Crude oil To state that crude oil is





- Slides: 15

Crude oil * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

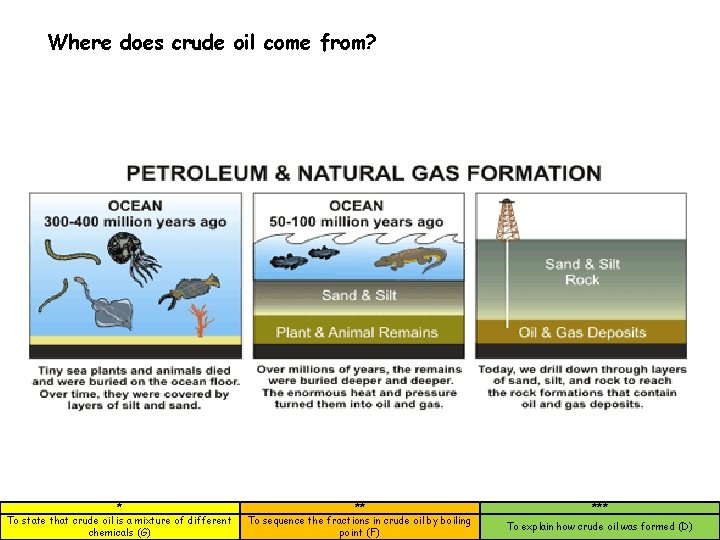
How crude oil was formed • Microscopic plants and animals die and fall to the sea bed • Layers of sand mud form on top • Pressure and high temperature cause oil to form • Oil obtained by drilling * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

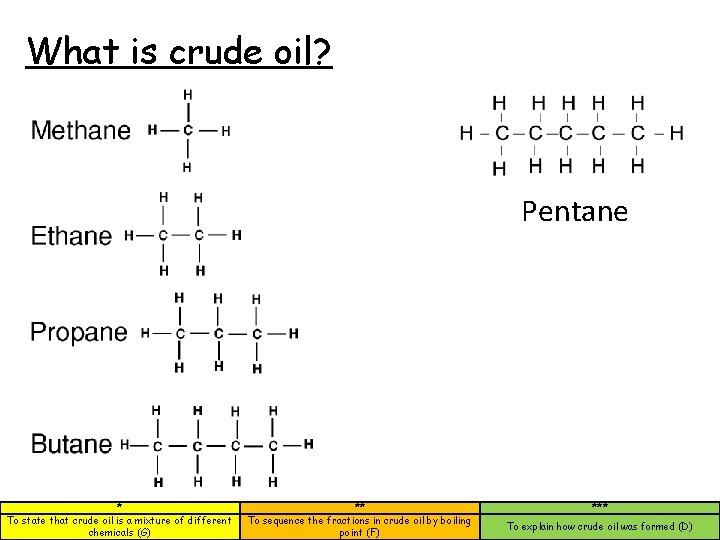
What is crude oil? Pentane * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

Where does crude oil come from? * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

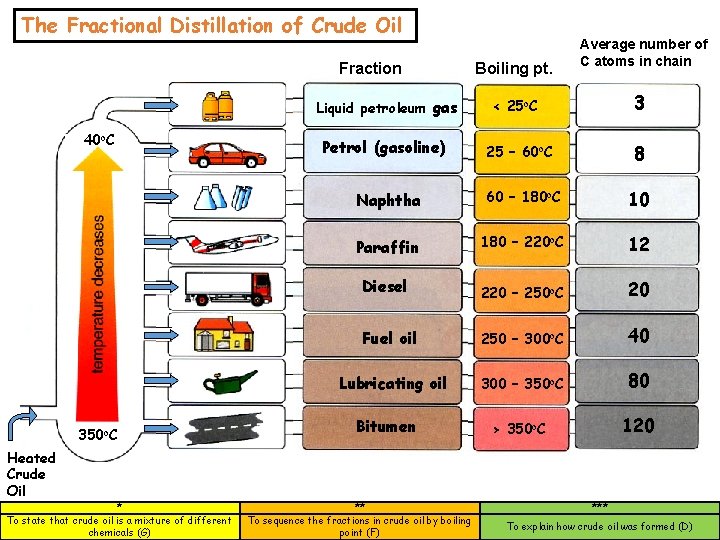
The Fractional Distillation of Crude Oil Fraction Liquid petroleum gas 40 o. C 350 o. C Heated Crude Oil * To state that crude oil is a mixture of different chemicals (G) Petrol (gasoline) Boiling pt. Average number of C atoms in chain 3 < 25 o. C 8 25 – 60 o. C Naphtha 60 – 180 o. C 10 Paraffin 180 – 220 o. C 12 Diesel 220 – 250 o. C 20 Fuel oil 250 – 300 o. C 40 Lubricating oil 300 – 350 o. C 80 > 350 o. C 120 Bitumen ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

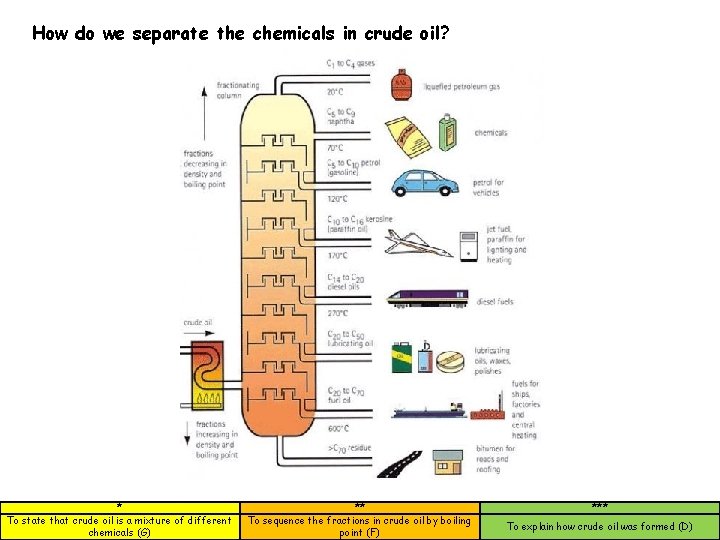
How do we separate the chemicals in crude oil? * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

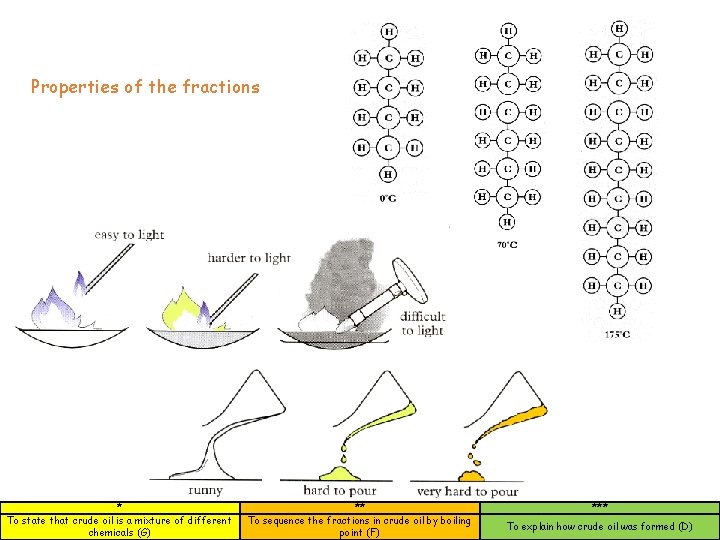
Properties of the fractions * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

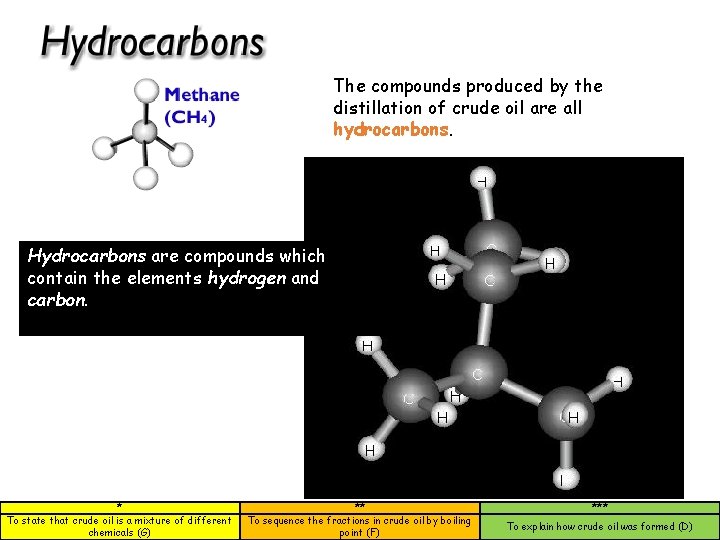
The compounds produced by the distillation of crude oil are all hydrocarbons. Hydrocarbons are compounds which contain the elements hydrogen and carbon. * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

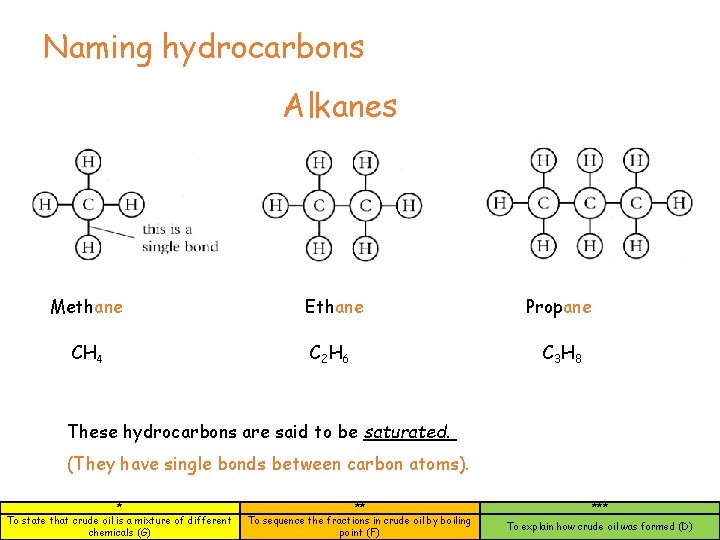
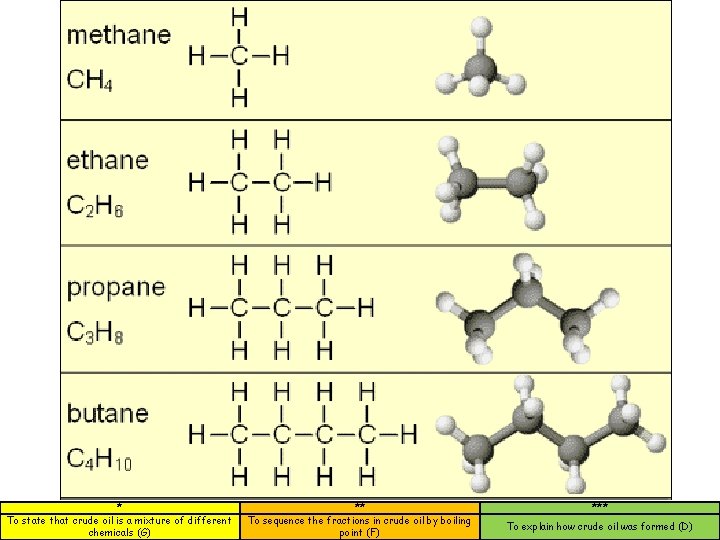
Naming hydrocarbons Alkanes Methane Ethane Propane CH 4 C 2 H 6 C 3 H 8 These hydrocarbons are said to be saturated. (They have single bonds between carbon atoms). * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

* To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

Fuels Most alkanes are used as fuels to produce useful forms of energy. When completely burned alkanes form carbon dioxide and water. * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

Equations for combustion CH 4 + methane 2 O 2 CO 2 oxygen + carbon dioxide C 3 H 8 + O 25 CO 2 3+ H 2 O C 2 H 4 + O 23 CO 2 2+ H 2 O * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) 2 H 2 O water 4 2 *** To explain how crude oil was formed (D)

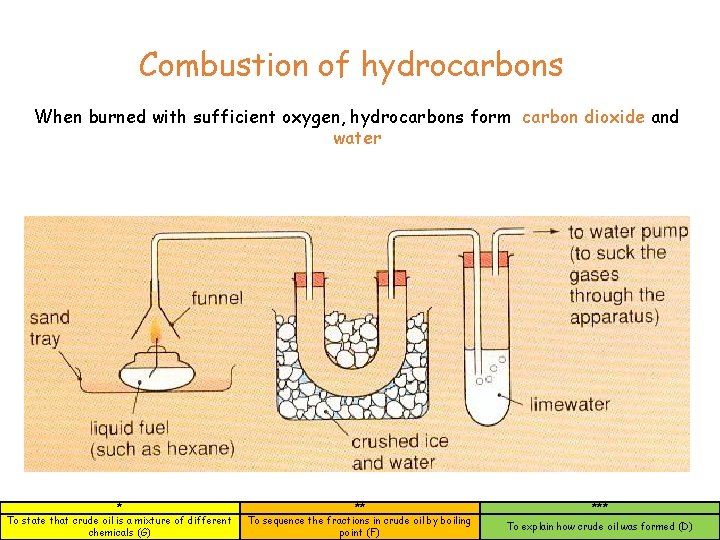
Combustion of hydrocarbons When burned with sufficient oxygen, hydrocarbons form carbon dioxide and water * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

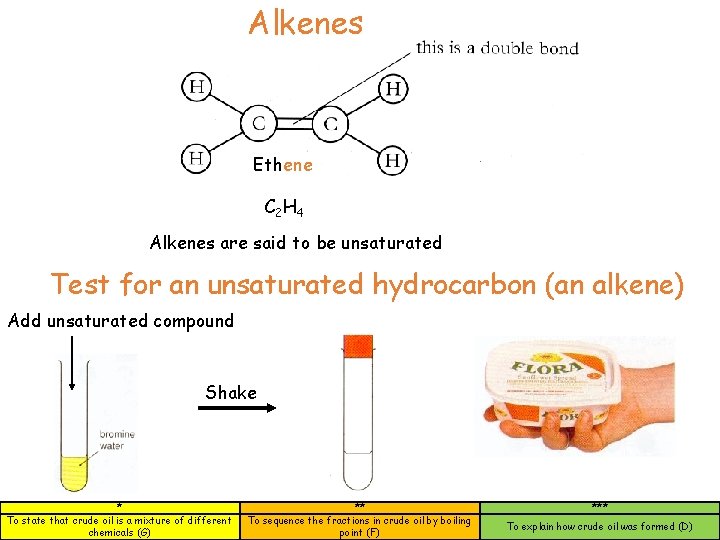
Alkenes Ethene C 2 H 4 Alkenes are said to be unsaturated Test for an unsaturated hydrocarbon (an alkene) Add unsaturated compound Shake * To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)

* To state that crude oil is a mixture of different chemicals (G) ** To sequence the fractions in crude oil by boiling point (F) *** To explain how crude oil was formed (D)