Ch 25 wkst 2 carbon compounds 1 Define

Ch 25 wkst 2 carbon compounds

1. Define conformations The structure of an organic molecule that differs from another structure by only one or more bond rotations. It is the same molecule, just twisted around and bent different ways.
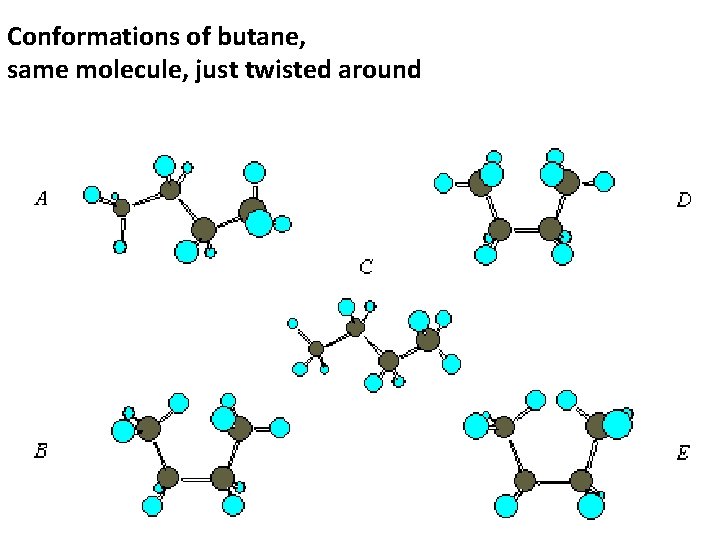
Conformations of butane, same molecule, just twisted around
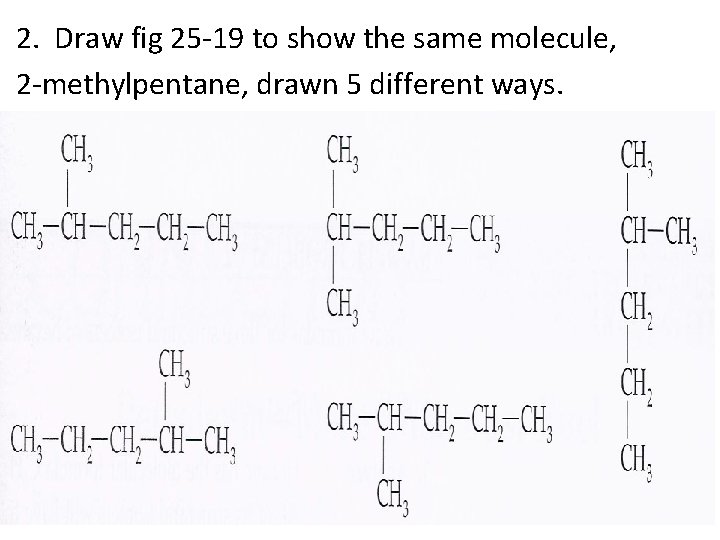
2. Draw fig 25 -19 to show the same molecule, 2 -methylpentane, drawn 5 different ways.

3. Define structural isomers. A molecule that has the same molecular formula as another molecule, but has atoms bonded in a different order.
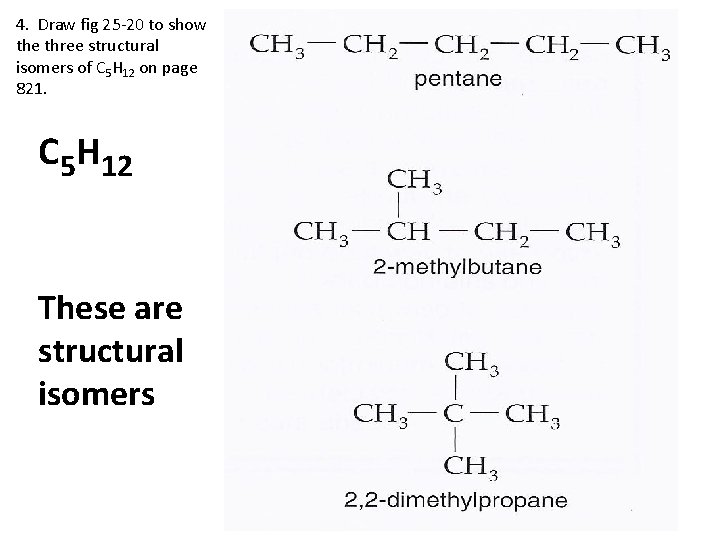
4. Draw fig 25 -20 to show the three structural isomers of C 5 H 12 on page 821. C 5 H 12 These are structural isomers
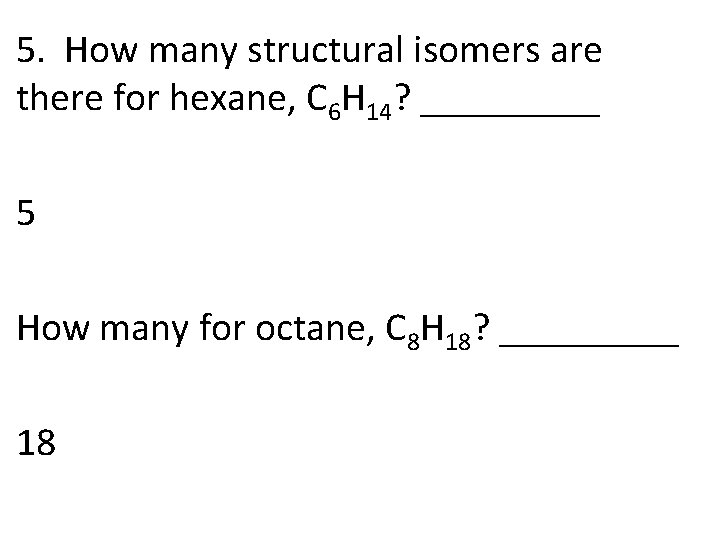
5. How many structural isomers are there for hexane, C 6 H 14? _____ 5 How many for octane, C 8 H 18? _____ 18

6. Try the practice problems on page 822. Draw 4 structural isomers of heptane. Name the isomers with only one branch. 3 -ethylhexane is a structural isomer of which straight chain alkane?

7. Read page 823 about petroleum. 8. What are some of the important products to come from refining petroleum or crude oil? Propane, butane, gasoline and other fuels, plastics

9. Define cyclic hydrocarbon. Hydrocarbons that contain a carbon ring. 10. Define cycloalkane. Cyclic hydrocarbons with only single bonds. 11. What is the general formula for cycloalkane? Cn. H 2 n
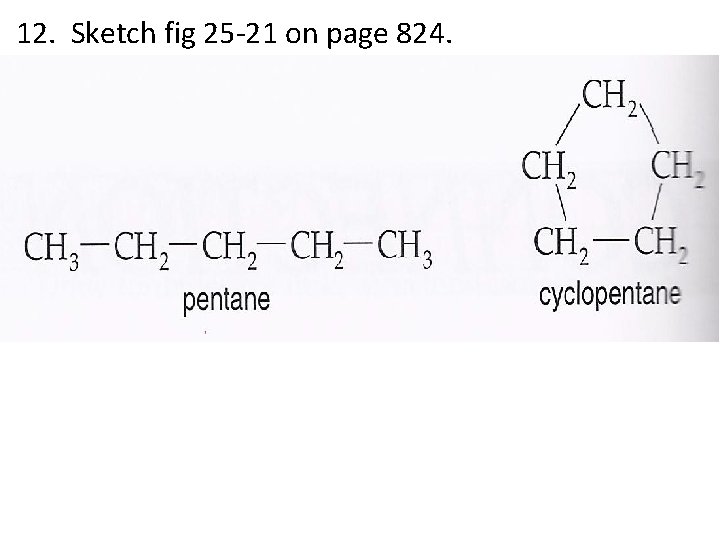
12. Sketch fig 25 -21 on page 824.

13. Sketch the structural formula for the following: Cyclopropane cyclobutane cyclohexane cyclooctane

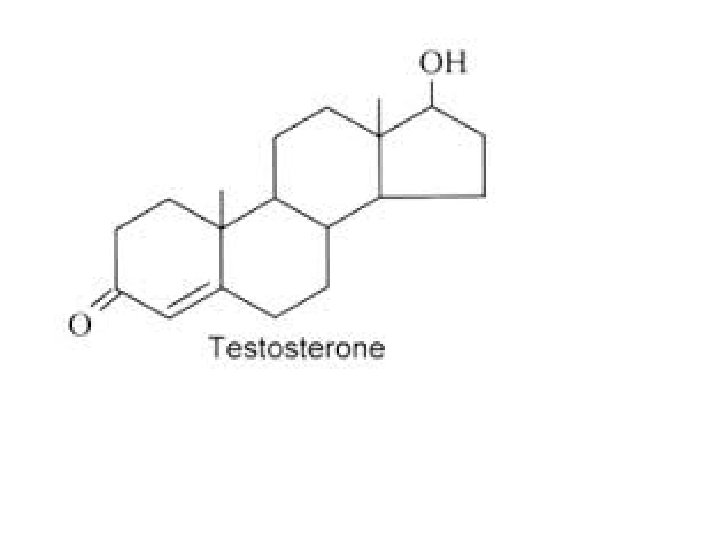
14. What are some important organic molecules that cyclohexane is a part of? Glucose which is the building block of most carbohydrates like sucrose, starch, etc Cholesterol needed for cell structure Steroid hormones
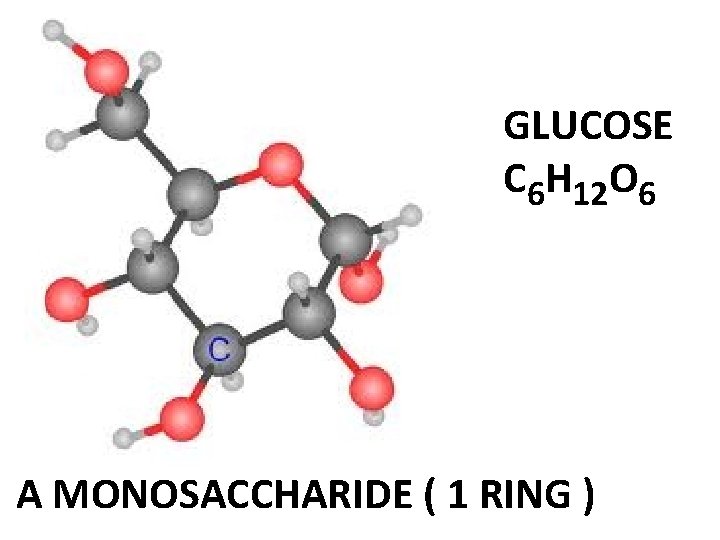
GLUCOSE C 6 H 12 O 6 A MONOSACCHARIDE ( 1 RING )
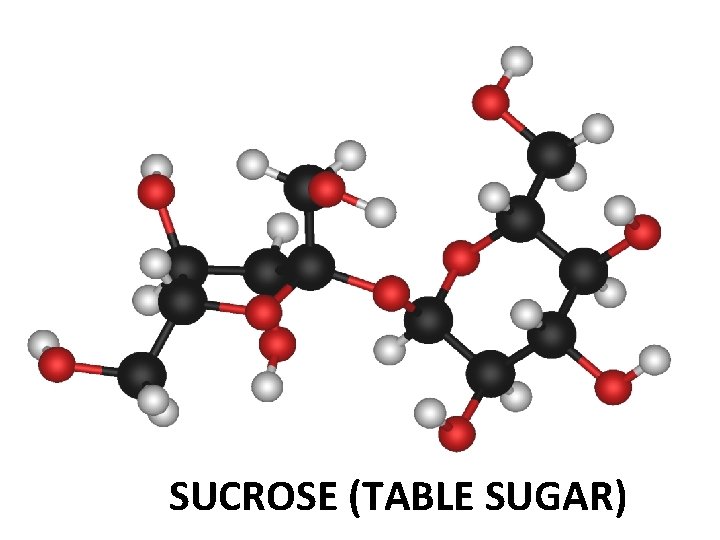
SUCROSE (TABLE SUGAR)
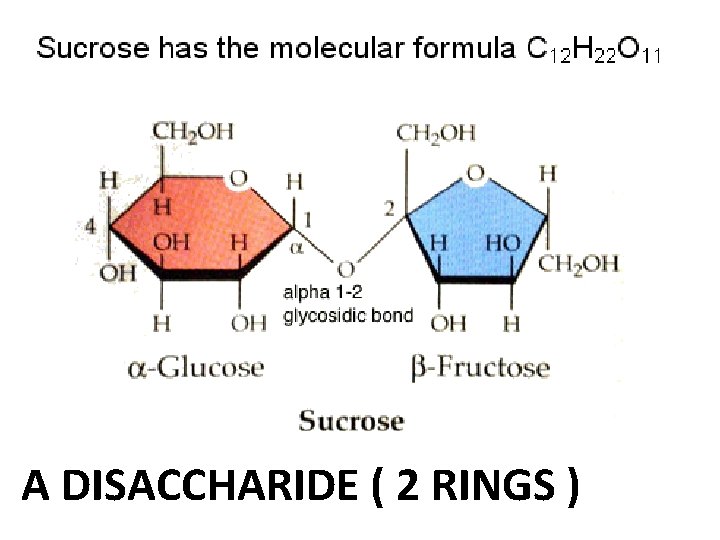
A DISACCHARIDE ( 2 RINGS )

15. Define unsaturated hydrocarbon and what do they contain at least one of? The class of hydrocarbons that are not filled to capacity. They contain at least one double bond or one triple bond between carbon atoms.
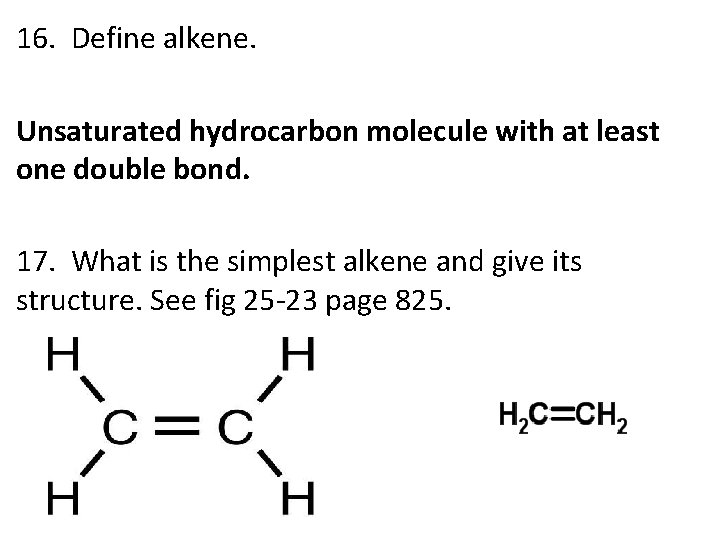
16. Define alkene. Unsaturated hydrocarbon molecule with at least one double bond. 17. What is the simplest alkene and give its structure. See fig 25 -23 page 825.
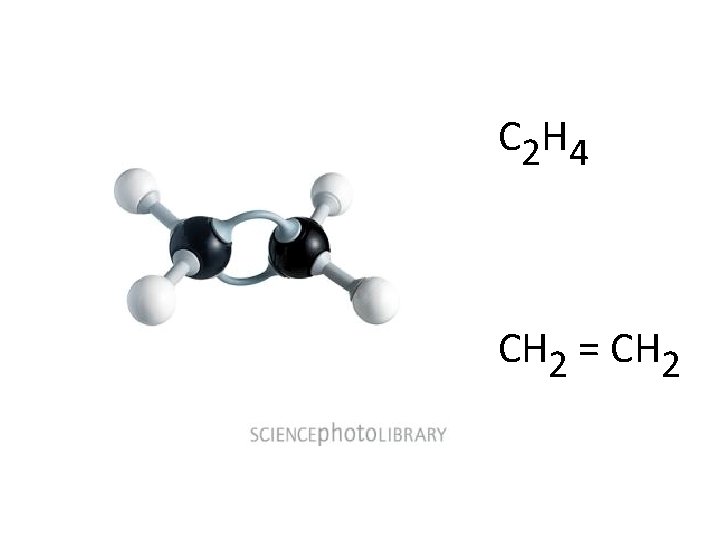
C 2 H 4 CH 2 = CH 2
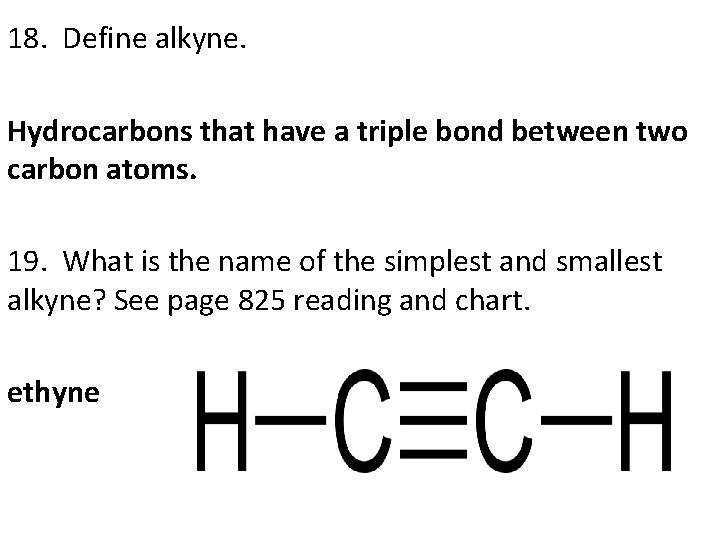
18. Define alkyne. Hydrocarbons that have a triple bond between two carbon atoms. 19. What is the name of the simplest and smallest alkyne? See page 825 reading and chart. ethyne

20. Give the general formula for: a. alkanes b. alkenes Cn. H 2 n+2 Cn. H 2 n c. alkynes Cn. H 2 n-2 21. Using the above formulae, determine the formula of: a. an alkane with 6 carbons C 6 H 14 b. an alkene with 6 carbons C 6 H 12 c. an alkyne with 6 carbons. C 6 H 10

22. Copy the chart fig 25 -24 page 825 on hydrocarbon structures.
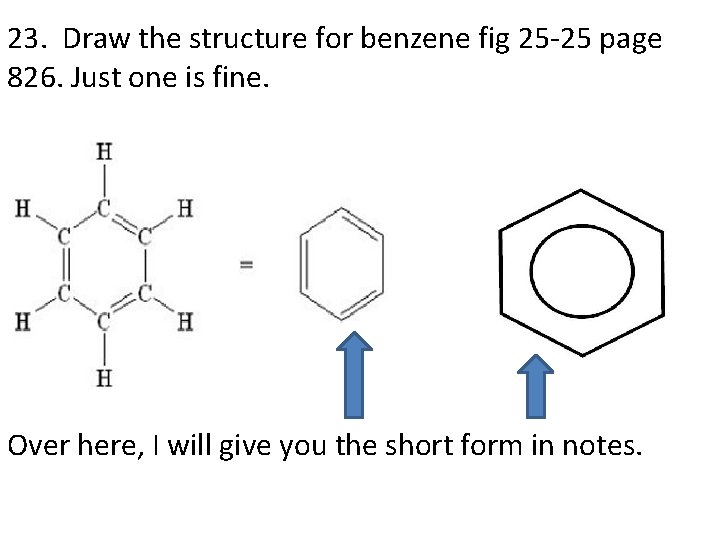
23. Draw the structure for benzene fig 25 -25 page 826. Just one is fine. Over here, I will give you the short form in notes.

24. Using both forms of benzene, draw the structures for: a. chlorobenzene b. 1, 2 -dichlorobenzene ortho c. 1, 3 -dichlorobenzene meta d. 1, 4 -dichlorobenzene para

25. Why are molecules of benzene called aromatic compounds and what other important biological molecules is benzene part of? Many benzene compounds have distinctive odors. eg cloves, vanilla, almonds, They are also part of larger biological molecules such as nucleic acids found in DNA, RNA, vitamins, hormones and many pharmaceutical drugs.
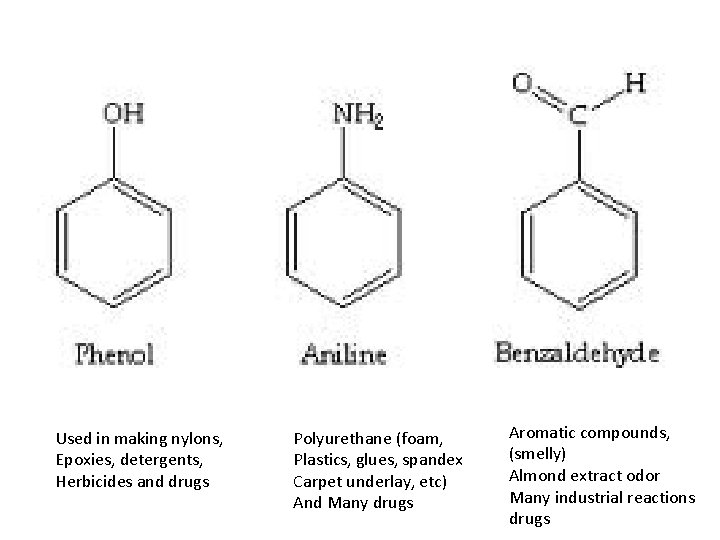
Used in making nylons, Epoxies, detergents, Herbicides and drugs Polyurethane (foam, Plastics, glues, spandex Carpet underlay, etc) And Many drugs Aromatic compounds, (smelly) Almond extract odor Many industrial reactions drugs
- Slides: 27