Chapter 18 Enols and Enolates Overview Enols Enolates


- Slides: 25

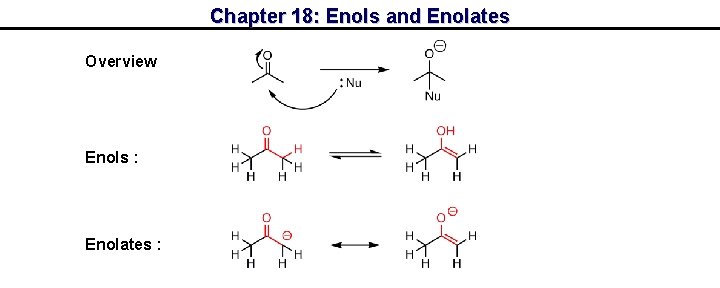
Chapter 18: Enols and Enolates Overview Enols : Enolates :

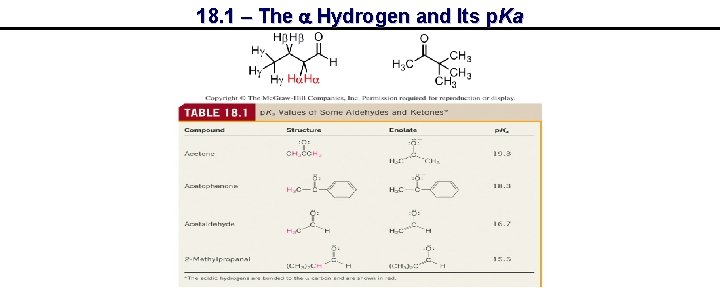
18. 1 – The a Hydrogen and Its p. Ka

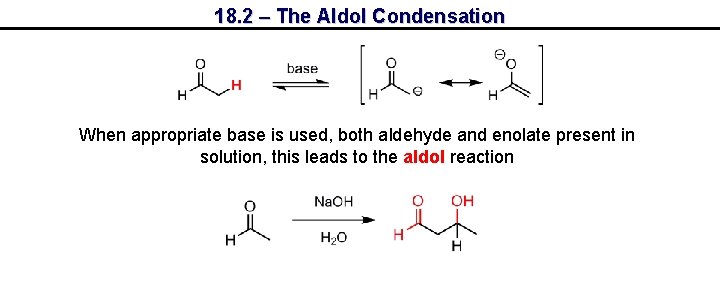
18. 2 – The Aldol Condensation When appropriate base is used, both aldehyde and enolate present in solution, this leads to the aldol reaction

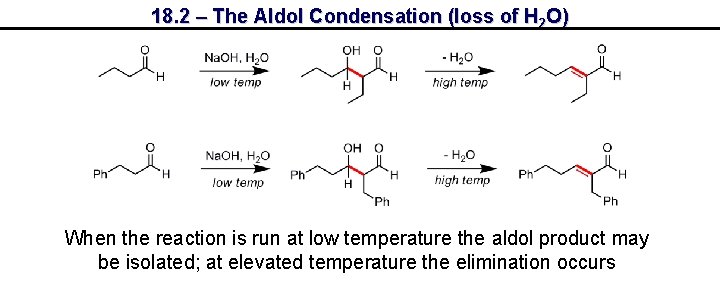
18. 2 – The Aldol Condensation (loss of H 2 O) When the reaction is run at low temperature the aldol product may be isolated; at elevated temperature the elimination occurs

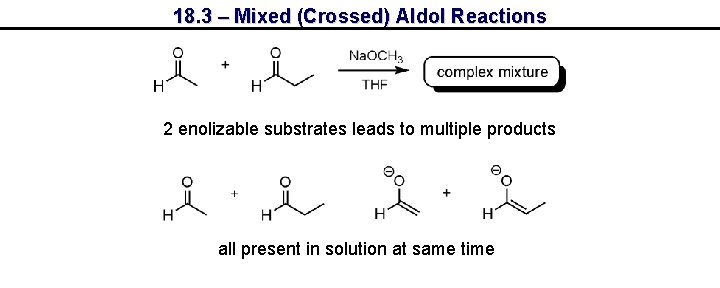
18. 3 – Mixed (Crossed) Aldol Reactions 2 enolizable substrates leads to multiple products all present in solution at same time

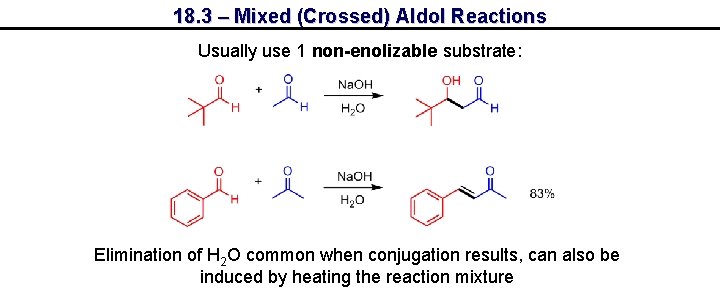
18. 3 – Mixed (Crossed) Aldol Reactions Usually use 1 non-enolizable substrate: Elimination of H 2 O common when conjugation results, can also be induced by heating the reaction mixture

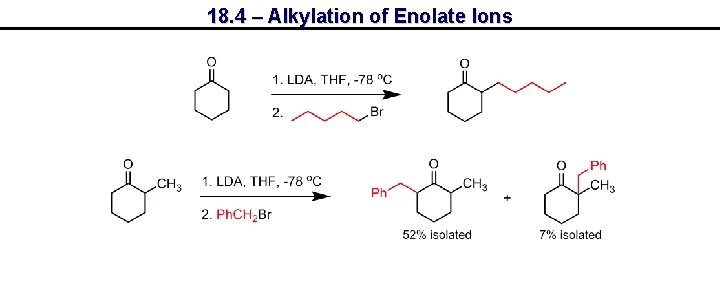
18. 4 – Alkylation of Enolate Ions

Staphylococcus aureus (MRSA, VRSA) • Greek staphyle meaning “a bunch of grapes” • Greek kokkos meaning “berry” • aureus = yellow

• Gram-positive, cluster-forming coccus • Cause food poisoning, endocarditis, osteomyelitis • Can cause septiceamia, infections on implants • Becoming increasingly resistant to antibiotics • MRSA strains appeared in 1961 • VRSA first reported in the USA in 2002

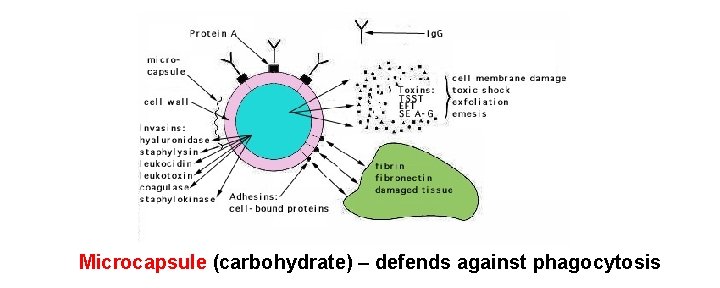
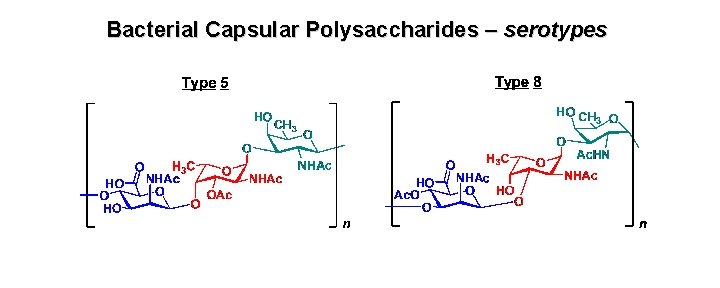
Microcapsule (carbohydrate) – defends against phagocytosis

Bacterial Capsular Polysaccharides – serotypes


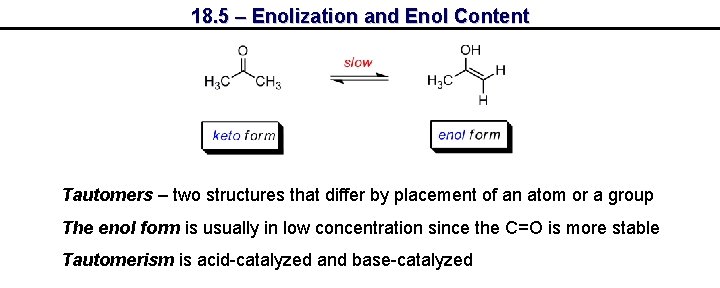
18. 5 – Enolization and Enol Content Tautomers – two structures that differ by placement of an atom or a group The enol form is usually in low concentration since the C=O is more stable Tautomerism is acid-catalyzed and base-catalyzed

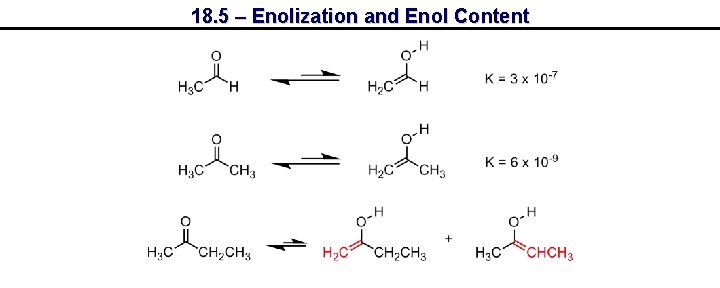
18. 5 – Enolization and Enol Content

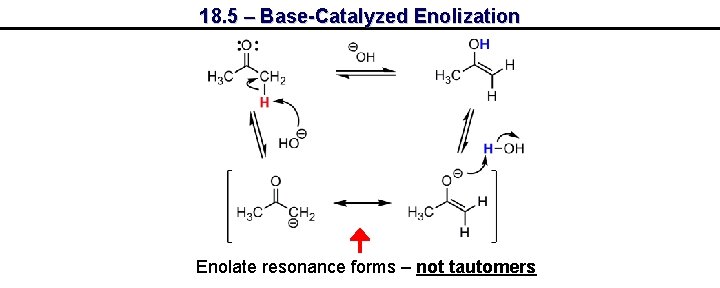
18. 5 – Base-Catalyzed Enolization Enolate resonance forms – not tautomers

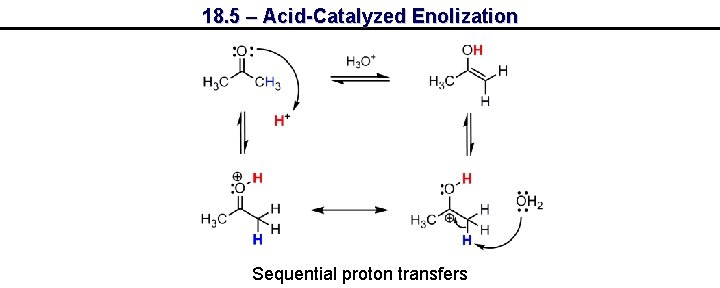
18. 5 – Acid-Catalyzed Enolization Sequential proton transfers

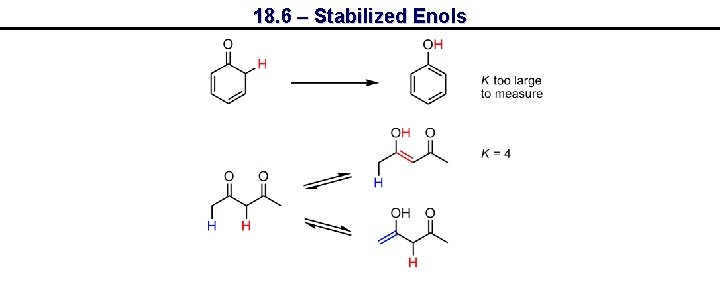
18. 6 – Stabilized Enols

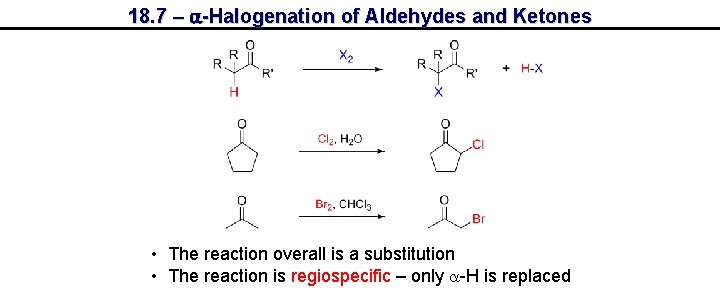
18. 7 – a-Halogenation of Aldehydes and Ketones • The reaction overall is a substitution • The reaction is regiospecific – only a-H is replaced

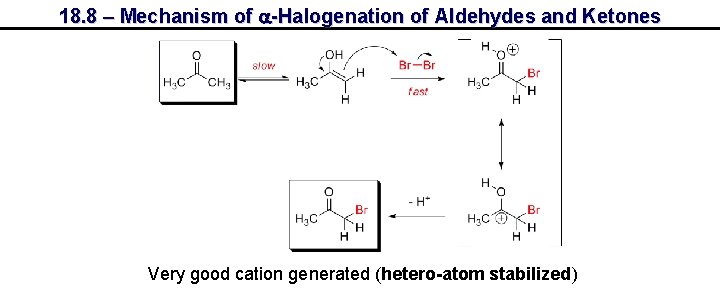
18. 8 – Mechanism of a-Halogenation of Aldehydes and Ketones Very good cation generated (hetero-atom stabilized)

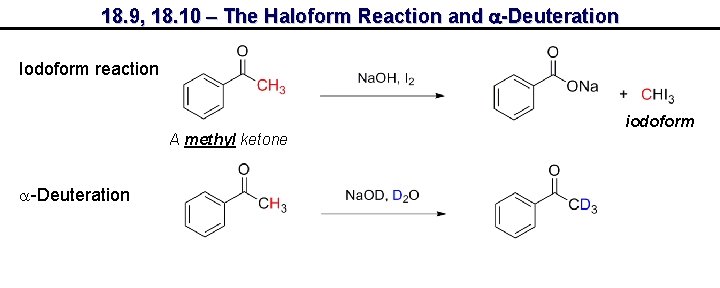
18. 9, 18. 10 – The Haloform Reaction and a-Deuteration Iodoform reaction iodoform A methyl ketone a-Deuteration

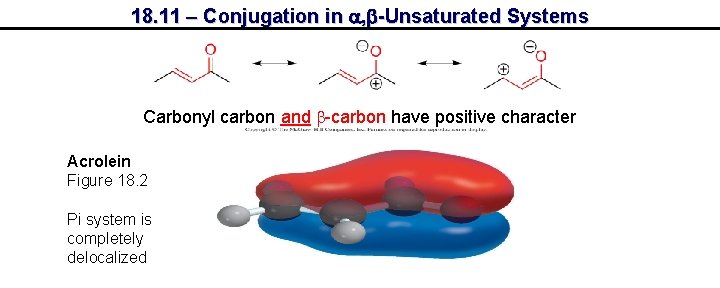
18. 11 – Conjugation in a, b-Unsaturated Systems Carbonyl carbon and b-carbon have positive character Acrolein Figure 18. 2 Pi system is completely delocalized

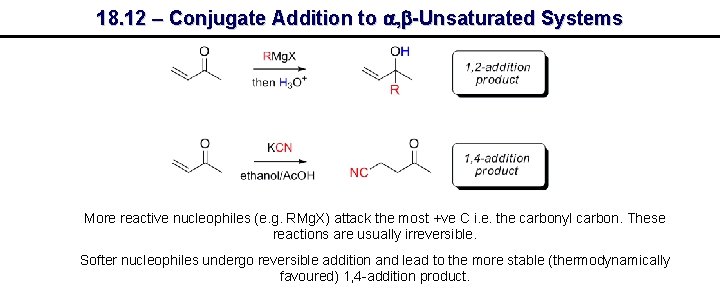
18. 12 – Conjugate Addition to a, b-Unsaturated Systems More reactive nucleophiles (e. g. RMg. X) attack the most +ve C i. e. the carbonyl carbon. These reactions are usually irreversible. Softer nucleophiles undergo reversible addition and lead to the more stable (thermodynamically favoured) 1, 4 -addition product.

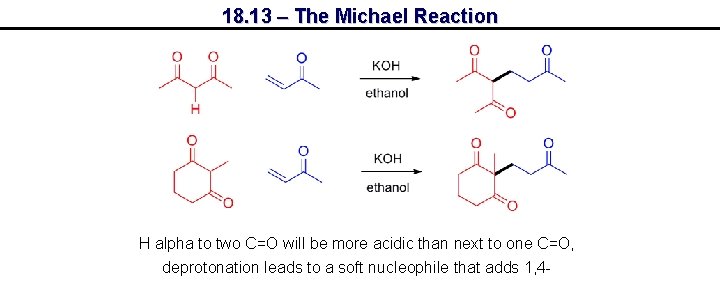
18. 13 – The Michael Reaction H alpha to two C=O will be more acidic than next to one C=O, deprotonation leads to a soft nucleophile that adds 1, 4 -

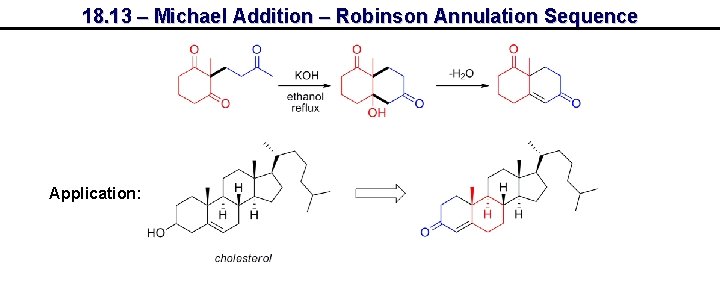
18. 13 – Michael Addition – Robinson Annulation Sequence Application:

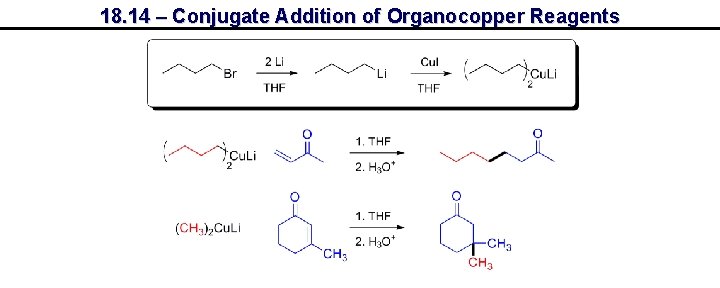
18. 14 – Conjugate Addition of Organocopper Reagents