Bell Work 1615 Turn in Reactions Wkst 1

Bell Work 1/6/15 • Turn in Reactions Wkst #1 • Have the following on your desk �Chemical Reactions Notes #2 �Reactions Wkst #2 �Reference Sheet from yesterday • What are the five proofs/evidence of a chemical change?

Unit 7 Chemical Reactions Powerpoint #2 Proof of Rxn, Reaction Types, and Activity Series

Indications/Evidence/ Proof of a Chemical Reaction Let me give you some hints.

Is it a chemical reaction? C. Johannesson If a gas is produced it might be a chemical reaction. Look for the bubbles!

Is it a chemical reaction? C. Johannesson If a change in odor is detected, it might be a chemical reaction.

Is it a chemical reaction? C. Johannesson If a precipitate is formed it might be a chemical reaction. A solid is left behind.

Is it a chemical reaction? C. Johannesson If it changes color it might be a chemical reaction. Like iron rusting!

Is it a chemical reaction? C. Johannesson If it gets hotter, it may be a chemical reaction. If evolution of heat and light, it might be a chemical reaction.

Is it a chemical reaction? C. Johannesson Or…if it gets colder it might be a chemical reaction.

Energy in Reactions • Endothermic. Energy moves from the surroundings into the source EX: Cold packs, Alka-seltzer • Exothermic. Energy moves from the source out to the surroundings EX: Light a match, fuel

What is a good way to remember the 5 indications/evidence/proof of a chemical reaction? GOPEC (gas, odor, precipitate, energy, color)

Five Types of Chemical Reactions Synthesis reaction A and B stand for either elements or compounds in all reactions. A + B AB Example: Solid sodium reacts with chlorine gas to produce solid sodium chloride Skeleton Equation: Na(s) + Cl 2(g) Na. Cl (s)

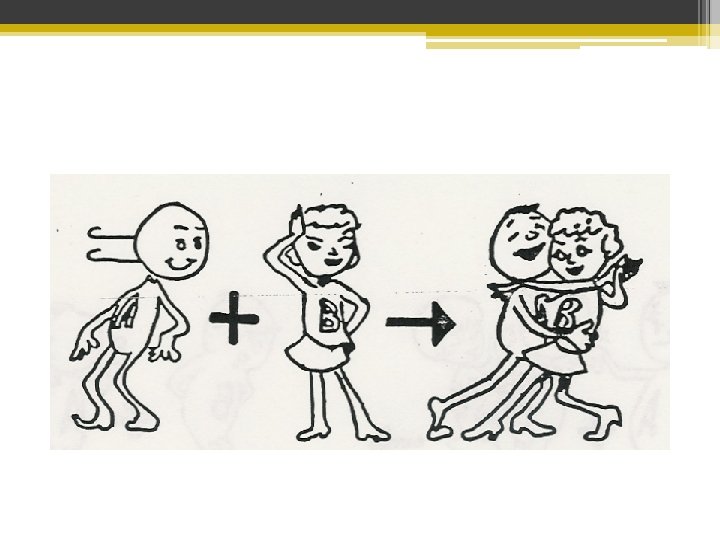
Synthesis Reaction Like Marriage

Decomposition reaction AB stands for a compound. A and B can either be elements or smaller compounds. AB A + B Examples: Liquid water decomposes into hydrogen gas and oxygen gas Skeleton Equation: H 2 O (l) H 2 (g) + O 2 (g)

Decomposition Reaction Like Divorce

Single-replacement reaction BX and AX are usually ionic compounds, A and B are elements. A + BX AX + B Example: Solid magnesium and aqueous copper (II) sulfate react form aqueous magnesium sulfate and solid copper. Skeleton Equation: Mg (s) + Cu. SO 4 (aq) Mg. SO 4 (aq) + Cu (s)
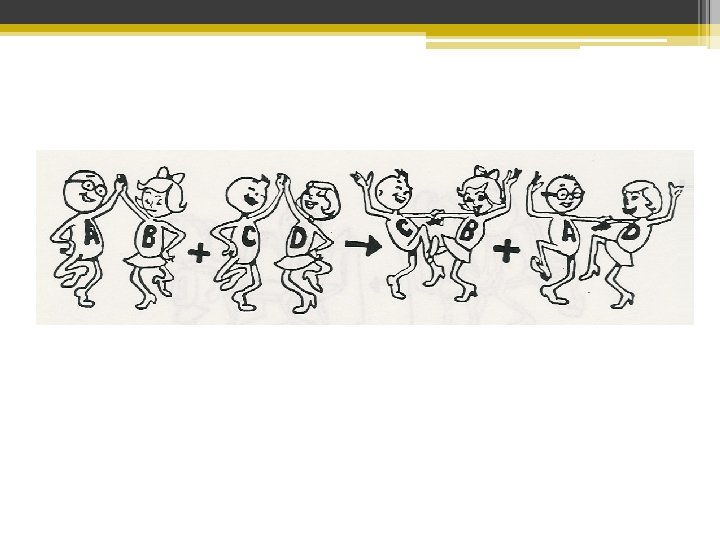
Single Replacement Like Switching Out One Dance Partner

Double-replacement Reaction AX, BY, AY, and BX are usually ionic compounds that are dissolved in water (aqueous). Sometimes a solid precipitate is formed. AX + BY AY + BX Example: Aqueous sodium bromide and aqueous magnesium sulfide react to form aqueous sodium sulfide and aqueous magnesium bromide. Skeleton Equation: Na. Br (aq) + Mg. S (aq) Na 2 S (aq) + Mg. Br 2 (aq)

Double Replacement Like Switching Both Dance Partners

Combustion Reaction Usually a compound containing carbon and hydrogen combines with oxygen to produce carbon dioxide and water Cx. Hy + O 2 CO 2 + H 2 O Example: Gaseous carbon tetrahydride burns in gaseous oxygen to produce gaseous carbon dioxide and liquid water Skeleton Equation: CH 4 (g) + O 2 (g) CO 2 (g) + H 2 O (l)

Type of Reaction? • Na. Cl + F 2 Na. F + Cl 2 • P + O 2 P 2 O 5 SR S • Fe. Cl 3 + Na. OH Fe(OH)3 + Na. Cl • Ag 2 O Ag + O 2 D • C 4 H 8 + O 2 CO 2+ H 2 O C DR
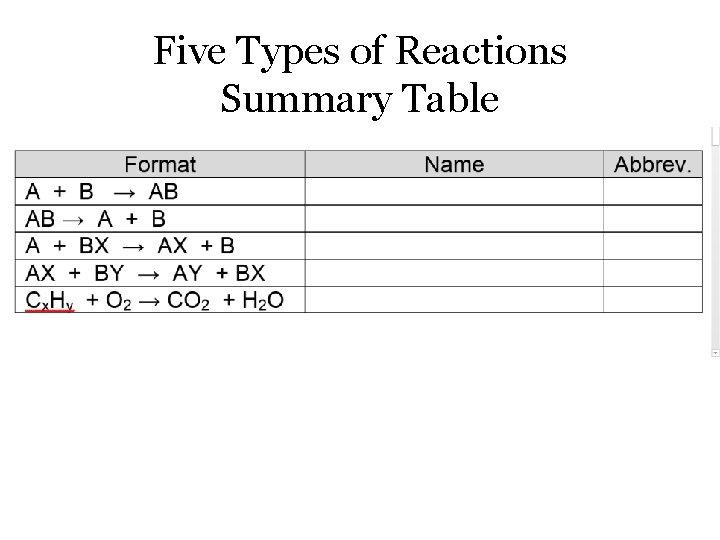
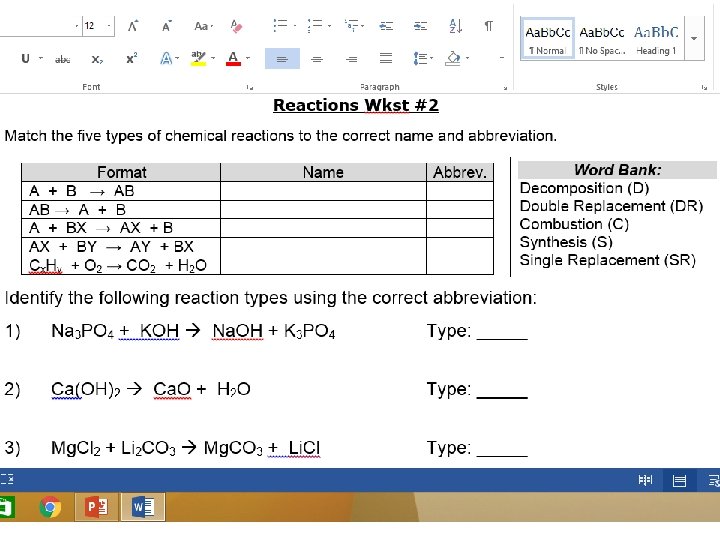
Five Types of Reactions Summary Table


How to determine if a single replacement reaction will happen? Use the Activity Series ▫ Ability of an element to react ▫ The easier it reacts, the higher the activity ▫ List of elements organized from highest to lowest activity

Activity Series • Metals ▫ greater activity, easier to lose electrons ▫ Why? easier to become a cation • Non-Metals ▫ greater activity, easier to gain electrons ▫ Why? easier to become an anion

An element can replace anything below Lithium rubidium potassium barium calcium sodium magnesium aluminum manganese zinc chromium iron cobalt nickel tin lead hydrogen copper mercury silver platinum gold fluorine chlorine bromine iodine D e c r e a x i n g A c t i v i t y Activity Series • Most active element is on top • an element can replace anything below it but not any above it • Ex: Ca + Pb(NO 3)2 Ca(NO 3)2+ Pb Cu + Li 2 SO 4 no reaction

Practice • Zn(s) + HCl(aq) Zn. Cl 2 + H 2 • Br 2 + Fe. Cl 2 no reaction

- Slides: 33