Asymmetric Synthesis Substitution using chiral enolates Outline n

Asymmetric Synthesis α-Substitution using chiral enolates

Outline n n Requirements for stereoselective enolate alkylation Imposing facial bias on alkylation Chiral auxiliaries leading to acids or alcohols Chiral auxiliaries leading to ketones

Requirements for stereoselective enolate alkylation n Only one geometrical isomer must be formed • • n Base Solvent Temperature Counter-ion Chiral auxiliary

Stereoelectronic requirements for deprotonation and alkylation n Maximum overlap of orbitals during bond-breaking and formation Preferred conformation during deprotonation Electrophile approach is slightly away from perpendicular, towards the auxiliary
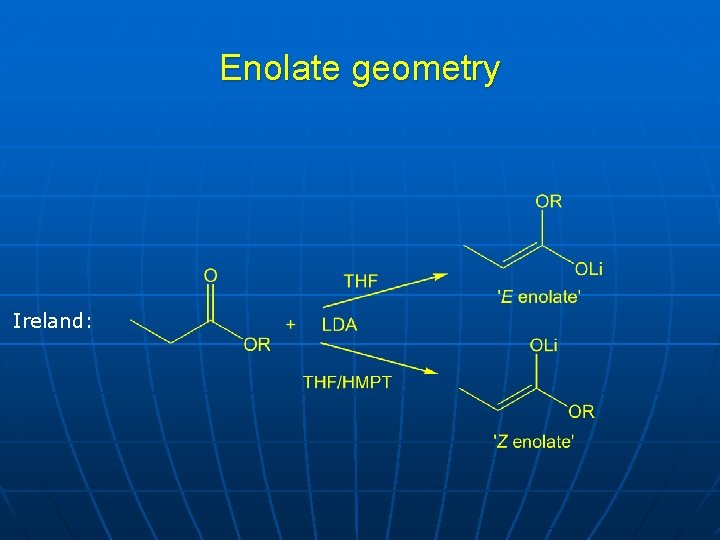
Enolate geometry Ireland:
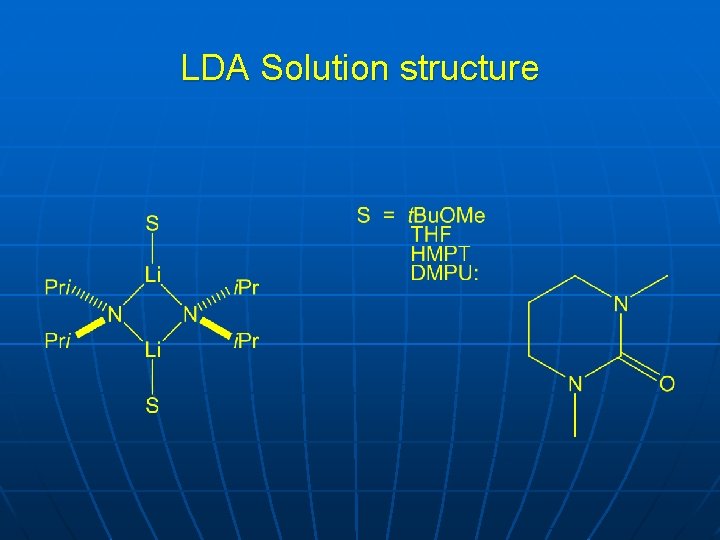
LDA Solution structure
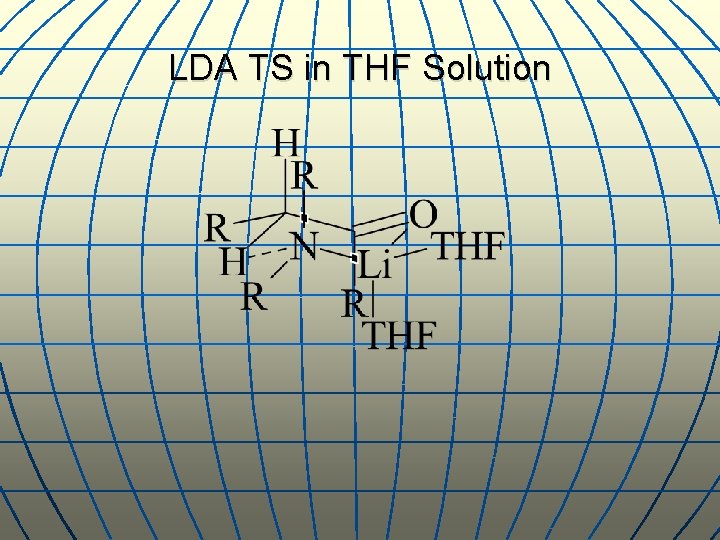
LDA TS in THF Solution
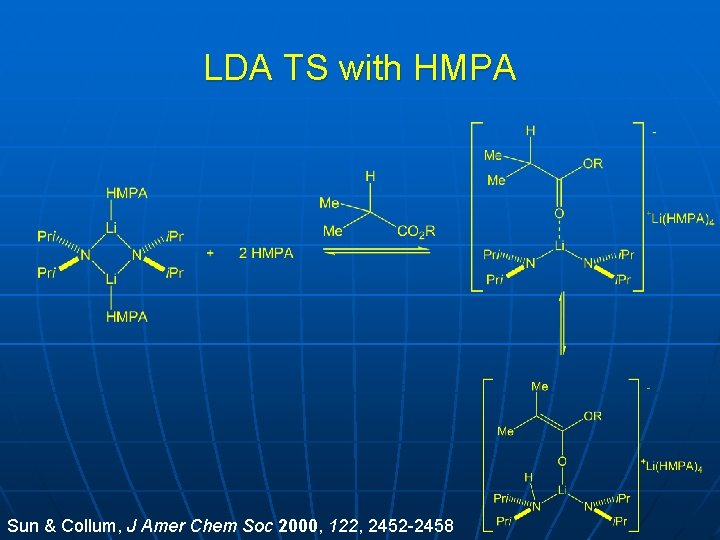
LDA TS with HMPA Sun & Collum, J Amer Chem Soc 2000, 122, 2452 -2458
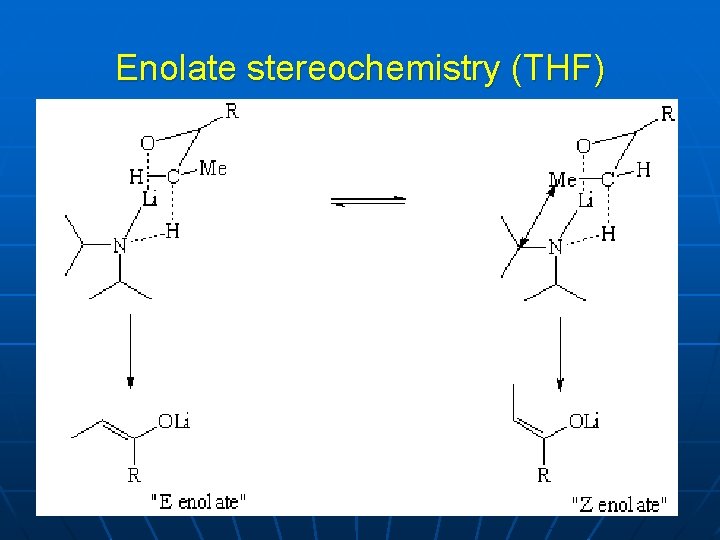
Enolate stereochemistry (THF)
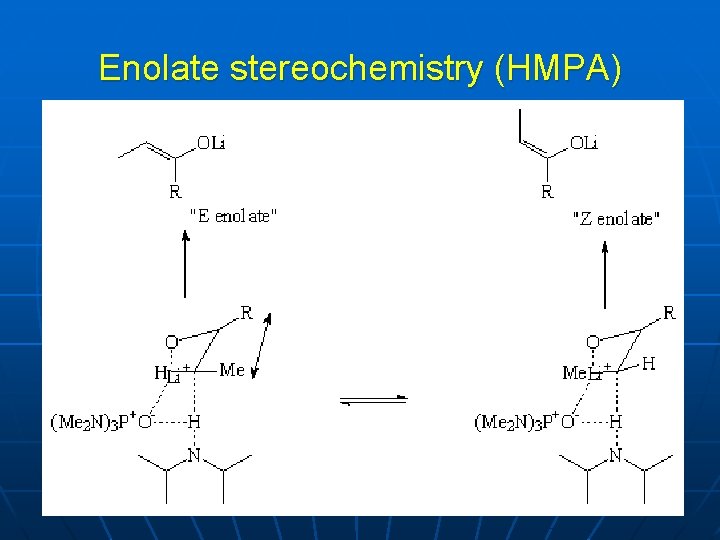
Enolate stereochemistry (HMPA)
![Other effects n n n % HMPA Substituent (R) size [Bu. Li] % hexane Other effects n n n % HMPA Substituent (R) size [Bu. Li] % hexane](http://slidetodoc.com/presentation_image/caf78e69a54d39f54cd12722fa4242e3/image-11.jpg)
Other effects n n n % HMPA Substituent (R) size [Bu. Li] % hexane [Li. Cl] or Li. Br] Other bases
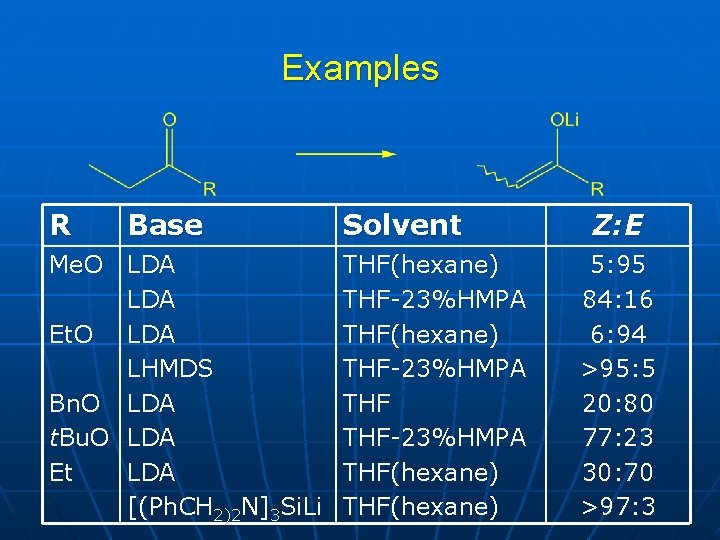
Examples R Base Me. O LDA Et. O LDA LHMDS Bn. O LDA t. Bu. O LDA Et LDA [(Ph. CH 2)2 N]3 Si. Li Solvent THF(hexane) THF-23%HMPA THF(hexane) Z: E 5: 95 84: 16 6: 94 >95: 5 20: 80 77: 23 30: 70 >97: 3
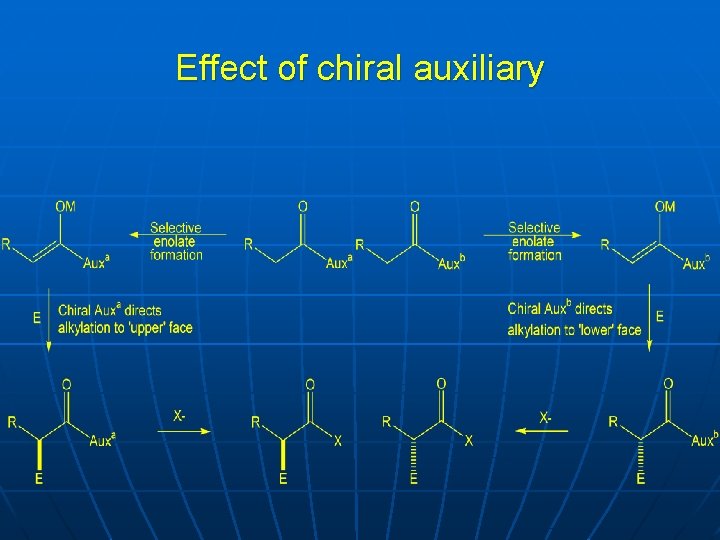
Effect of chiral auxiliary
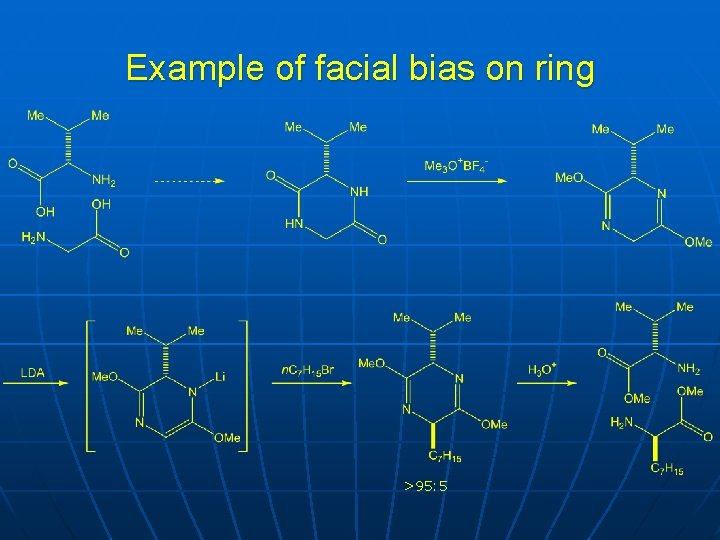
Example of facial bias on ring >95: 5
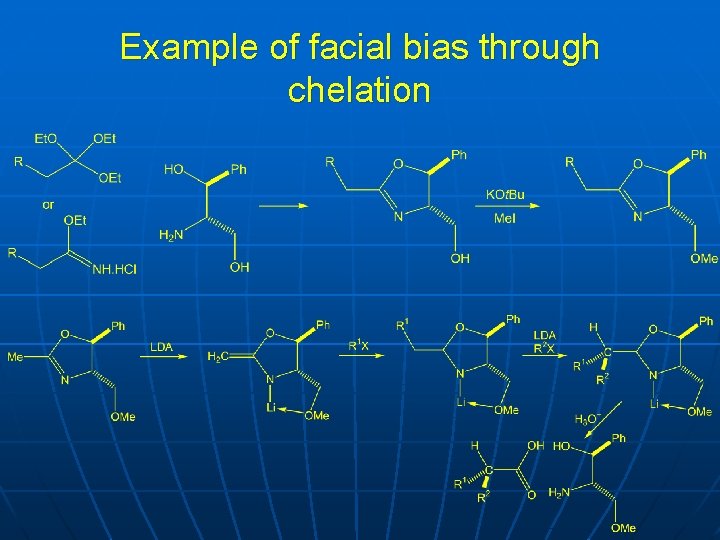
Example of facial bias through chelation
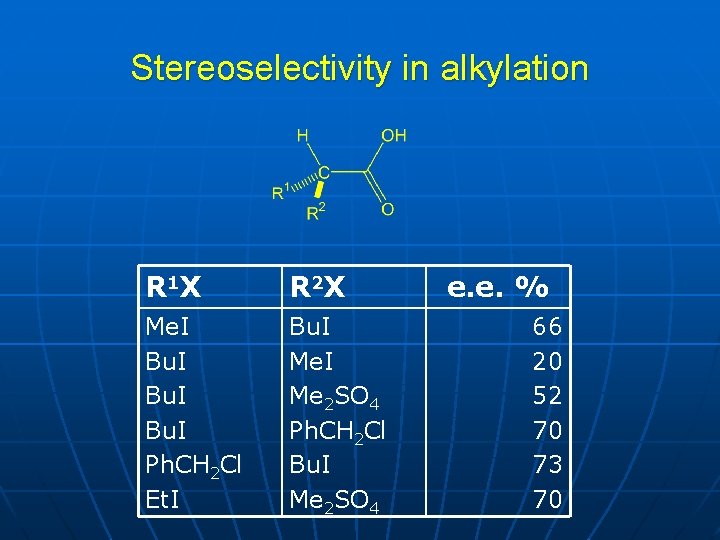
Stereoselectivity in alkylation R 1 X R 2 X Me. I Bu. I Ph. CH 2 Cl Et. I Bu. I Me 2 SO 4 Ph. CH 2 Cl Bu. I Me 2 SO 4 e. e. % 66 20 52 70 73 70
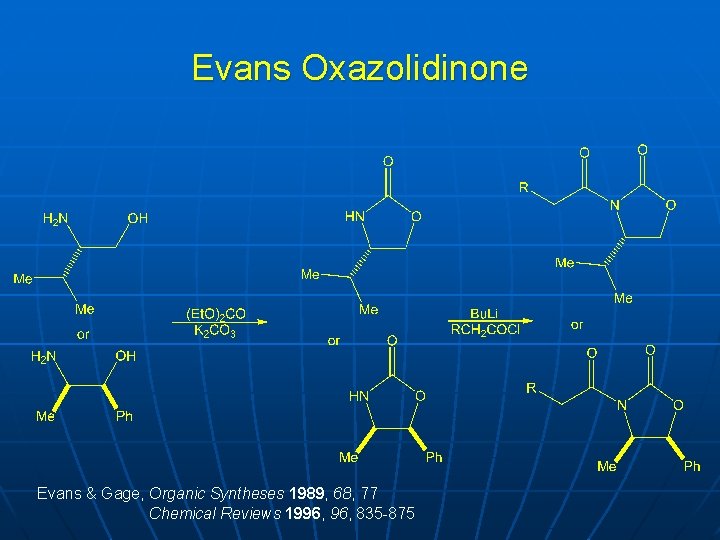
Evans Oxazolidinone Evans & Gage, Organic Syntheses 1989, 68, 77 Chemical Reviews 1996, 835 -875
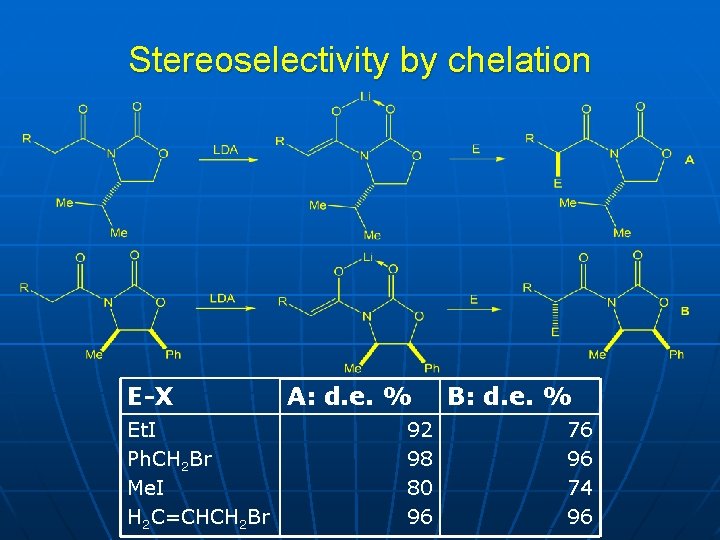
Stereoselectivity by chelation E-X Et. I Ph. CH 2 Br Me. I H 2 C=CHCH 2 Br A: d. e. % 92 98 80 96 B: d. e. % 76 96 74 96
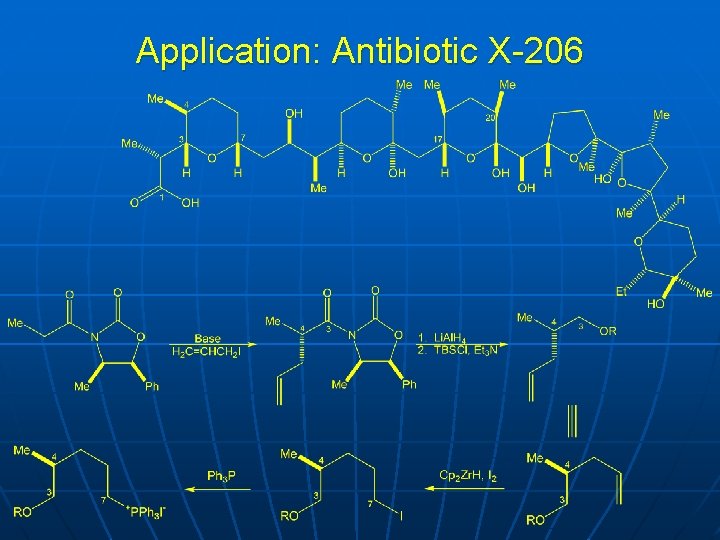
Application: Antibiotic X-206
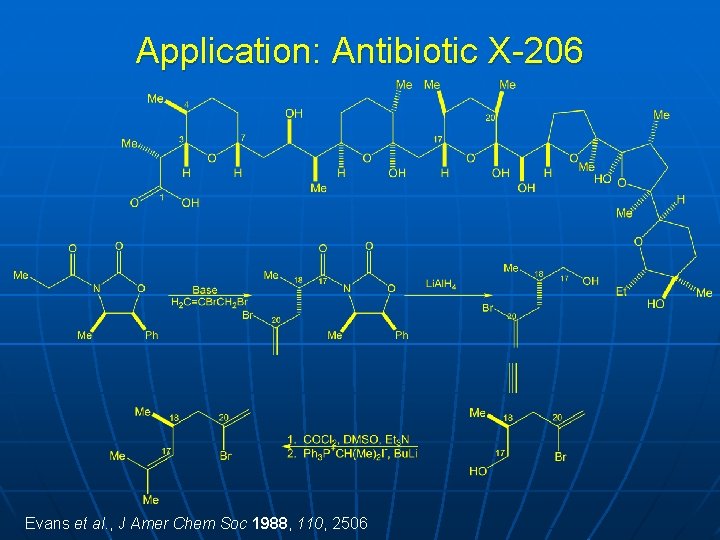
Application: Antibiotic X-206 Evans et al. , J Amer Chem Soc 1988, 110, 2506

Other electrophiles n n Hydroxylation Bromination Acylation
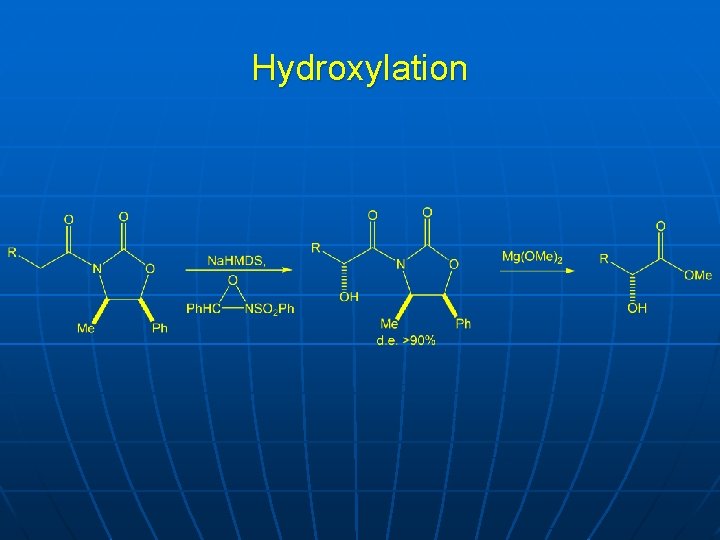
Hydroxylation
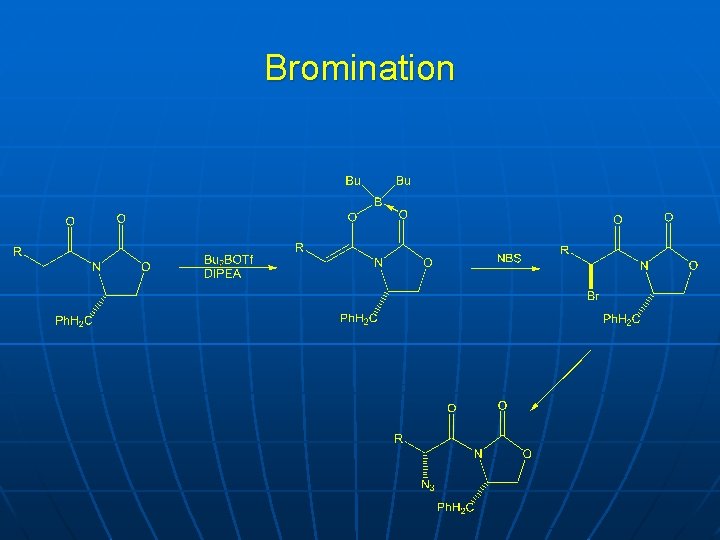
Bromination
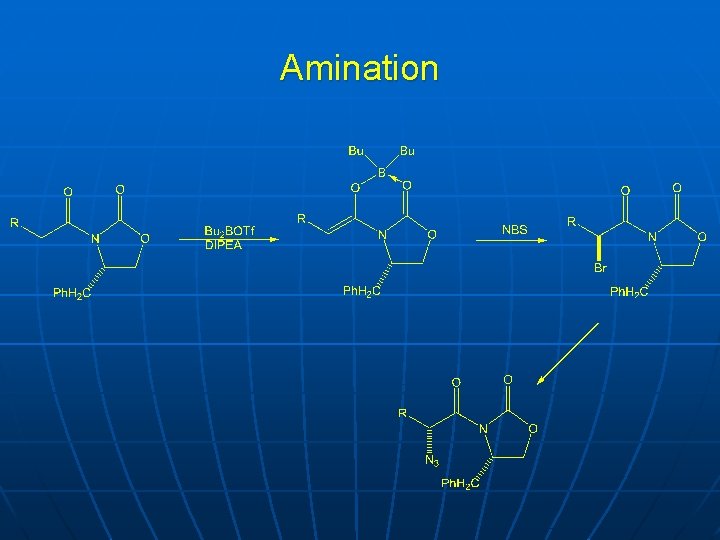
Amination
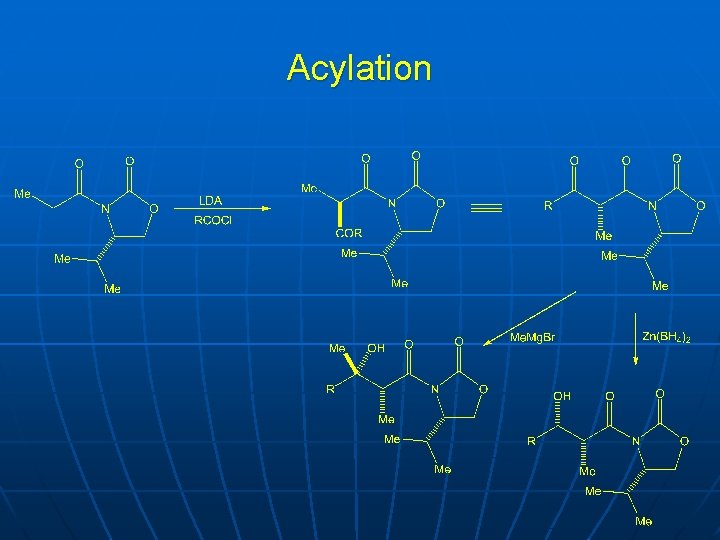
Acylation
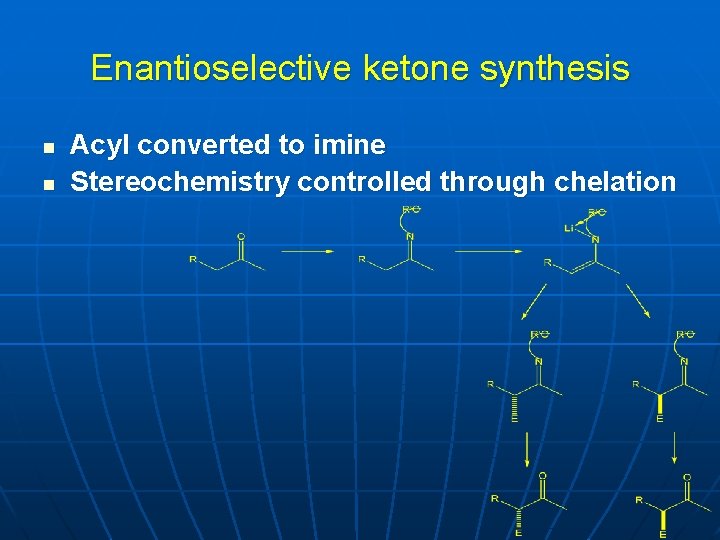
Enantioselective ketone synthesis n n Acyl converted to imine Stereochemistry controlled through chelation
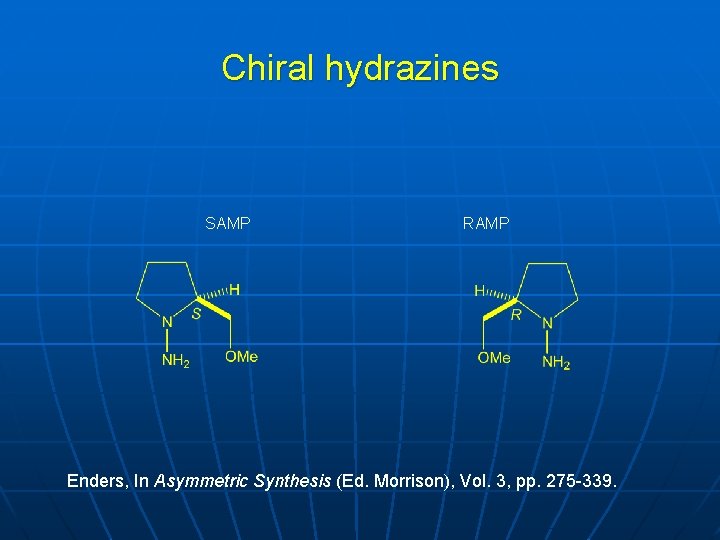
Chiral hydrazines SAMP RAMP Enders, In Asymmetric Synthesis (Ed. Morrison), Vol. 3, pp. 275 -339.
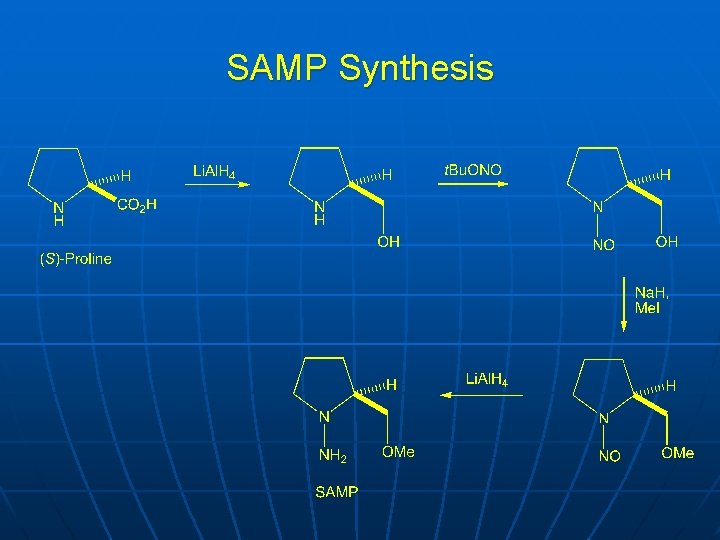
SAMP Synthesis
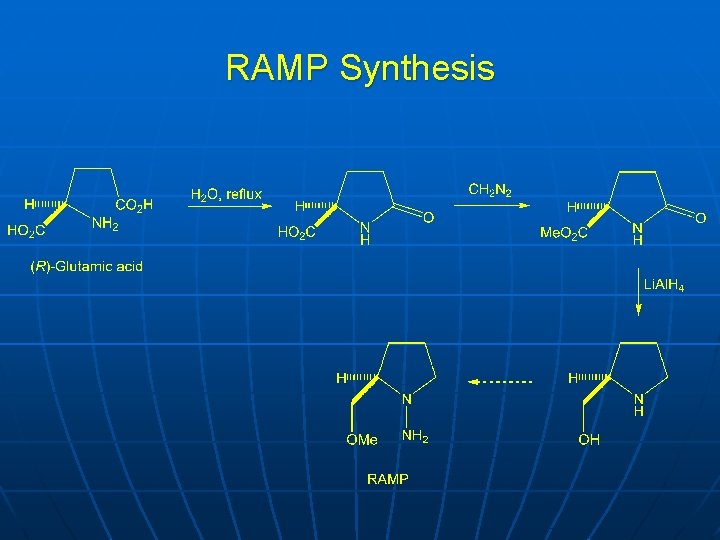
RAMP Synthesis
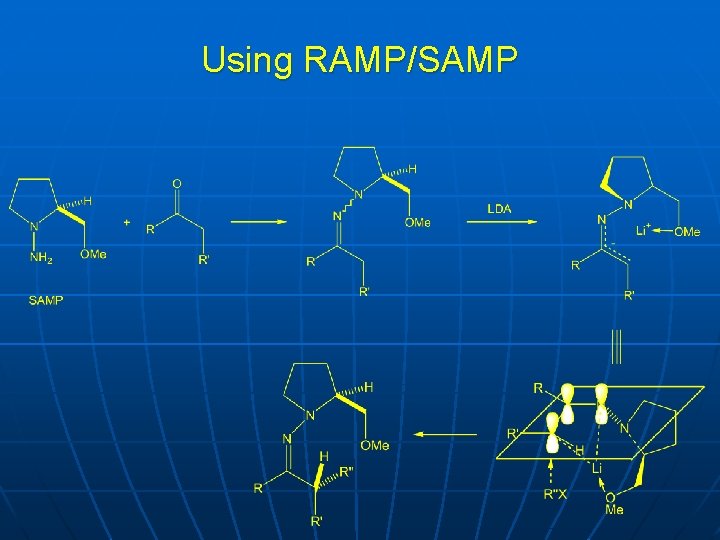
Using RAMP/SAMP
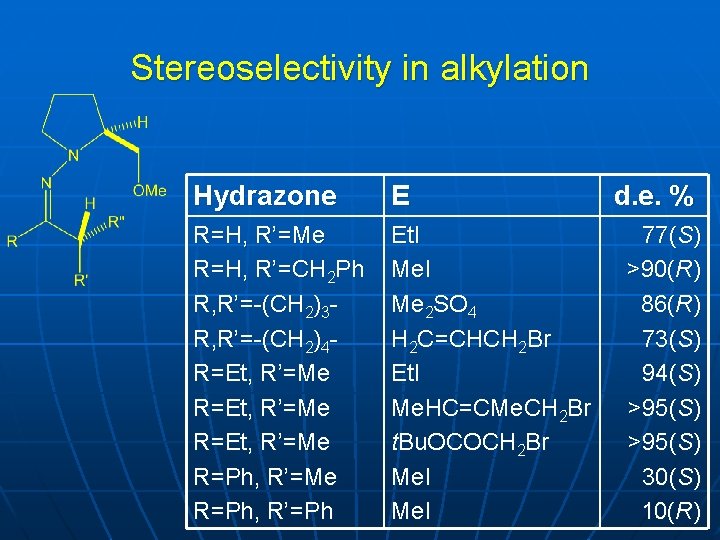
Stereoselectivity in alkylation Hydrazone E R=H, R’=Me R=H, R’=CH 2 Ph R, R’=-(CH 2)3 R, R’=-(CH 2)4 R=Et, R’=Me R=Ph, R’=Ph Et. I Me 2 SO 4 H 2 C=CHCH 2 Br Et. I Me. HC=CMe. CH 2 Br t. Bu. OCOCH 2 Br Me. I d. e. % 77(S) >90(R) 86(R) 73(S) 94(S) >95(S) 30(S) 10(R)

Rationalization n n Reaction with acyclic and cyclic ketones and aldehydes Methoxy group is important for high stereoselectivity Therefore chelation, as in oxazolines Removal by acid hydrolysis or ozonolysis
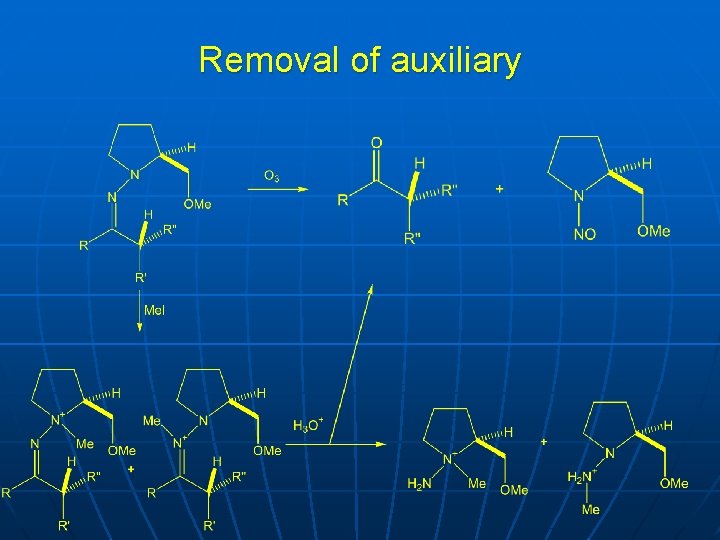
Removal of auxiliary

Application Leioburnum vittatum & L. calcar Enders & Baus, Liebigs Ann Chem 1983, 1439
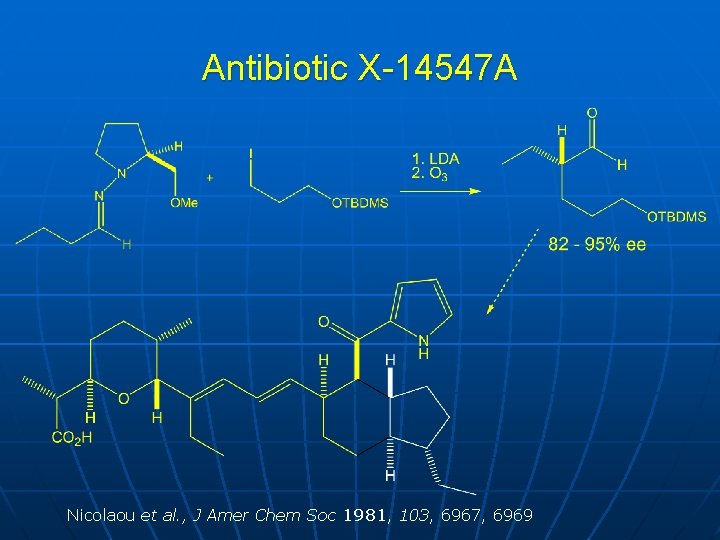
Antibiotic X-14547 A Nicolaou et al. , J Amer Chem Soc 1981, 103, 6967, 6969

Questions ?
- Slides: 36