Chem 31 1018 Lecture Announcements Quiz 3 Today
















- Slides: 18

Chem. 31 – 10/18 Lecture

Announcements • Quiz 3 Today • Today’s Lecture – Chapter 6 • Acids, Bases and Salts – Chapter 7 – Titrations • Overview and Definitions

Acids, Bases and Salts Definitions of Acids and Bases - Lewis Acids/Bases (defined before, most general category) - Brønsted-Lowry Acids/Bases: acid = proton donor base = proton acceptor (must have free electron pair so also is a Lewis base) - definitions are relative

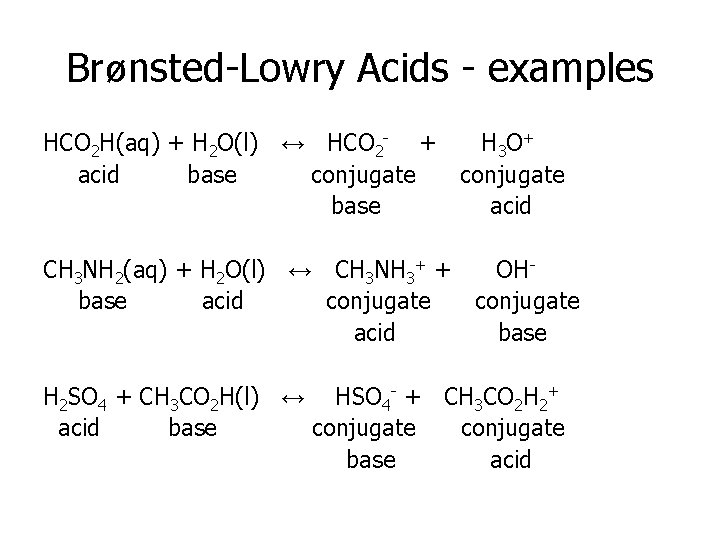
Brønsted-Lowry Acids - examples HCO 2 H(aq) + H 2 O(l) ↔ HCO 2 - + H 3 O+ acid base conjugate base acid CH 3 NH 2(aq) + H 2 O(l) ↔ CH 3 NH 3+ + base acid conjugate acid OHconjugate base H 2 SO 4 + CH 3 CO 2 H(l) ↔ HSO 4 - + CH 3 CO 2 H 2+ acid base conjugate base acid

Brønsted-Lowry Acids Note: for most acids, the reaction with water is simplified: Example: HNO 2 (nitrous acid) HNO 2 ↔ H+ + NO 2 -

Autoprotolysis and the p. H Scale Autoprotolysis refers to proton transfer in protic solvents like water: H 2 O(l) ↔ H+ + OHK = Kw = [H+][OH-] = 1. 0 x 10 -14 (T = 25°C) In pure water [H+] = [OH-] = Kw 0. 5 = 1. 0 x 10 -7 M p. H = -log[H+] = 7. 0 Acidic is p. H < 7; basic is p. H > 7

Strong Acids • Strong acids completely dissociate in water (except at very high concentrations) – HX(aq) → H+ + X- (no HX(aq) exists) • Ka > 1 • Major strong acids: HCl, HNO 3, H 2 SO 4 • Note: – For H 2 SO 4, 1 st dissociation is that of a strong acid, but 2 nd dissociation is that of a weak acid (Ka ~ 0. 01)

Weak Acids • • Partially dissociate in water Most have H that can dissociate HX(aq) ↔ H+ + X- (HX(aq) exists) Example: HNO 2 ↔ H+ + NO 2 Degree of dissociation given by Ka value Ka = [H+][NO 2 -]/[HNO 2] Metal cations can be acids through the reaction: Mn+ + H 2 O(l) ↔ MOH(n-1)+ + H+ (although for +1 and some +2 metals the above reactions favor reactants so strongly the metals can be considered “neutral”)

Ionic Compounds in Water • First step should be dissociation to respective ions: example: Na. Cl(s) → Na+ + Cl • In subsequent steps, determine how anion/cation react: - anions usually only react as bases - cations may react as acids - see if ions are recognizable conjugate acids or bases - polyprotic acids are somewhat different

Ionic Compounds in Water Conjugate bases of weak acids are basic. NO 2 - + H 2 O(l) ↔ HNO 2 (aq) + OH- Conjugate bases of weaker weak acids are stronger bases. Kb = Kw/Ka CN- is a stronger base than NO 2 - because Ka(HCN) = 6. 2 x 10 -10 and Ka(HNO 2) = 7. 1 x 10 -3

Acidity of Ionic Compounds Determine if the ionic compounds are acidic or basic in the following examples: 1. Na. Cl 2. NH 4 Cl 3. Na. CH 3 CO 2 4. Fe(NO 3)3 5. NH 4 CN

Polyprotic Acids • Release more than 1 H+ per molecule Examples: H 2 SO 4, H 3 PO 4, H 2 C 2 O 4 H 3 PO 4 has 3 Ka values (Ka 1, Ka 2, Ka 3) for 3 reactions losing H+: 1) H 3 PO 4 ↔ H 2 PO 4 - + H+ Ka 1 2) H 2 PO 4 - ↔ HPO 42 - + H+ Ka 2 3) HPO 42 - ↔ PO 43 - + H+ Ka 3

Chapter 7 - Titrations Introduction – Overview A. Chapter 7 covers general titrations (quantitation, practical aspects, types of titrations, shape of precipitation titration curve – not covering calculations due to time) B. Chapter 11 covers titration curves for acidbase titrations - covered later C. Other Chapters (12, 16) cover other types of titrations – not covered

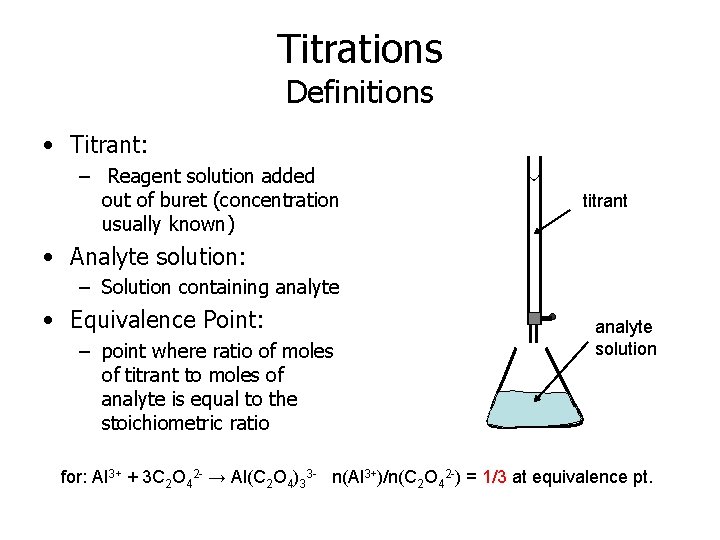
Titrations Definitions • Titrant: – Reagent solution added out of buret (concentration usually known) titrant • Analyte solution: – Solution containing analyte • Equivalence Point: – point where ratio of moles of titrant to moles of analyte is equal to the stoichiometric ratio analyte solution for: Al 3+ + 3 C 2 O 42 - → Al(C 2 O 4)33 - n(Al 3+)/n(C 2 O 42 -) = 1/3 at equivalence pt.

Titrations Practical Requirements • The equilibrium constant must be large – Size of K value depends on desired precision and concentration of analyte – Typically K ~ 106 is marginal, K > 1010 is better • The reaction must be fast • It must be possible to “observe” the equivalence point – observed equivalence point = end point

Titrations Detection of Endpoints • An endpoint is defined as the point in the titration when the equivalence point is observed • Ways to detect endpoints: – Use of colored reactants example: Mn. O 4 - + H 2 C 2 O 4 (aq) → Mn 2+ + CO 2 (g) PINK Clear – Use of indicators An indicator changes color in response to the change in a reactant’s concentration – Use of simple instruments Must respond quickly, but typical equipment is low cost

Titrations Detection of Endpoints • Simple instruments – electrodes (typically respond to log of ion concentrations) – spectroscopic measurements (measurement of absorption of light) – Can improve titration precision vs. using indicators • Titration Error = Difference between end point and equivalence point = systematic error • Note: It is possible to have large errors or uncertainties in detection of reagent conc. by various methods without having great titration errors to meter

Titrations Other Definitions • Standardization vs. Analyte Titrations – To accurately determine an analyte’s concentration, the titrant concentration must be well known – This can be done by preparing a primary standard (high purity standard) – Alternatively, the titrant concentration can be determined in a standardization titration (e. g. vs. a known standard) • Rationale: – many solutions can not be prepared accurately from available standards • Example: – determination of [H 2 O 2] by titration with Mn. O 4– neither compound is very stable so no primary standard – instead, [Mn. O 4 -] determined by titration with H 2 C 2 O 4 in standardization titration – then, H 2 O 2 titrated using standardized Mn. O 4 -