Chem 11 ch 15 wkst 1 solutions 1




















- Slides: 22

Chem 11 ch 15 wkst 1 solutions

1. Define solution. A homogeneous mixture of two or more substances in a single physical state.

2. State 3 properties of solutions as discussed on page 502. Particles are very small in a solution. Particles are evenly distributed. Particles will not separate.

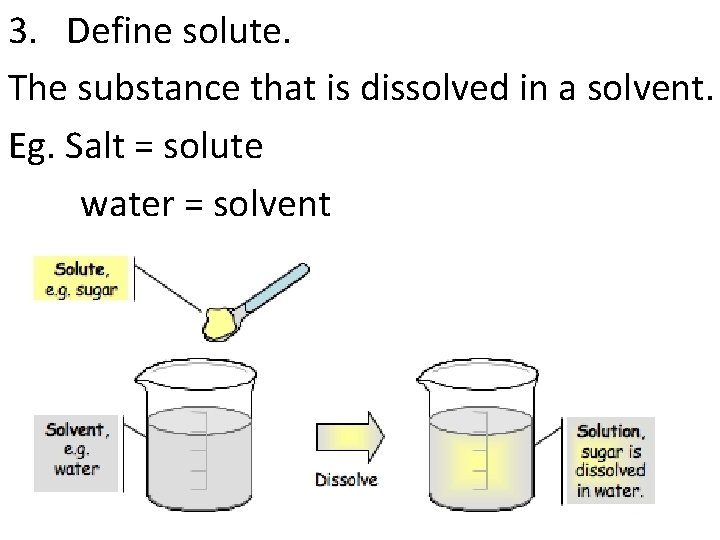
3. Define solute. The substance that is dissolved in a solvent. Eg. Salt = solute water = solvent

4. Define solvent. The substance that does the dissolving. Eg. Salt = solute water = solvent

5. Define soluble. The ability of a substance to dissolve in another substance. Usually means dissolves.

6. Define insoluble. The inability of a substance to dissolve in another substance. ie does not dissolve.

7. What are the 3 types of solutions and give and example of each. Solid solution Sterling silver = copper and silver Liquid solution Alcohol / water, salt/water, O 2 / water Gas solution Air = oxygen, nitrogen, argon, CO 2



8. Define miscible. The ability of a liquid to form a solution with another liquid in all proportions. Eg. The liquids will mix with each other like alcohol and water.

9. Define immiscible. The inability of a liquid to form a solution with another liquid in all proportions. Eg. The liquids will NOT mix with each other like oil and water

10. Define aqueous solutions. A solution in which the solvent is water

11. Define electrolyte. A substance that dissolves in water to form a solution that will conduct electricity. Eg. Salt dissolved in water = electrolytic solution due to the sodium ions and chloride ions.

12. What is the difference between an electrolyte solution and a nonelectrolyte solution and give an example of each. Electrolyte solution conducts electricity (like salt dissolved in water). Non-electrolytic solution does not conduct electricity ( like sugar dissolved in water)

13. Define concentration of solution. The amount of solute dissolved in a given amount of solution / solvent.

14. Define molarity and write the equation. The concentration of a solution in moles per liter. M = moles liter
15. What is a polar solvent? Give some examples. A liquid made up of polar molecules. Eg. Water, alcohol, acetone, acetic acid ( vinegar.

16. What is a non-polar solvent? Give some examples. A solution that is made up of non-polar molecules. Eg. Gasoline ( hexane ) oils, wax, grease, carbon tetrachloride

17. What is the rule concerning the dissolving of solutes in solvents? See chart page 515. Likes dissolves LIKE !! Polar molecules will dissolve in polar solvents. Non-polar molecules dissolve in non-polar solvents.

