CHEMSHEETS STRUCTURE TYPES Chemsheets AS 1024 07 June2015









- Slides: 38

CHEMSHEETS STRUCTURE TYPES Chemsheets AS 1024 07 -June-2015

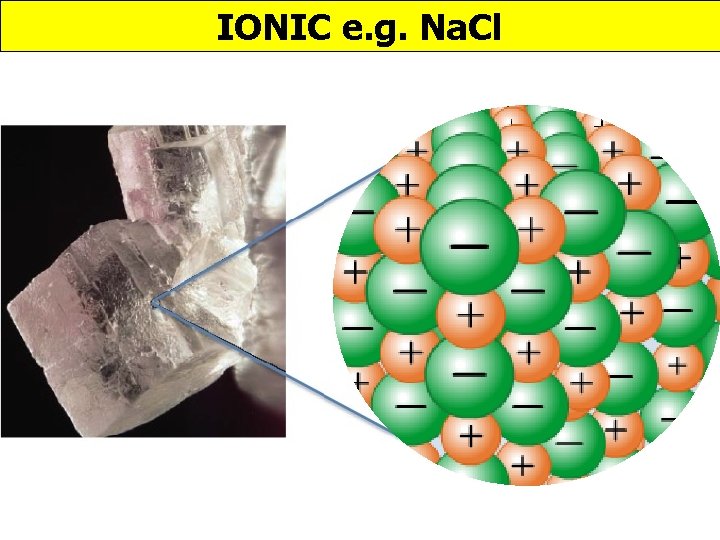
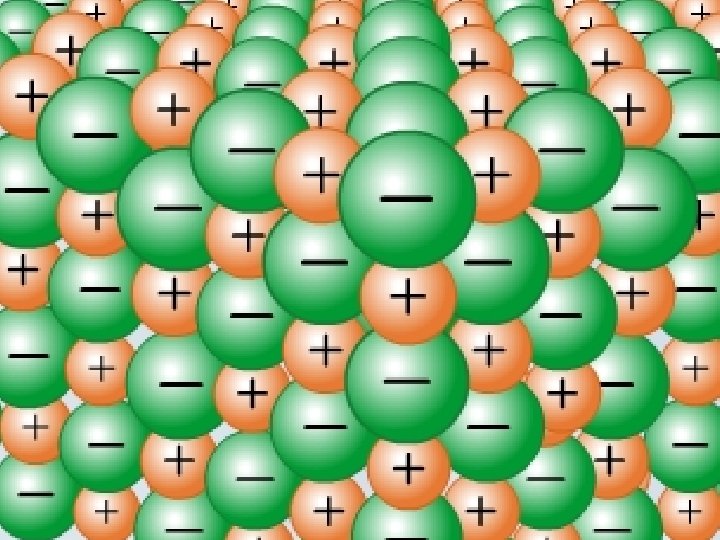
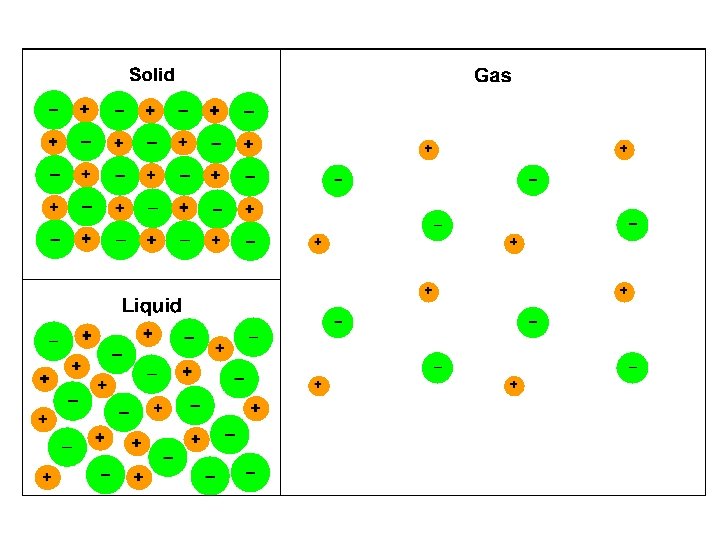
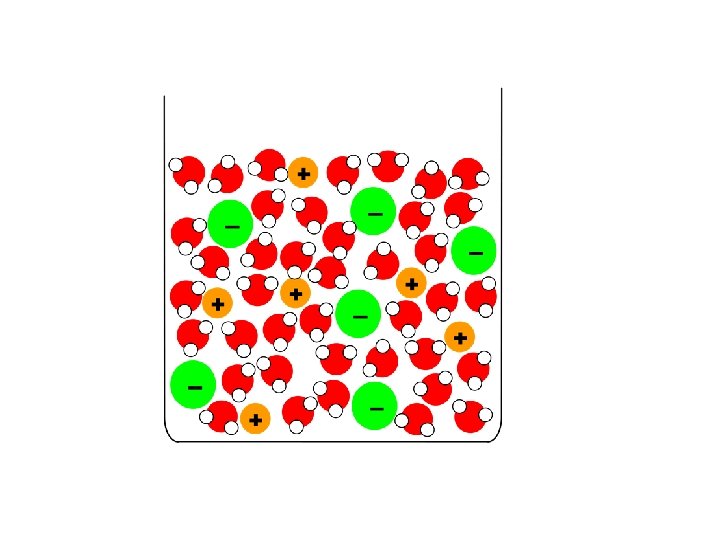
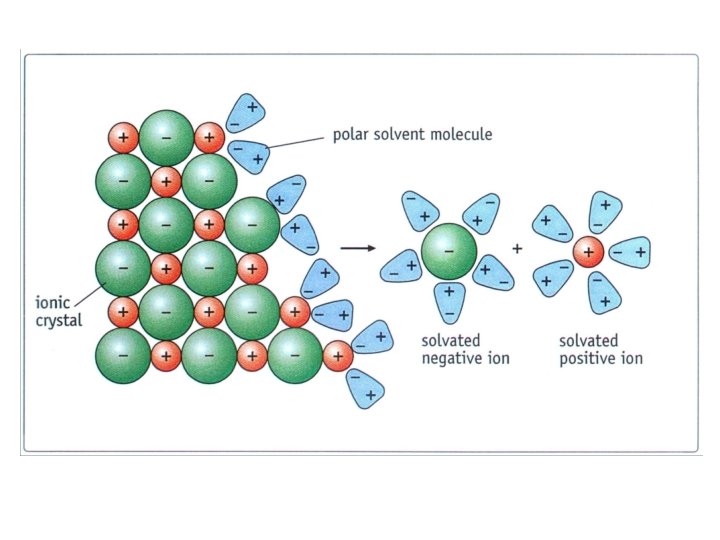
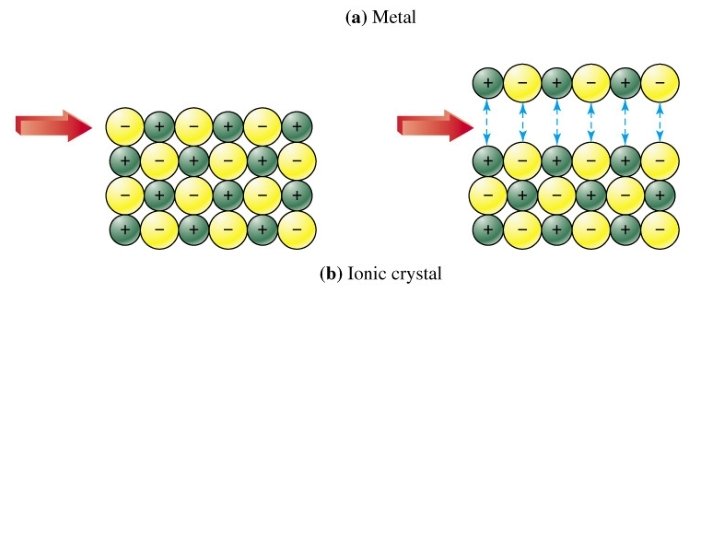
IONIC e. g. Na. Cl






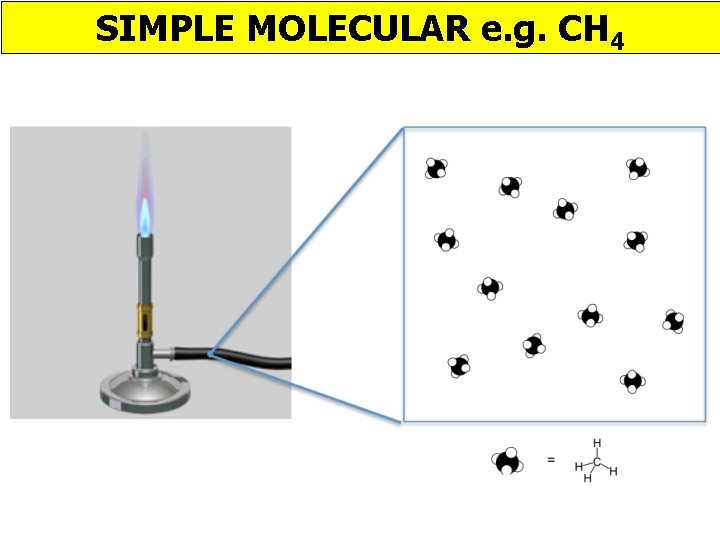
SIMPLE MOLECULAR e. g. CH 4

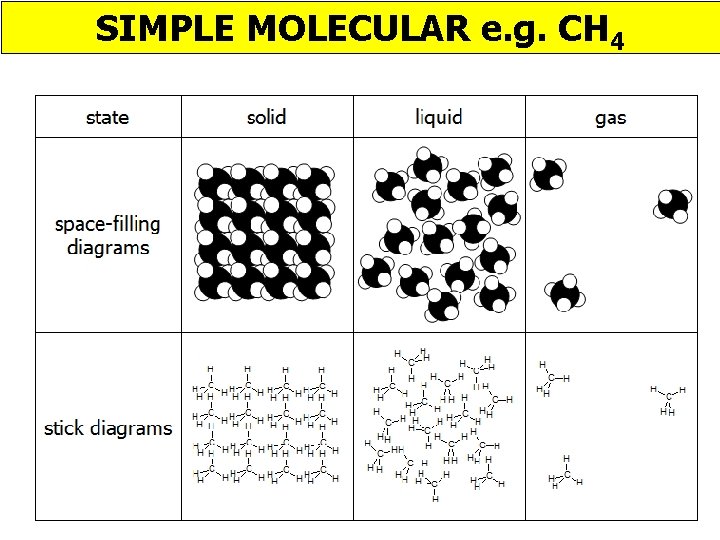
SIMPLE MOLECULAR e. g. CH 4


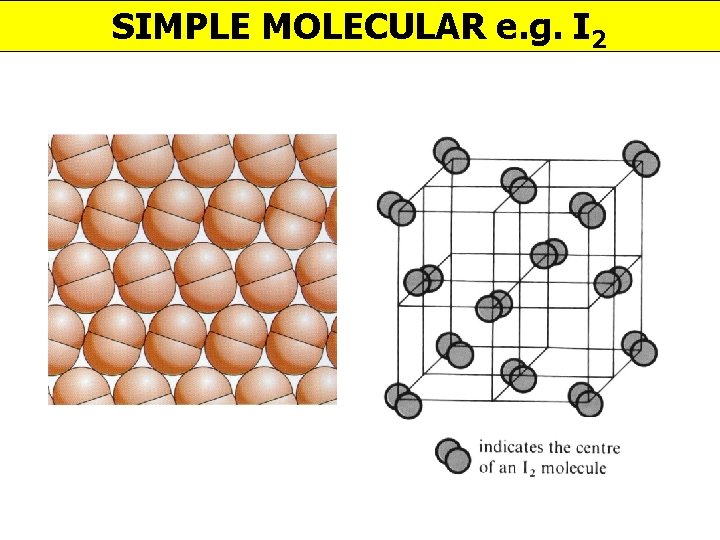
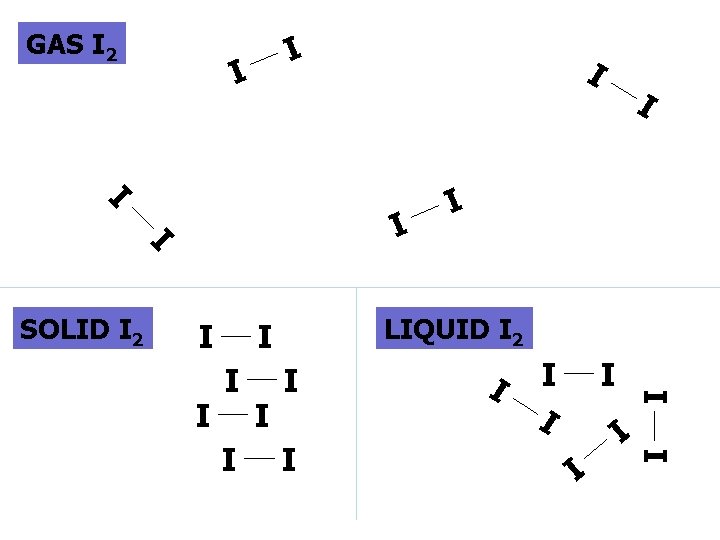
SIMPLE MOLECULAR e. g. I 2

I I SOLID I 2 I I I LIQUID I 2 I I I I GAS I 2

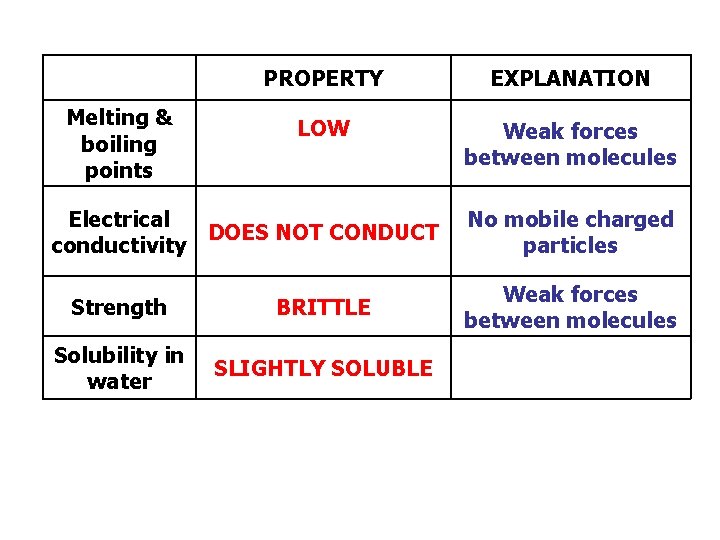
Melting & boiling points PROPERTY EXPLANATION LOW Weak forces between molecules Electrical DOES NOT CONDUCT conductivity Strength BRITTLE Solubility in water SLIGHTLY SOLUBLE No mobile charged particles Weak forces between molecules

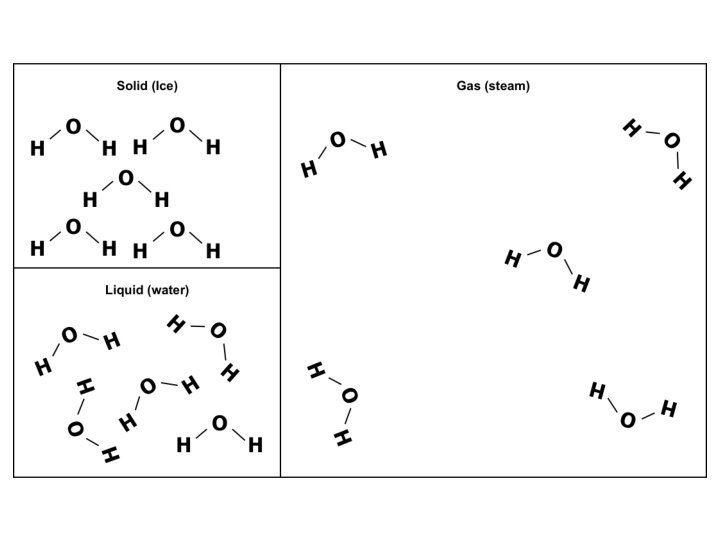
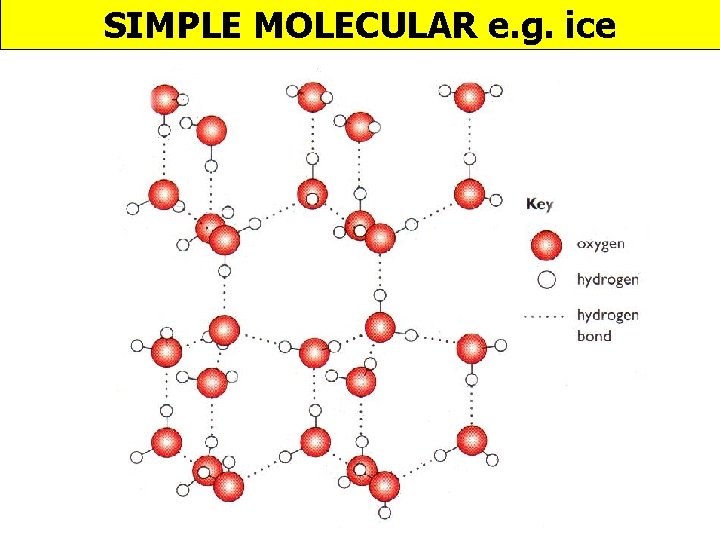
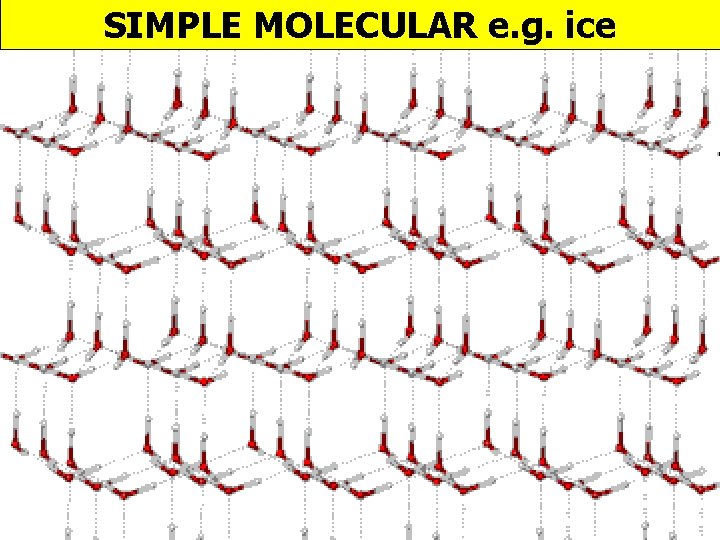
SIMPLE MOLECULAR e. g. ice

SIMPLE MOLECULAR e. g. ice

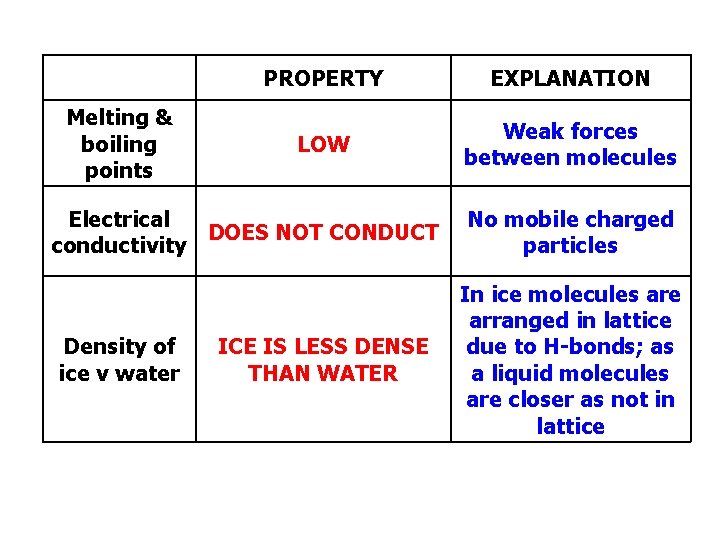
Melting & boiling points PROPERTY EXPLANATION LOW Weak forces between molecules Electrical DOES NOT CONDUCT conductivity Density of ice v water ICE IS LESS DENSE THAN WATER No mobile charged particles In ice molecules are arranged in lattice due to H-bonds; as a liquid molecules are closer as not in lattice

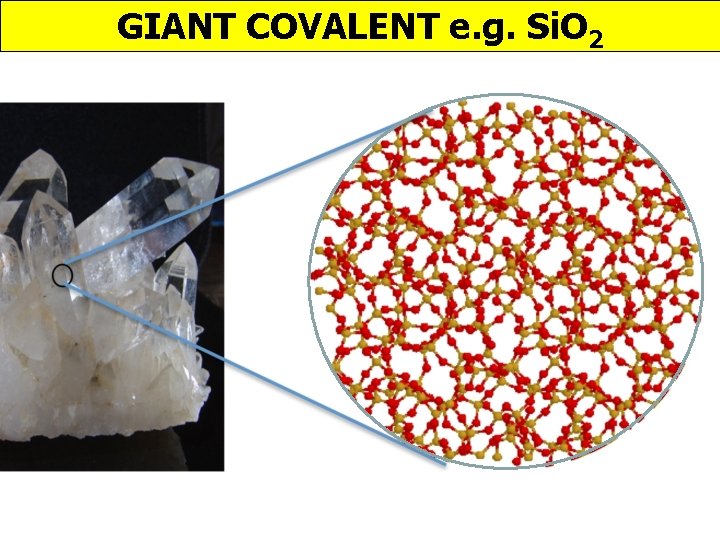
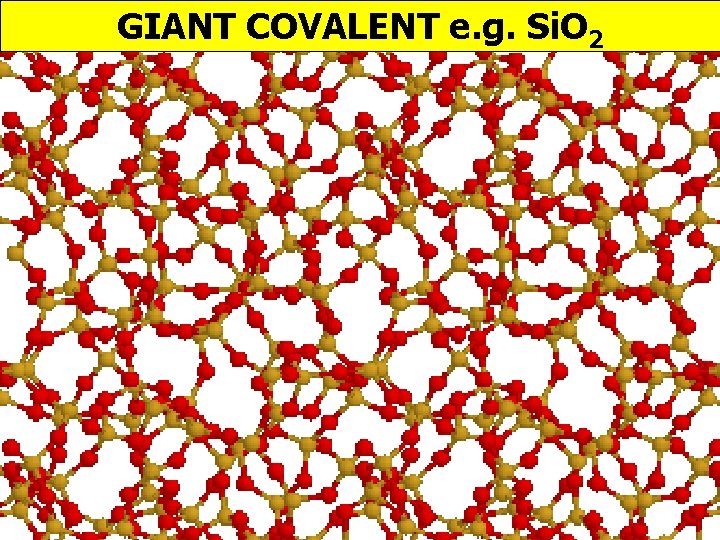
GIANT COVALENT e. g. Si. O 2

GIANT COVALENT e. g. Si. O 2

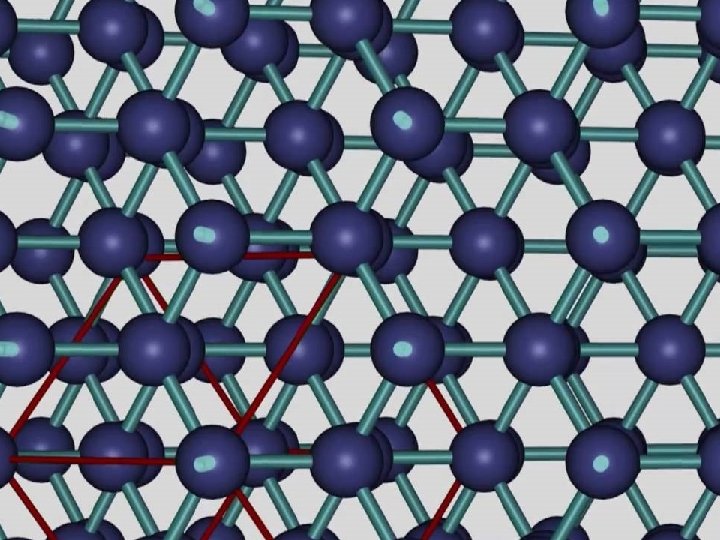
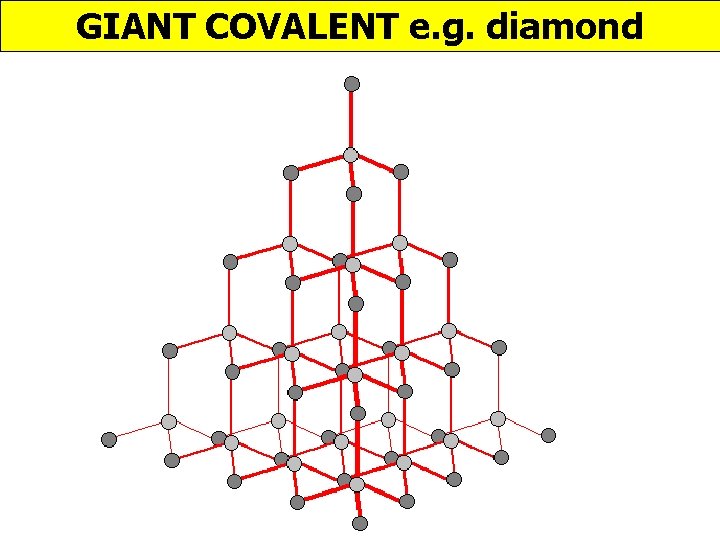
GIANT COVALENT e. g. diamond http: //www. edinformatics. com/interactive_molecules/diamond. htm Great rotatable structures (but they are NOT molecules!!)



GIANT COVALENT e. g. diamond

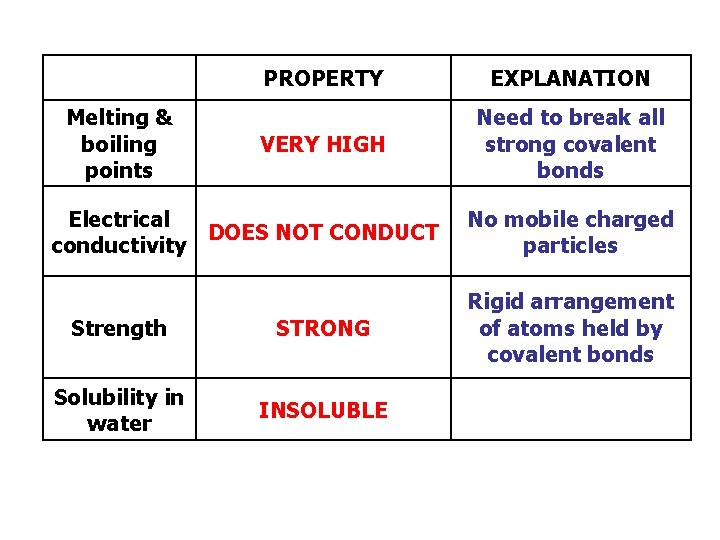
Melting & boiling points PROPERTY EXPLANATION VERY HIGH Need to break all strong covalent bonds Electrical DOES NOT CONDUCT conductivity Strength STRONG Solubility in water INSOLUBLE No mobile charged particles Rigid arrangement of atoms held by covalent bonds

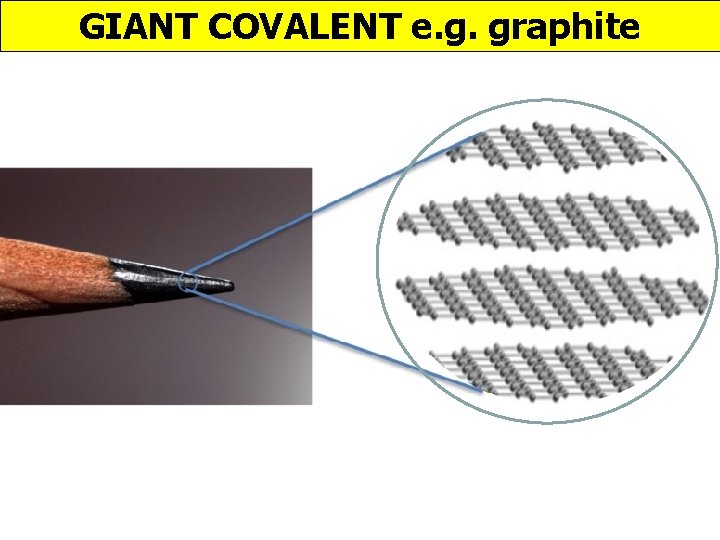
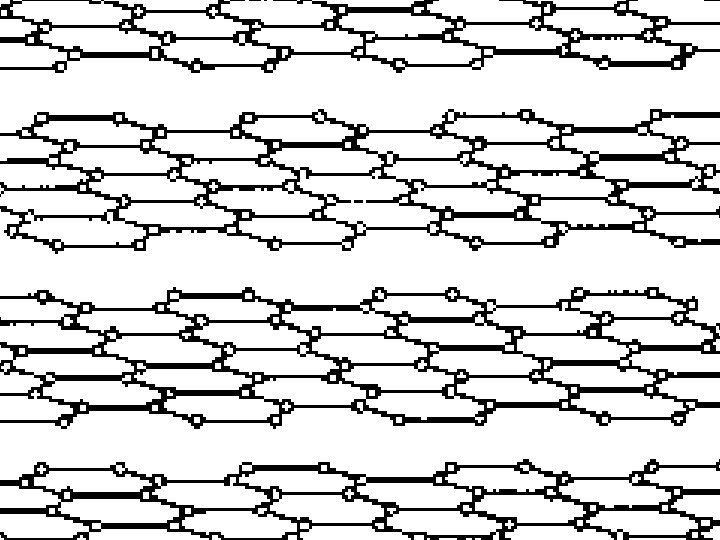
GIANT COVALENT e. g. graphite http: //www. edinformatics. com/interactive_molecules/diamond. htm Great rotatable structures (but they are NOT molecules!!)


GIANT COVALENT e. g. graphite


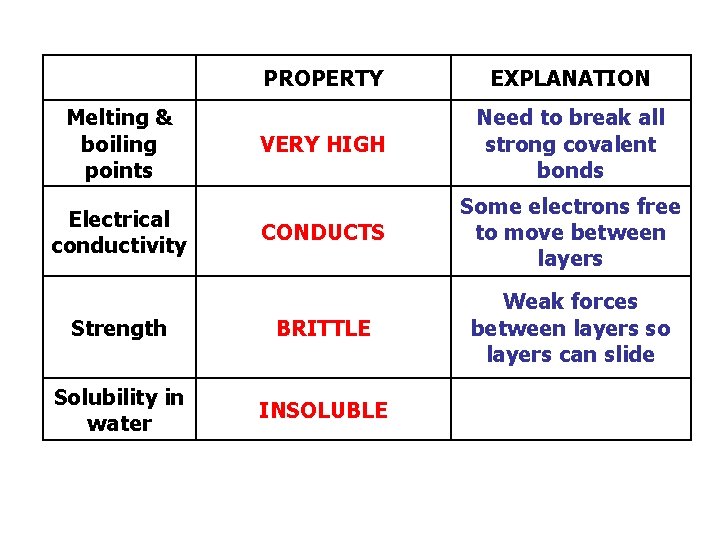
PROPERTY EXPLANATION VERY HIGH Need to break all strong covalent bonds CONDUCTS Some electrons free to move between layers Strength BRITTLE Weak forces between layers so layers can slide Solubility in water INSOLUBLE Melting & boiling points Electrical conductivity

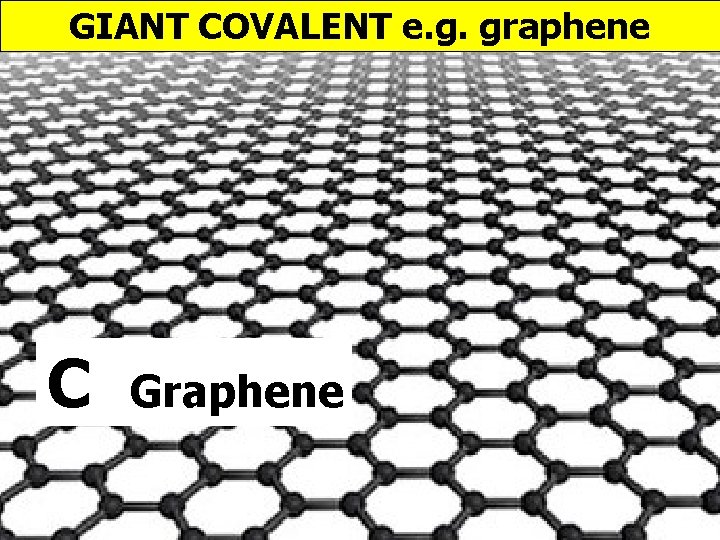
GIANT COVALENT e. g. graphene C Graphene

GIANT COVALENT e. g. graphene

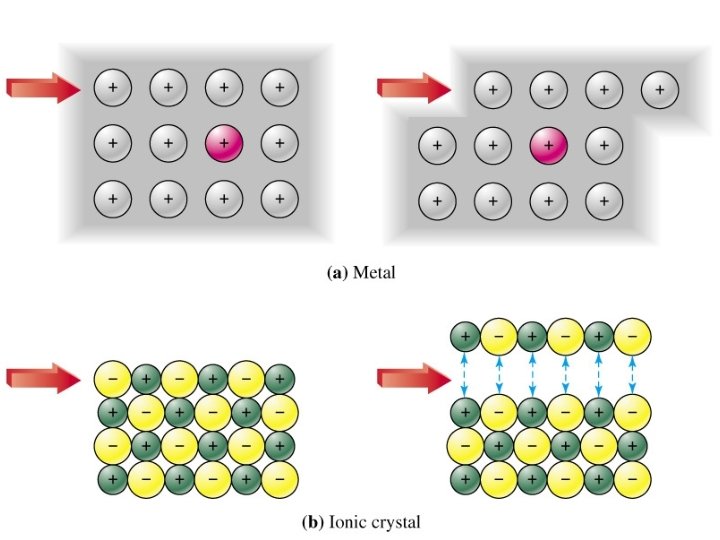
METALLIC e. g. Mg



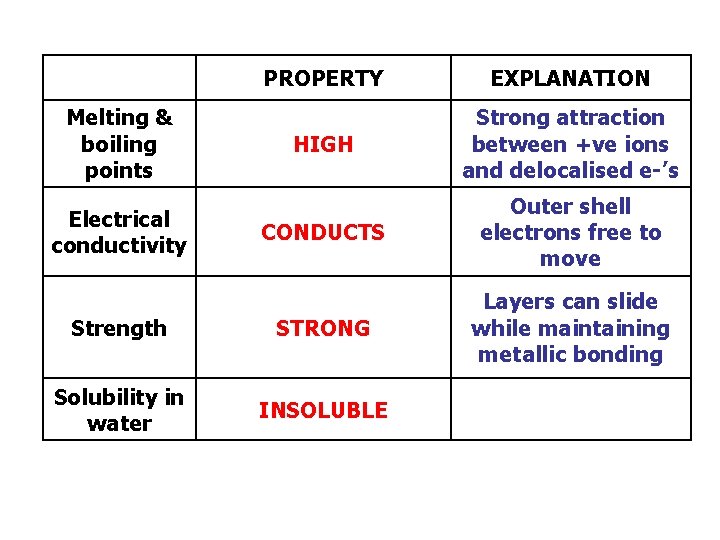
PROPERTY EXPLANATION HIGH Strong attraction between +ve ions and delocalised e-’s CONDUCTS Outer shell electrons free to move Strength STRONG Layers can slide while maintaining metallic bonding Solubility in water INSOLUBLE Melting & boiling points Electrical conductivity

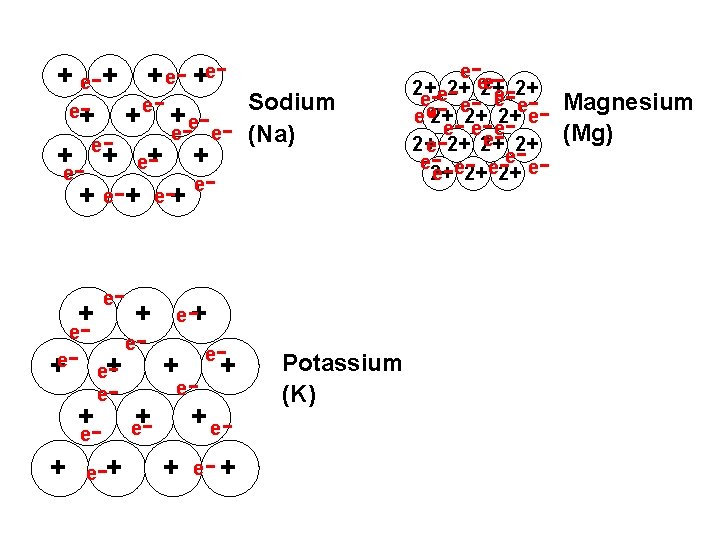
Rank these metals in order of melting points: Sodium Potassium Magnesium

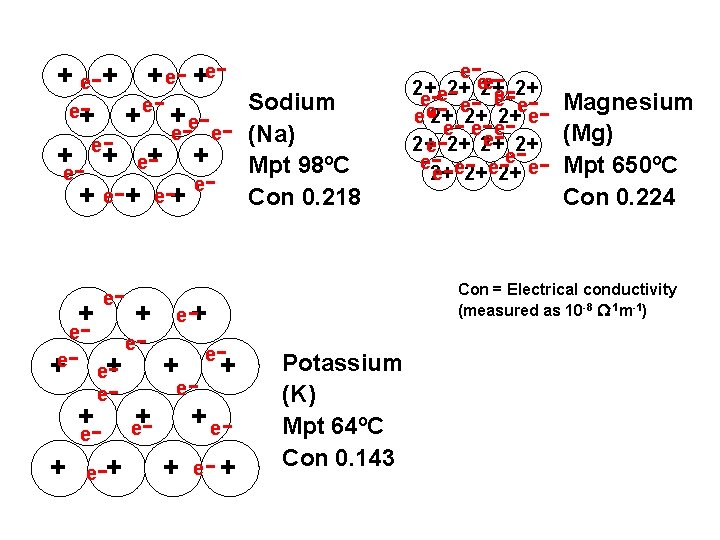
+ e-+ + e- +ee+ - + e- Sodium e- e- (Na) e+ + eee e + + e e - - +e- e+ + e - + e-+ e - - e+ + e e+ - e - -+ + e- + Potassium (K) - ---- - - - e e 2+ 2+ e e e e 2+ 2+ 2+ e e ee e 2+ 2+ Magnesium (Mg)

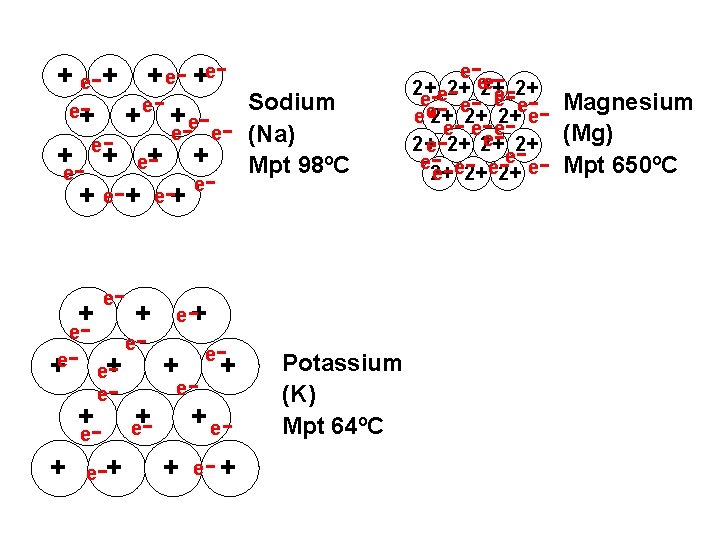
+ e-+ + e- +ee+ - + e- Sodium e- e- (Na) e+ + Mpt 98ºC eee e + + e e - - +e- e+ + e - + e-+ e - - e+ + e e+ - e - -+ + e- + Potassium (K) Mpt 64ºC - ---- - - - e e 2+ 2+ e e e e 2+ 2+ 2+ e e ee e 2+ 2+ Magnesium (Mg) Mpt 650ºC

+ e-+ + e- +ee+ - + ee- ee+ + eee e + + e e - - +e- e+ + e - + e-+ e - - + - e - Magnesium (Mg) Mpt 650ºC Con 0. 224 Con = Electrical conductivity (measured as 10 -8 -1 m-1) e+ e e+ Sodium (Na) Mpt 98ºC Con 0. 218 - ---- - - - e e 2+ 2+ e e e e 2+ 2+ 2+ e e ee e 2+ 2+ -+ + e- + Potassium (K) Mpt 64ºC Con 0. 143