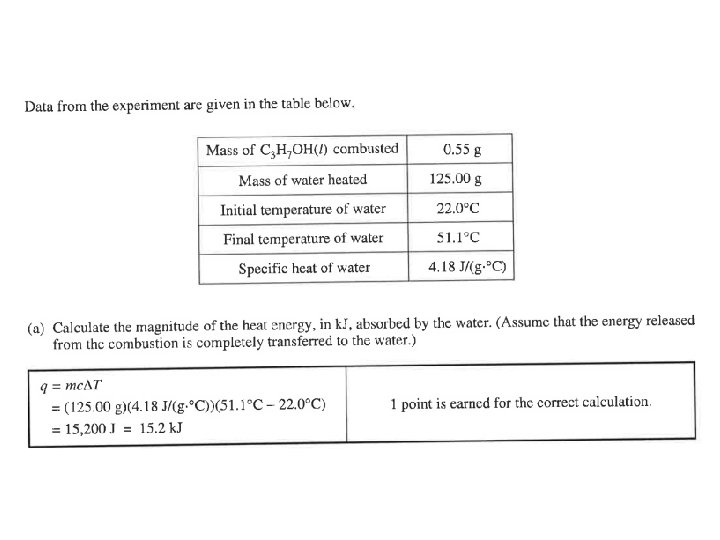


Question 5 Most common error was adding 0. 55 grams to the 125. 00 grams of water. Or……they just used the 0. 55 grams. They used q=mcΔT for part b. The combustion of H 2 O in part c. Adding water to combustion shifts rxn left.

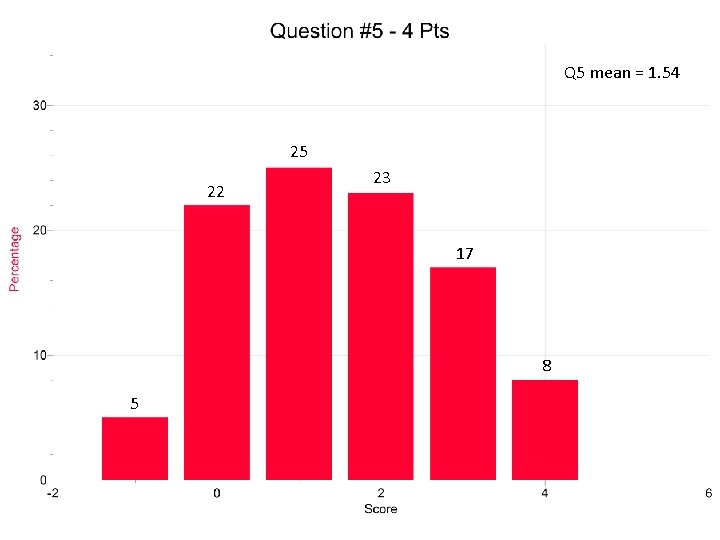
Q 5 mean = 1. 54 25 22 23 17 8 5




Solubility is not Ksp Question 6 MATH: squares and cubes PV=n. RT Molar volume of a gas at STP only applies to …. gases at STP Shielding…. ”you keep using that word…” Ions, atoms and molecules…. OH MY! The word IT!

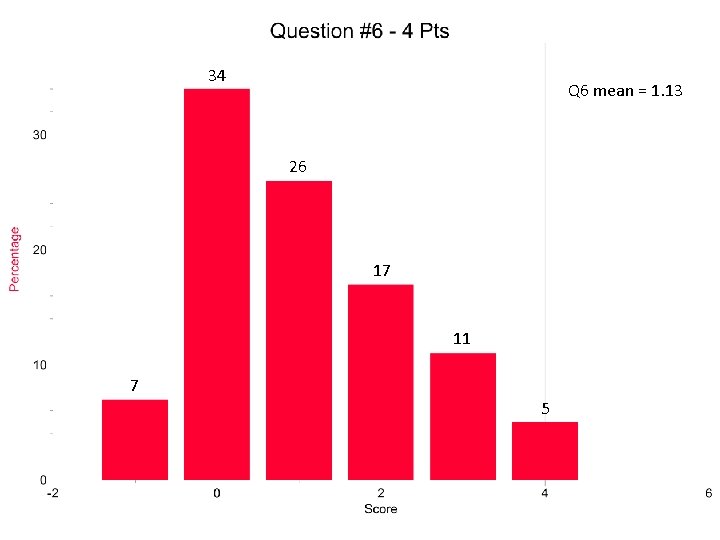
34 Q 6 mean = 1. 13 26 17 11 7 5




Question 7 Students didn’t know when to reverse the sign of the standard reduction potential. Eo is an intensive quantity. Don’t multiply by a stoichiometric factor. The answers in part a) needed to coordinate with the answers in part b).


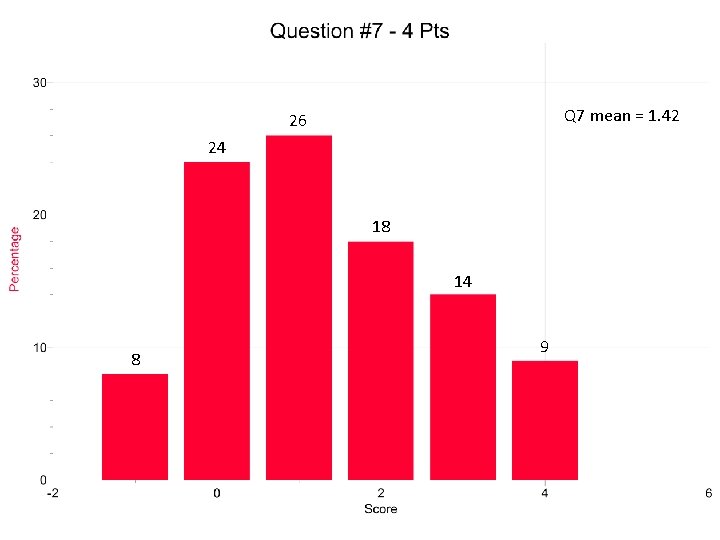
Q 7 mean = 1. 42 26 24 18 14 8 9
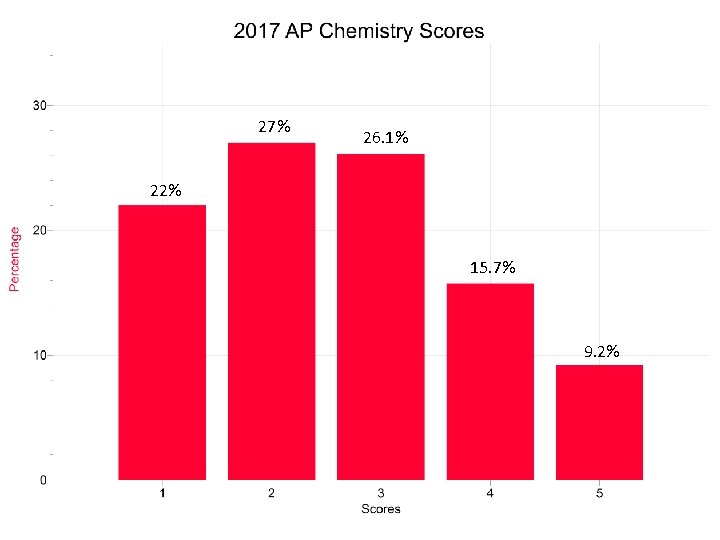
27% 26. 1% 22% 15. 7% 9. 2%

- Slides: 63