Final Exam Review pick up final exam review













































- Slides: 67

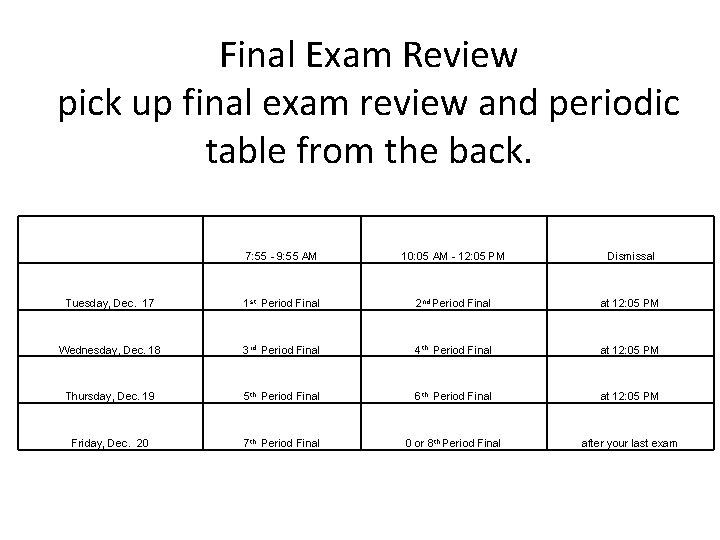
Final Exam Review pick up final exam review and periodic table from the back. 7: 55 - 9: 55 AM 10: 05 AM - 12: 05 PM Dismissal Tuesday, Dec. 17 1 st Period Final 2 nd Period Final at 12: 05 PM Wednesday, Dec. 18 3 rd Period Final 4 th Period Final at 12: 05 PM Thursday, Dec. 19 5 th Period Final 6 th Period Final at 12: 05 PM Friday, Dec. 20 7 th Period Final 0 or 8 th Period Final after your last exam

Announcements • If you have not taking any exam…you MUST do it by Thursday afternoon • I will be here Thursday until 4: 30 pm • If you would like to do a makeup on a final grade, it is due at the latest Monday during lunch. – For the paper exam you can do test corrections. – After Monday, I will not allow any more makeup exams.

Tips for studying for final exams… • DO NOT PROCRASTINATE • Start from what you just learned and work your way back. • Take study breaks! • Get enough sleep. • Teach someone else. If you can teach it, you understand it!!! • RELAX! It is JUST a final…. NO NEED TO STRESS!

Make sure you add Trigonal Pyramidal to note cards to study. It’s 3 -1 (107)

Rules for Naming Covalent Compounds 1. The “Most Metallic” Atom goes first 2. The amount of atoms goes first in the name followed by the name of the atom then the last atom has nonmetal root -ide 1. eg: N 2 S Di. Nitrogen monosulfide 3. If “O” is repeating, take out an O 1. eg: CO = Carbon Monoxide, NOT Carbon monooxide 4. Do not put Mono if the first atom only has one 1. Eg. CO is NOT monocarbon monoxide

Pre-Fixes to memorize # Prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona

We will start with # 47…

Ionic Vs. Covalent Ionic Between a metal and a nonmetal (left of staircase) (right of staircase) Covalent Between two nonmetals (right of staircase) Electrons are transferred from the metal to Electrons are shared between two nonmetals 1) metal + nonmetal root – ide 2) Transition metals and group 4 Copper (II) = Cu 2+ 3) Polyatomic Ions #nonmetal (most electronegative) + #nonmetalroot –ide

Needing Exams from! Lab Practical Elias Frederick Exam Mariella Mariel

Announcements • Thursday Afternoon is tentative – Due to illness

Diatomic Gases – Gasses found naturally as diatomic molecules • Hydrogen (H 2) Nitrogen (N 2) Oxygen (O 2) Fluorine (F 2) Chlorine (Cl 2) Iodine (I 2) Bromine (Br 2) • Have No Fear Of Ice Cold ROOTBeer

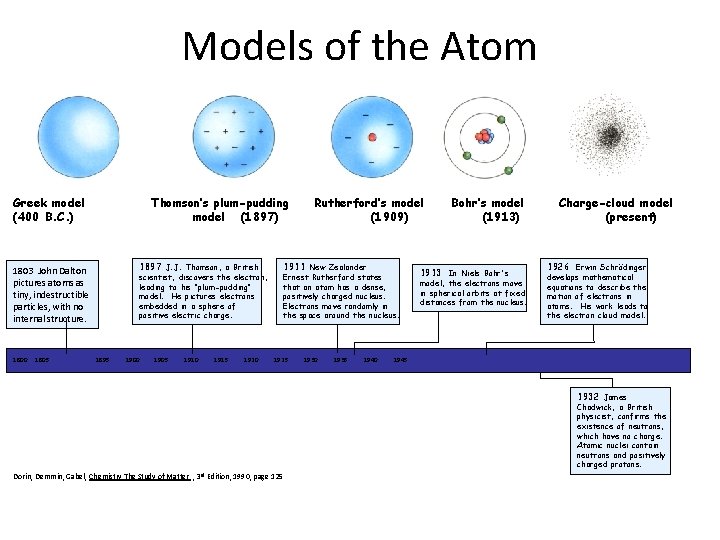
Models of the Atom Dalton’s Greek model (400 B. C. ) (1803) 1803 John Dalton pictures atoms as tiny, indestructible particles, with no internal structure. Thomson’s plum-pudding model (1897) 1897 J. J. Thomson, a British scientist, discovers the electron, leading to his "plum-pudding" model. He pictures electrons embedded in a sphere of positive electric charge. Rutherford’s model (1909) 1911 New Zealander Ernest Rutherford states that an atom has a dense, positively charged nucleus. Electrons move randomly in the space around the nucleus. Bohr’s model (1913) 1913 In Niels Bohr's model, the electrons move in spherical orbits at fixed distances from the nucleus. Charge-cloud model (present) 1926 Erwin Schrödinger develops mathematical equations to describe the motion of electrons in atoms. His work leads to the electron cloud model. 1800 1805 . . 1895 1900 1905 1910 1915 1920 1925 1930 1935 1940 1945 1932 James Chadwick, a British physicist, confirms the existence of neutrons, which have no charge. Atomic nuclei contain neutrons and positively charged protons. Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 125

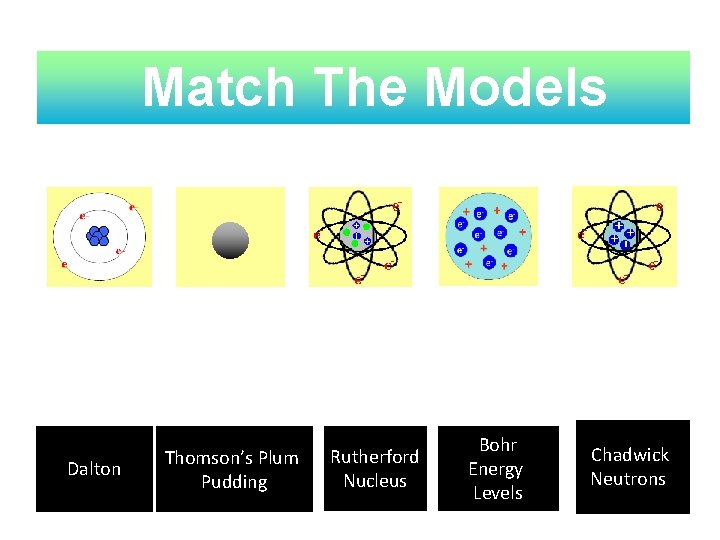
Match The Models Dalton Thomson’s Plum Pudding Rutherford Nucleus Bohr Energy Levels Chadwick, Neutrons

Sub-Particles in the Atom Location in the Atom Sub-Particle Mass Charge proton ~1 a. m. u. 1+ in nucleus neutron ~1 a. m. u. 0 in nucleus electron ~0 a. m. u. 1 - orbits nucleus atomic. mass. unit= a. m. u. ~ 1. 992 × 10− 27 kg or 1/12 the mass of C-12

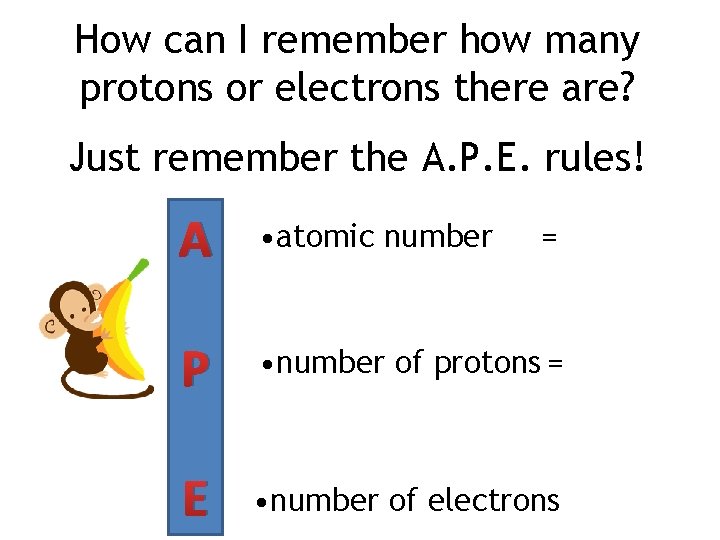
How can I remember how many protons or electrons there are? Just remember the A. P. E. rules! A • atomic number P • number of protons = E • number of electrons =

Now, how do I remember how many protons there are? Just remember the M. A. N. ! M • mass number - A • Atomic number = N • number of neutrons

Mass Number • mass number= protons + neutrons • always a whole number • NOT on the Periodic Table! e. Nucleus ee- Electrons e- e- e- + + Nucleus Carbon-12 Neutrons 6 Protons 6 Electrons 6 Neutron Proton

Writing Isotopes • Put the mass number after the name of the element • carbon- 12 • carbon -14 • uranium-235 – Or Mass # 12 Atomic # 6 California WEB C

Isotopes • Dalton was wrong. • Atoms of the same element can have different numbers of neutrons. • different mass numbers. • called isotopes.

Average Atomic Mass • massed average of all isotopes on the Periodic Table • round to 2 decimal places Avg. (mass)(%) + (mass)(%) Atomic = 100 Mass Avg. (mass)(decimal) + (mass)(decimal) Atomic = Mass

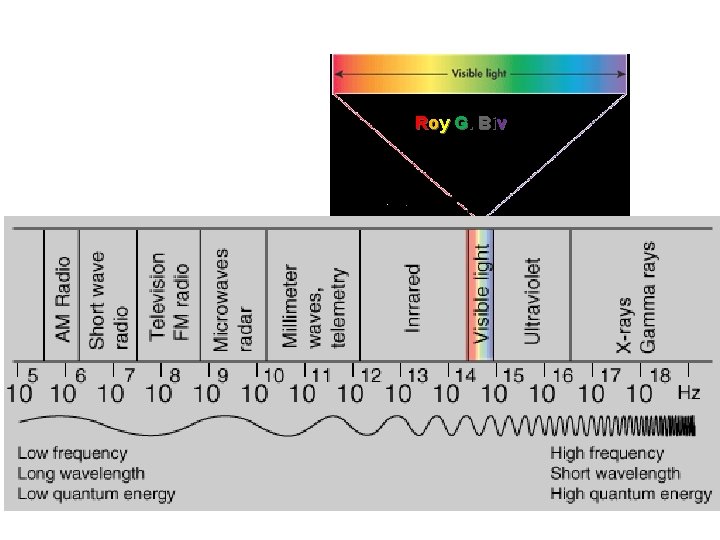
Roy G. Biv

Electrons Absorbing Energy Photon coming into atom collides with electron. Photons are energy. + The electron is “excited” to a higher energy level with is newly increased energy from absorbing the photon.

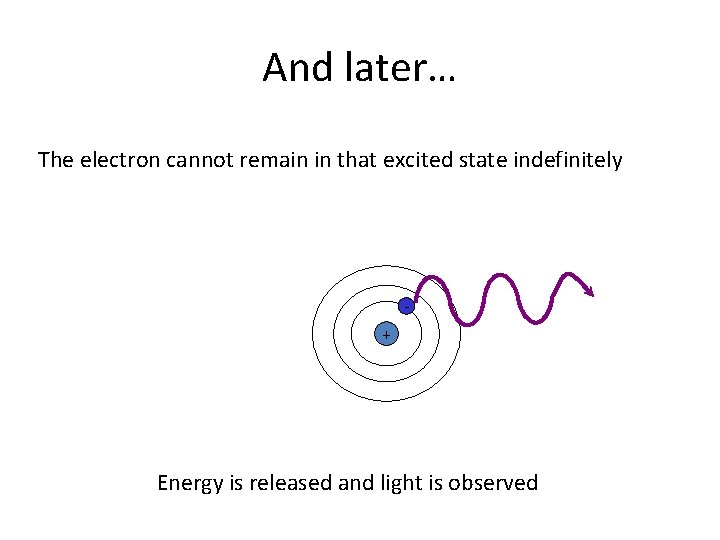
And later… The electron cannot remain in that excited state indefinitely + Energy is released and light is observed

Hydrogen Spectrum The black bars are the colors that a hydrogen atom absorbs. The other colors pass through the atom un-absorbed.

What does this have to do with electron arrangement in atoms? • When all electrons are in the lowest possible energy levels, an atom is said to be in its GROUND STATE. • When an atom absorbs energy so that its electrons are “boosted” to higher energy levels, the atom is said to be in an EXCITED STATE.

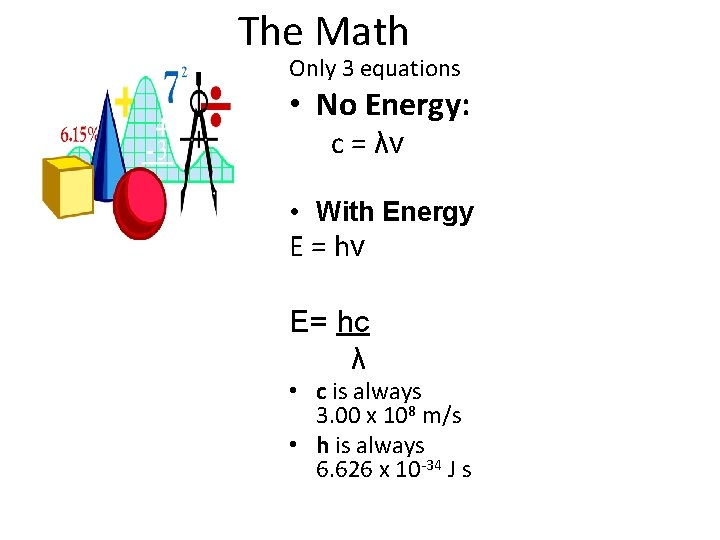
The Math Only 3 equations • No Energy: c = λv • With Energy E = hv E= hc λ • c is always 3. 00 x 108 m/s • h is always 6. 626 x 10 -34 J s

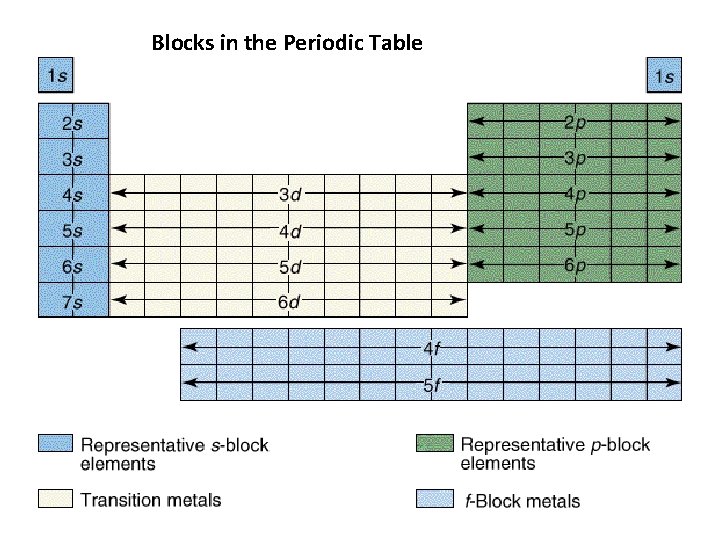
Blocks in the Periodic Table

Taking Measurements

Scientific Notation • For example, instead of writing 0. 000056, we write 5. 6 x 10 -9. • Always one number to the left of decimal…NO MORE NO LESS! • Exponent determines whether number is really big (positive) or really small (negative)

Significant Figures • Series of Questions: • Is the decimal absent or present? • Present – Start from the pacific ocean side (left). – Start counting from the first number that’s NOT 0 – Keep counting don’t stop • Atlantic – Start from the Atlantic ocean side (right). – Start counting from the first number that’s NOT 0 – Keep counting don’t stop

Density • D = m/V • Density = mass (g) / volume (m. L)

Metric Conversions

Physical and Chemical Changes • Physical – A physical change does not produce a new substance. Changes in state or phase (melting, freezing, vaporization, condensation, sublimation) are physical changes. Examples of physical changes include crushing a can, melting an ice cube, and breaking a bottle. • Chemical – A chemical change makes a substance that wasn't there before. There may be clues that a chemical reaction took place, such as light, heat, color change, gas production, odor, or sound. The starting and ending materials of a physical change are the same, even though they may look different.

Signs of Chemical Changes • Bubbles of gas appear • A precipitate forms. When mixing a pair of soluble reactants in solution, the sudden appearance of a solid that 'rains down' (precipitates) into the bottom of the container is a sign that a reaction has occurred. Sometimes the precipitate particles are too small to settle out; in this case, look for a cloudy solution. • A color change occurs • The temperature changes • Light is emitted. Sometimes energy is released by bond-forming reactions in the form of light. This occurs in most combustion reactions. Living things that glow in the dark- such as fireflies, funguses, and deep sea creatures- produce light without heat, using chemical reactions

Law of Conservation of Mass • In a reaction, the starting material must equal the ending material • Example: • What is the mass of the Na. Cl after the experiment ?

Elements, Compounds, Mixtures • Element: consists of only one kind of atom, cannot be broken down into a simpler type of matter by either physical or chemical means, and can exist as either atoms (e. g. argon) or molecules (e. g. , nitrogen). A molecule consists of two or more atoms of the same element, or different elements, that are chemically bound together. Note that the two nitrogen atoms which comprise a nitrogen molecule move as a unit. • Compound: consists of atoms of two or more different elements bound together, can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), has properties that are different from its component elements, and always contains the same ratio of its component atoms. • Mixture: consists of two or more different elements and/or compounds physically intermingled, can be separated into its components by physical means, and often retains many of the properties of its components. http: //www. chem. purdue. edu/gchelp/atoms/elements. html

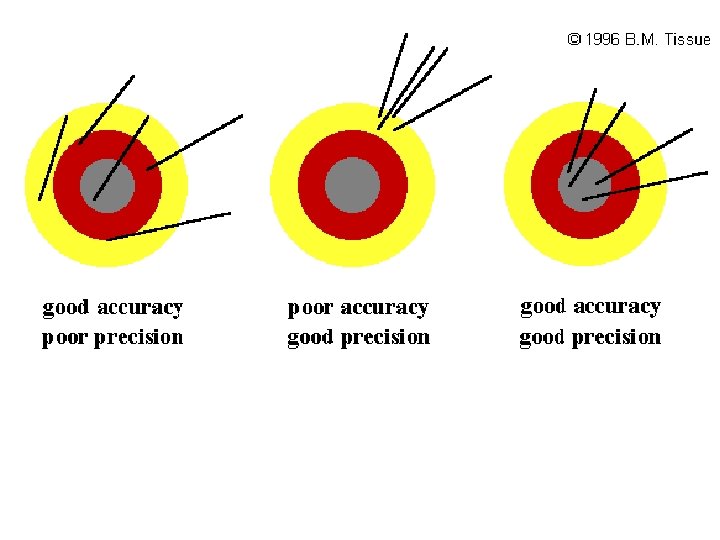
Accuracy and Precision • Accuracy refers to the agreement between experimental data and a known value. You can think of it in terms of a bullseye in which the target is hit close to the center, yet the marks in the target aren't necessarily close to each other. • Precision refers to how well experimental values agree with each other. If you hit a bullseye precisely, then you are able to hit the same spot on the target each time, even though that spot may be distant from the center.


Intensive vs. Extensive • Intensive – Depends on the substance – Same for all substances – Eg: aluminum has a density of 2. 7 g/m. L no matter the size, shape, or mass • Extensive – depends on the outside amount – Not the same – Eg: mass, volume…exc

Periodic Table • Periodicity: regular variations (or patterns) of properties with increasing atomic number. Both chemical and physical properties vary in a periodic (repeating) pattern. • Group: vertical column of elements (“family”) • Period: horizontal row of elements

Periodic Table and Periodicity Chemistry: The Periodic Table and Periodicity 1. Who first published the classification of the elements that is the basis of our periodic table today? DMITRI MENDELEEV 2. By what property did Mendeleev arrange the elements? ATOMIC MASS 3. By what property did Moseley suggest that the periodic table be arranged? ATOMIC NUMBER 4. What is the periodic law? THE PROPERTIES OF THE ELEMENTS REPEAT PERIODICALLY 5. What is a period? How many are there in the periodic table? A HORIZONTAL ROW IN THE PERIODIC TABLE; 7 6. What is a group (also called a family)? How many are there in the periodic table? A VERTICAL COLUMN IN THE PERIODIC TABLE; 18

The Periodic Law • Mendeleev understood the ‘Periodic Law’ which states: • When arranged by increasing atomic number, the chemical elements display a regular and repeating pattern of chemical and physical properties.

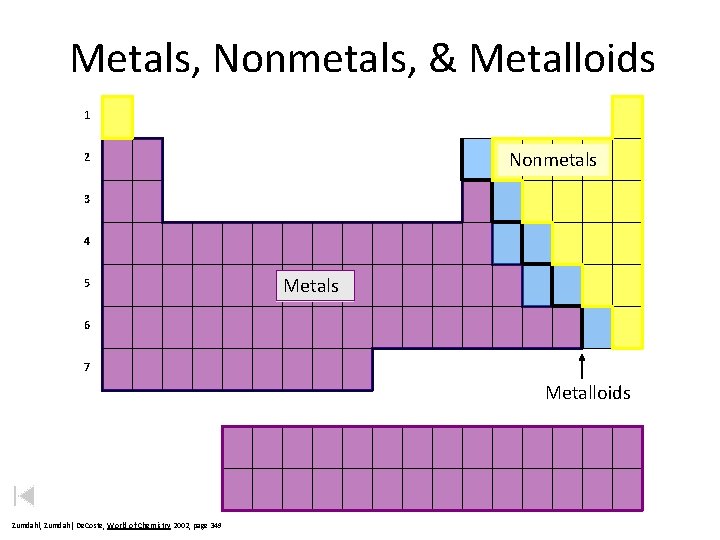
Metals, Nonmetals, & Metalloids 1 Nonmetals 2 3 4 5 Metals 6 7 Metalloids Zumdahl, De. Coste, World of Chemistry 2002, page 349

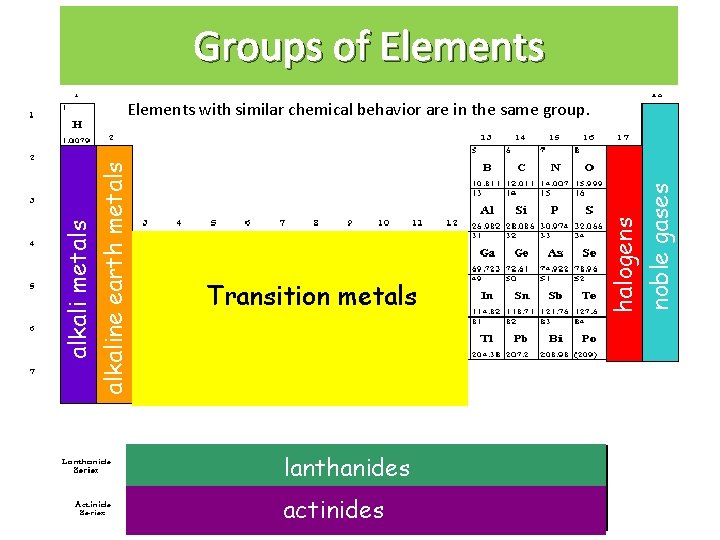
Groups of Elements Transition metals lanthanides actinides halogens noble gases alkali metals alkaline earth metals Elements with similar chemical behavior are in the same group.

Metals • Metals are lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • They are mostly solids at room temp. • What is one exception? Mercury (Hg)

Nonmetals • Nonmetals are the opposite. • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and Bromine is a liquid.

Metalloids • Metalloids, aka semi-metals are just that. • They have characteristics of both metals and nonmetals. • They are shiny but brittle. • And they are semiconductors. • What is our most important semiconductor? Silicon (Si)

Periodic trends Identifying the patterns

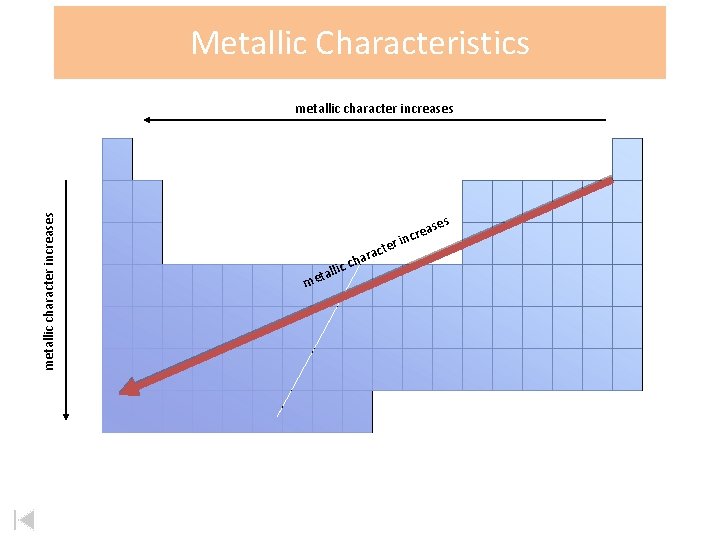
Metallic Characteristics metallic character increases s m llic eta ch ct ara er e eas r c n i

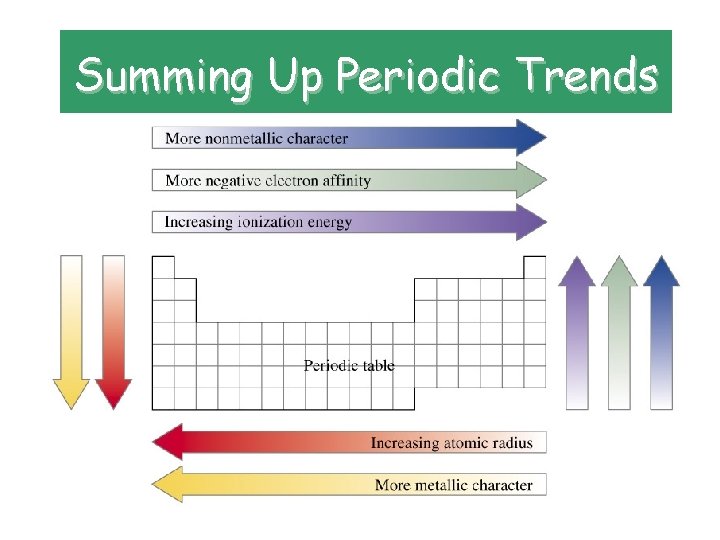
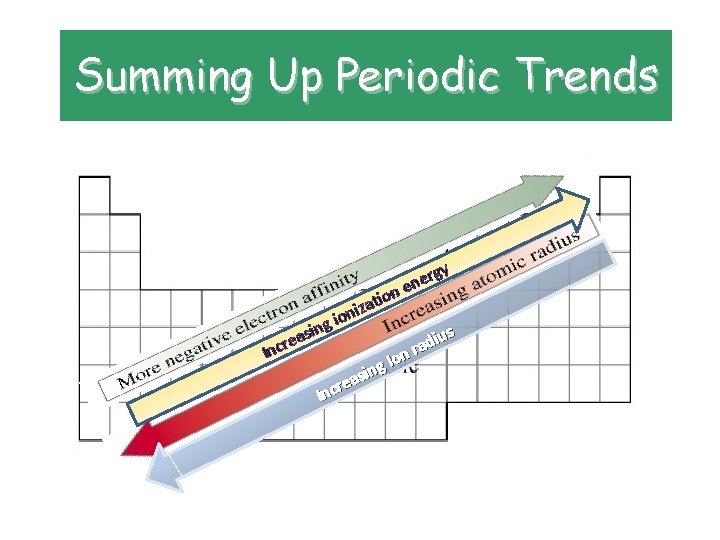
Periodic Trends • atomic radius. • Radius is the distance from the center of the nucleus to the “edge” of the electron cloud.

Atomic Radii

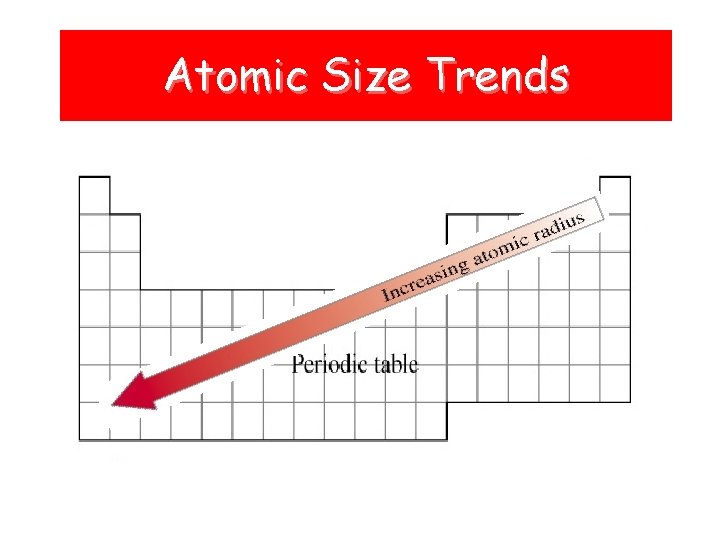
Atomic Radii • Trend: decreases across a period WHY? ? ? – As the # of protons in the nucleus increases, the positive charge increases and as a result, the “pull” on the electrons increases. • Trend: increases down a group WHY? ? ? – The atomic radius gets bigger because electrons are added to energy levels farther away from the nucleus.

Atomic Size Trends

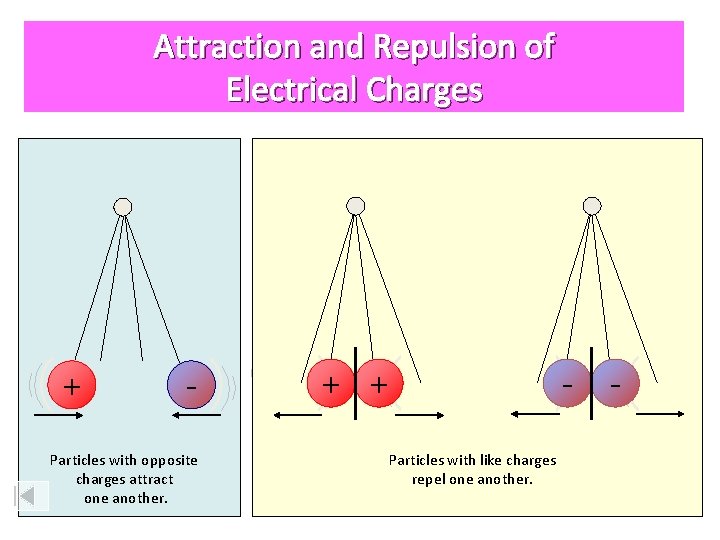
Attraction and Repulsion of Electrical Charges + - Particles with opposite charges attract one another. + + Particles with like charges repel one another. -

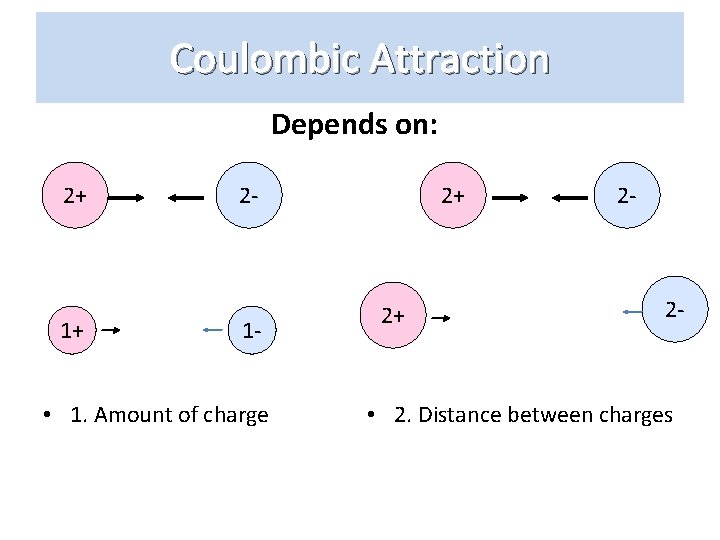
Coulombic Attraction Depends on: 2+ 1+ 2 - 1 - • 1. Amount of charge 2+ 2+ 2 - 2 - • 2. Distance between charges

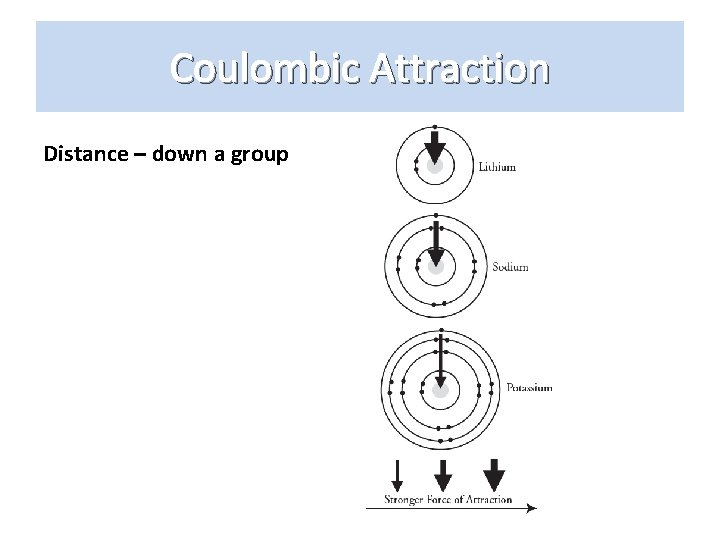
Coulombic Attraction Distance – down a group

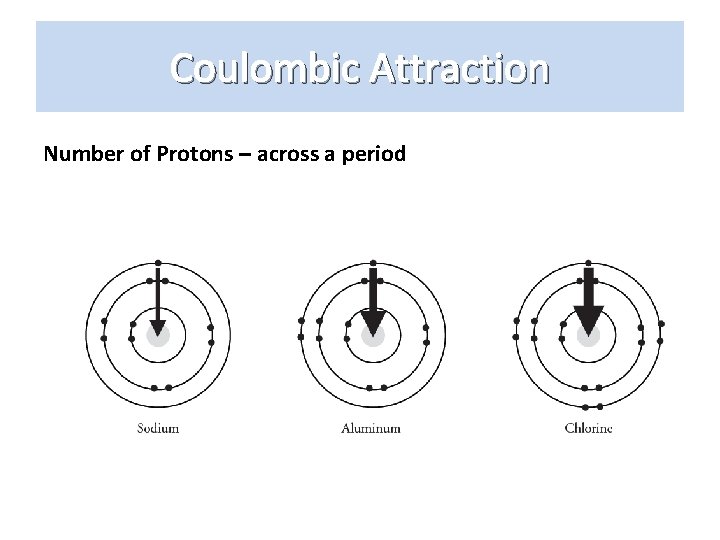
Coulombic Attraction Number of Protons – across a period

Coulombic Attraction So which one do we use? ? ? • Distance trumps number of protons. • Otherwise we would see an increase in the attractive force as you move from smaller to larger atoms down a column.

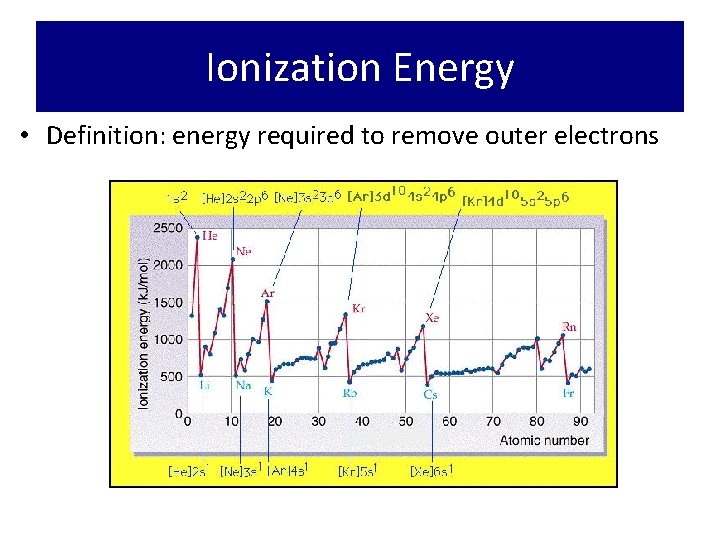
Ionization Energy • Definition: energy required to remove outer electrons

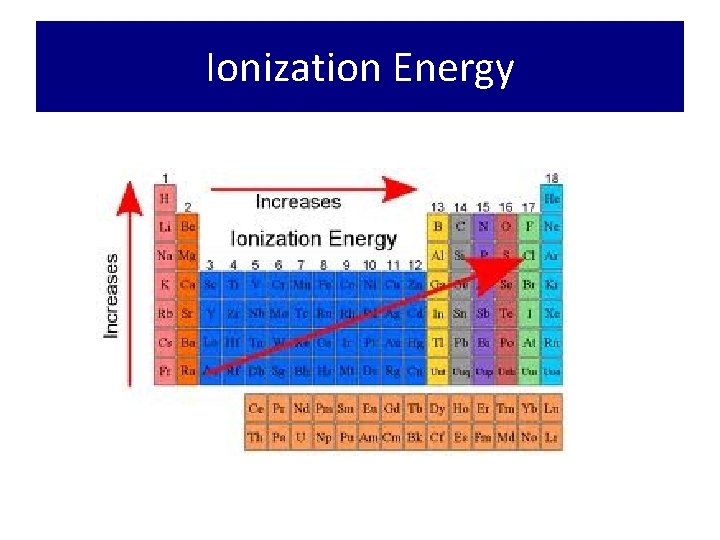
Ionization Energy

Ionization Energy • Trend: increases across a period • WHY? ? ? • All the atoms in the same period – Increasing nuclear charge • Trend: decreases down a group • WHY? ? ? – Electrons are further away from the positive “pull” of the nucleus and therefore easier to remove.

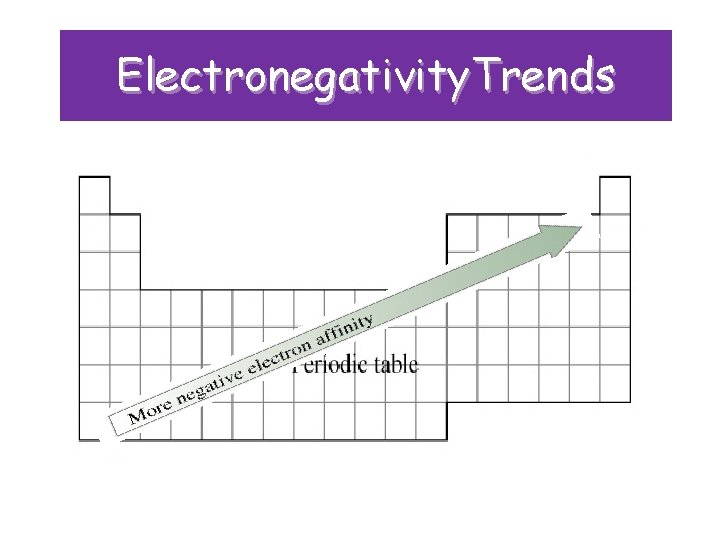
Electronegativity • Definition: the tendency of an atom to attract electrons to itself when chemically combined with another element

Electronegativity • Trend: increases across a period (noble gases excluded!) • WHY? ? ? – Nuclear charge is increasing, atomic radius is decreasing; attractive force that the nucleus can exert on another electron increases. • Trend: decreases down a group • WHY? ? ? – The electrons are farther away from the nucleus; decreased attraction, so decreased electronegativity

Electronegativity. Trends

Summing Up Periodic Trends

Summing Up Periodic Trends In as e r c in iza n o g i gy r e en n o ti us i d ra n o I g n i s rea c n I

Which has the largest atomic radius? 7. Br- Br 8. Al+ Al As an atom gains electrons, the atom gets bigger As an atom loses electrons, the atom gets smaller