22 1 Introduction Alpha Carbon Chemistry Enols and

22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • Why would a chemist want to form an enolate? • To form an enolate, a base must be used to remove the alpha protons • The appropriate base depends on how acidic the alpha protons are • What method do we have to quantify how acidic something is? Copyright 2012 John Wiley & Sons, Inc. 22 -1
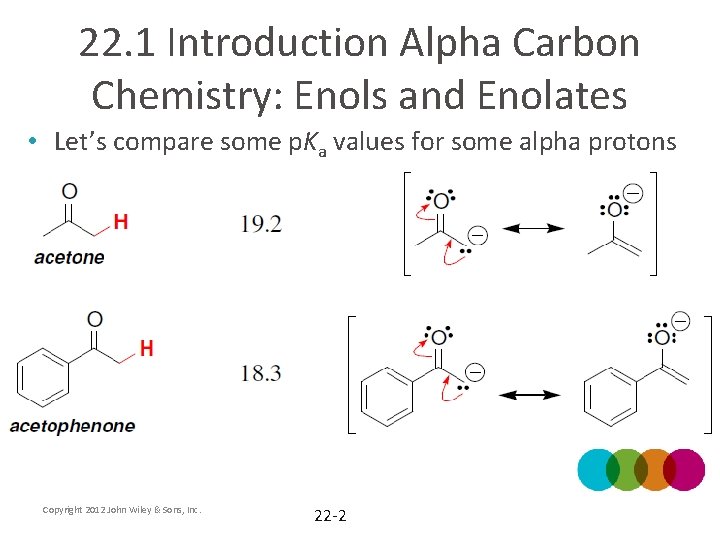
22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • Let’s compare some p. Ka values for some alpha protons Copyright 2012 John Wiley & Sons, Inc. 22 -2

22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • When p. Ka values are similar, both products and reactants are present in significant amounts • Which side will this equilibrium favor? Copyright 2012 John Wiley & Sons, Inc. 22 -3

22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • In this case, it is an advantage to have both enolate and aldehyde in solution so they can react with one another • Show the electrons might move in the reaction between the enolate and the aldehyde Copyright 2012 John Wiley & Sons, Inc. 22 -4
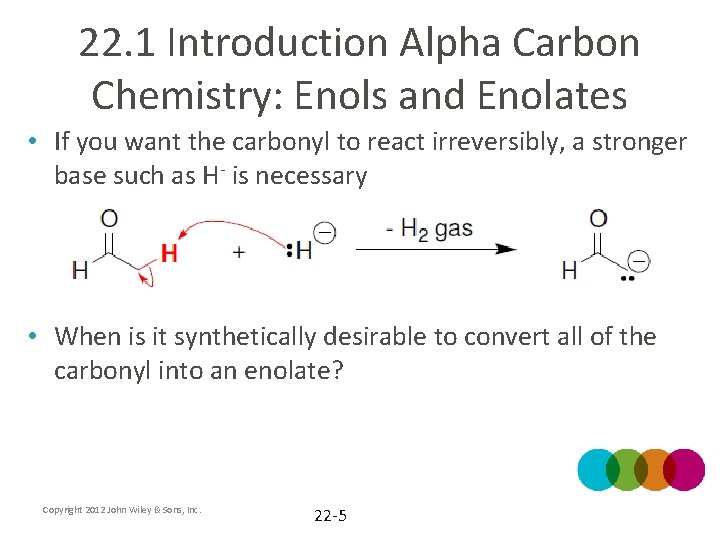
22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • If you want the carbonyl to react irreversibly, a stronger base such as H- is necessary • When is it synthetically desirable to convert all of the carbonyl into an enolate? Copyright 2012 John Wiley & Sons, Inc. 22 -5

22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • LDA is an even stronger base that is frequently used to promote irreversible enolate formation • Why is the reaction affectively irreversible? • LDA features two bulky isopropyl groups. Why would such a bulky base be desirable? Copyright 2012 John Wiley & Sons, Inc. 22 -6
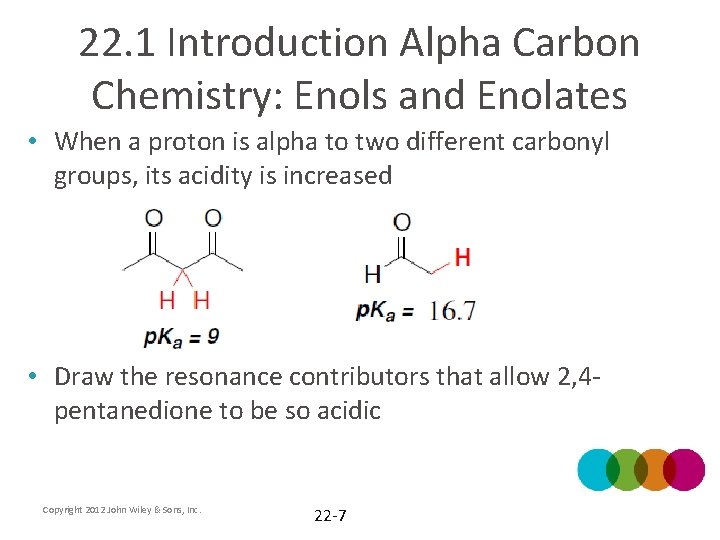
22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • When a proton is alpha to two different carbonyl groups, its acidity is increased • Draw the resonance contributors that allow 2, 4 pentanedione to be so acidic Copyright 2012 John Wiley & Sons, Inc. 22 -7
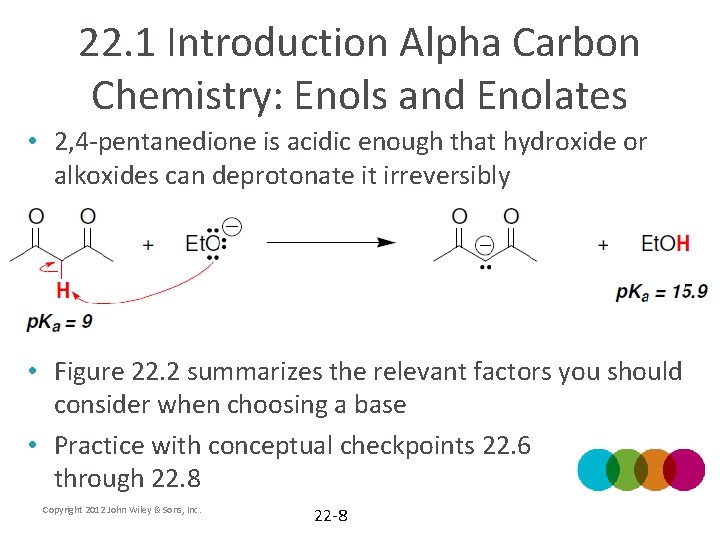
22. 1 Introduction Alpha Carbon Chemistry: Enols and Enolates • 2, 4 -pentanedione is acidic enough that hydroxide or alkoxides can deprotonate it irreversibly • Figure 22. 2 summarizes the relevant factors you should consider when choosing a base • Practice with conceptual checkpoints 22. 6 through 22. 8 Copyright 2012 John Wiley & Sons, Inc. 22 -8
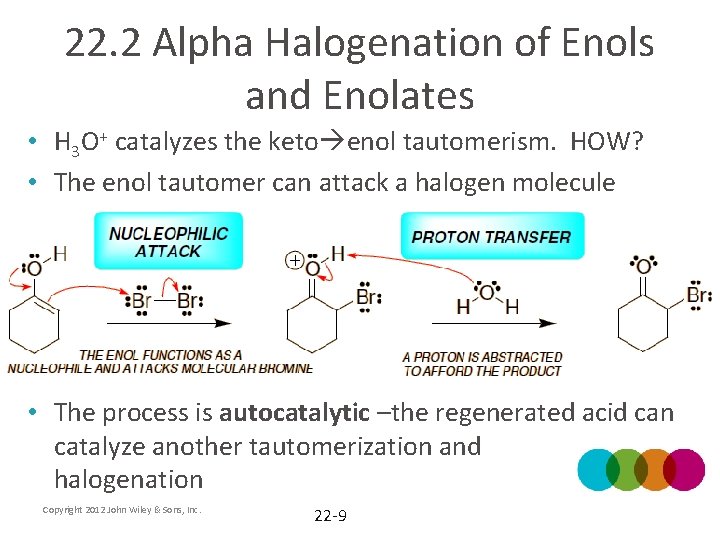
22. 2 Alpha Halogenation of Enols and Enolates • H 3 O+ catalyzes the keto enol tautomerism. HOW? • The enol tautomer can attack a halogen molecule • The process is autocatalytic –the regenerated acid can catalyze another tautomerization and halogenation Copyright 2012 John Wiley & Sons, Inc. 22 -9
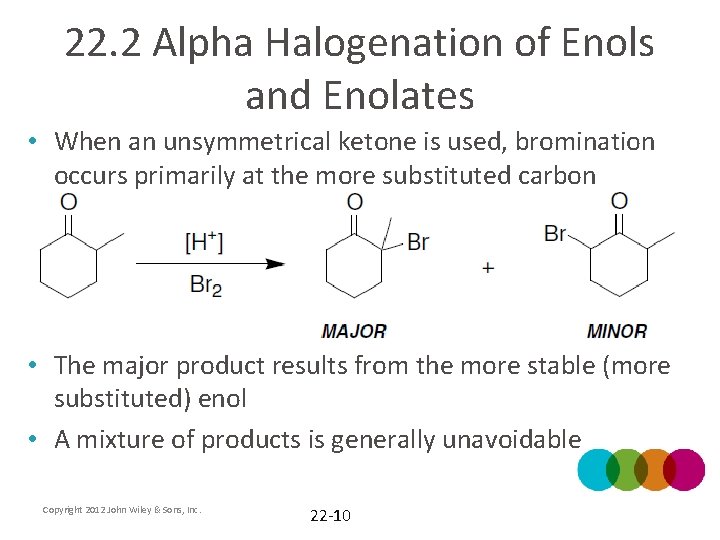
22. 2 Alpha Halogenation of Enols and Enolates • When an unsymmetrical ketone is used, bromination occurs primarily at the more substituted carbon • The major product results from the more stable (more substituted) enol • A mixture of products is generally unavoidable Copyright 2012 John Wiley & Sons, Inc. 22 -10
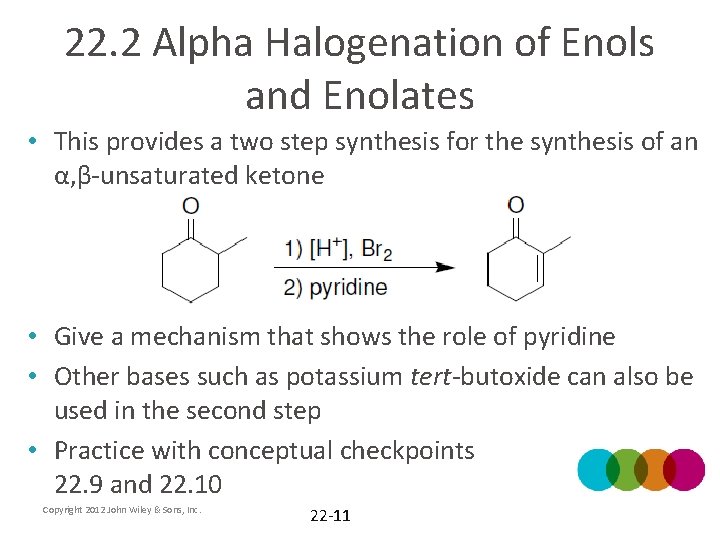
22. 2 Alpha Halogenation of Enols and Enolates • This provides a two step synthesis for the synthesis of an α, β-unsaturated ketone • Give a mechanism that shows the role of pyridine • Other bases such as potassium tert-butoxide can also be used in the second step • Practice with conceptual checkpoints 22. 9 and 22. 10 Copyright 2012 John Wiley & Sons, Inc. 22 -11

22. 2 Alpha Halogenation of Enols and Enolates • Alpha halogenation can also be achieved under basic conditions • The formation of the enolate is not favored, but the equilibrium is pushed forward by the second step • Will the presence of the α bromine make the remaining α proton more or less acidic? Copyright 2012 John Wiley & Sons, Inc. 22 -12
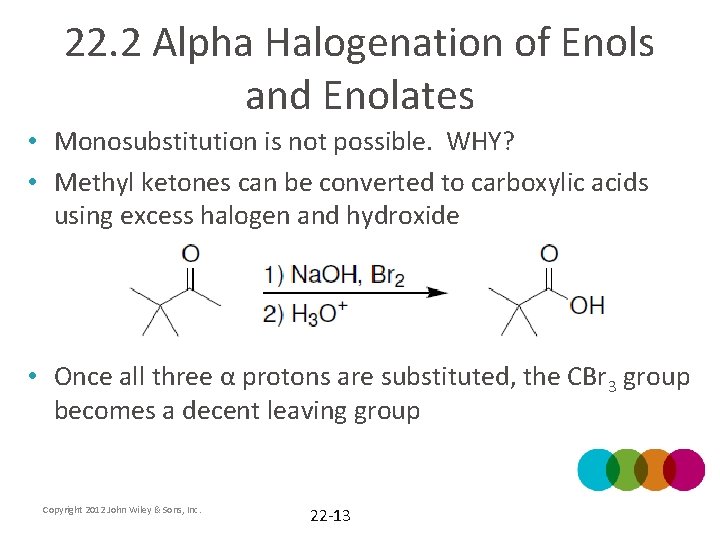
22. 2 Alpha Halogenation of Enols and Enolates • Monosubstitution is not possible. WHY? • Methyl ketones can be converted to carboxylic acids using excess halogen and hydroxide • Once all three α protons are substituted, the CBr 3 group becomes a decent leaving group Copyright 2012 John Wiley & Sons, Inc. 22 -13
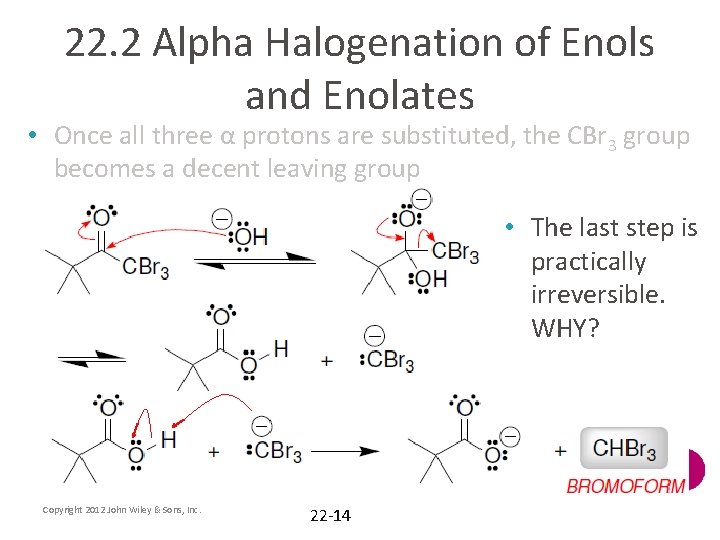
22. 2 Alpha Halogenation of Enols and Enolates • Once all three α protons are substituted, the CBr 3 group becomes a decent leaving group • The last step is practically irreversible. WHY? Copyright 2012 John Wiley & Sons, Inc. 22 -14

22. 2 Alpha Halogenation of Enols and Enolates • The carboxylate produced on the last slide can be protonated with H 3 O+ • The reaction works well with Cl 2, Br 2, and I 2, and it is known as the haloform reaction • The iodoform reaction may be used to test for methyl ketones, because iodoform can be observed as a yellow solid when it forms • Practice with conceptual checkpoints 22. 13 and 22. 14 Copyright 2012 John Wiley & Sons, Inc. 22 -15
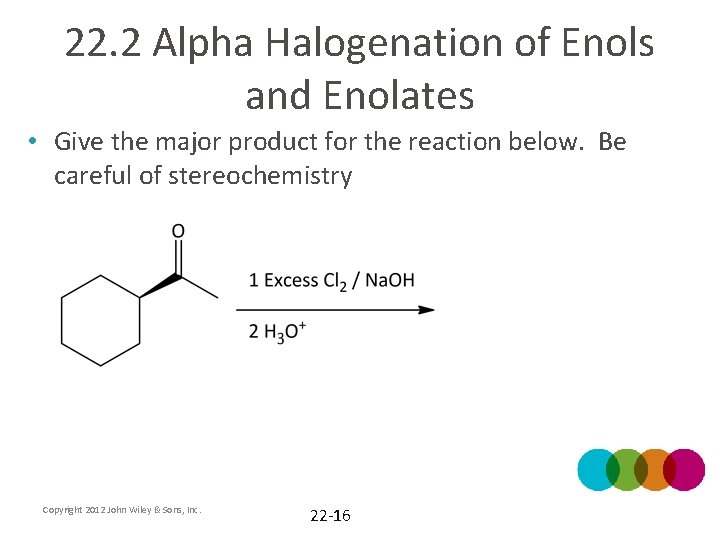
22. 2 Alpha Halogenation of Enols and Enolates • Give the major product for the reaction below. Be careful of stereochemistry Copyright 2012 John Wiley & Sons, Inc. 22 -16

22. 3 Aldol Reactions • Recall that when an aldehyde is treated with hydroxide (or alkoxide), an equilibrium forms where significant amounts of both enolate and aldehyde are present • If the enolate attacks the aldehyde, an aldol reaction occurs • The product features both aldehyde and alcohol groups • Note the location of the –OH group on the beta carbon Copyright 2012 John Wiley & Sons, Inc. 22 -17
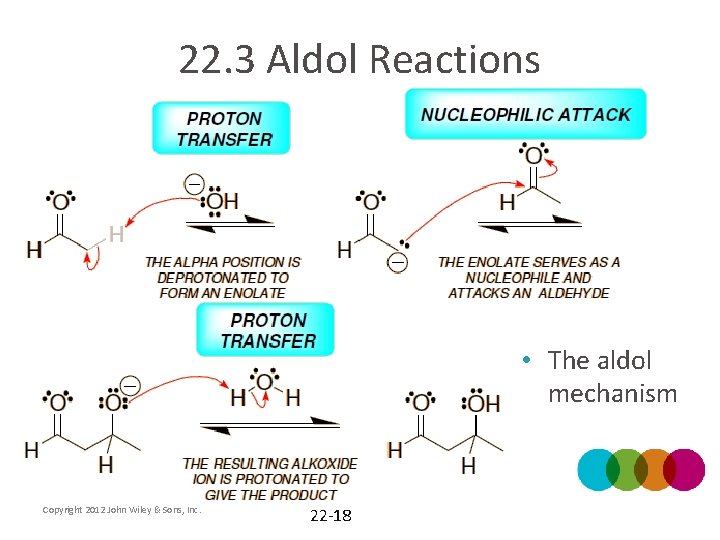
22. 3 Aldol Reactions • The aldol mechanism Copyright 2012 John Wiley & Sons, Inc. 22 -18
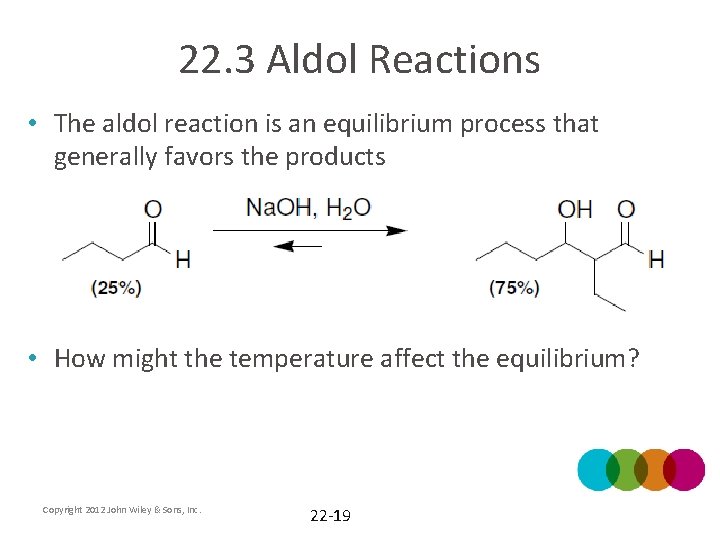
22. 3 Aldol Reactions • The aldol reaction is an equilibrium process that generally favors the products • How might the temperature affect the equilibrium? Copyright 2012 John Wiley & Sons, Inc. 22 -19
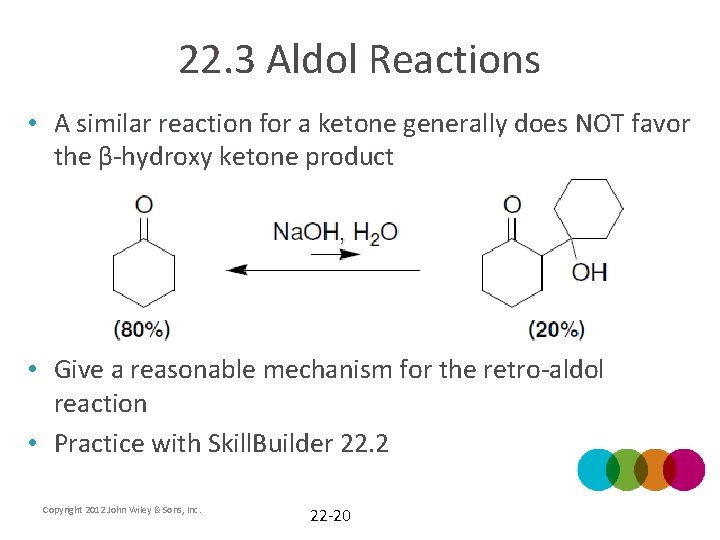
22. 3 Aldol Reactions • A similar reaction for a ketone generally does NOT favor the β-hydroxy ketone product • Give a reasonable mechanism for the retro-aldol reaction • Practice with Skill. Builder 22. 2 Copyright 2012 John Wiley & Sons, Inc. 22 -20
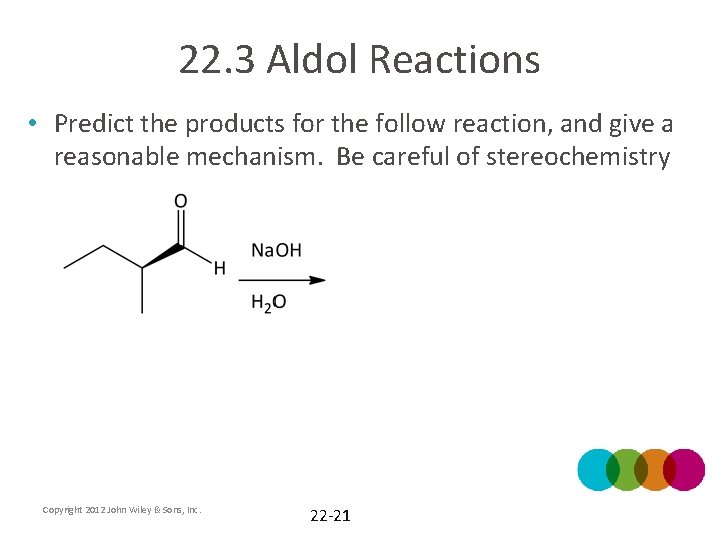
22. 3 Aldol Reactions • Predict the products for the follow reaction, and give a reasonable mechanism. Be careful of stereochemistry Copyright 2012 John Wiley & Sons, Inc. 22 -21

22. 3 Aldol Reactions • When an aldol product is heated under acidic or basic conditions, an α, β-unsaturated carbonyl forms • Such a process is called an aldol condensation, because water is given off • The elimination reaction above is an equilibrium, which generally favors the products • WHY? Consider enthalpy and entropy Copyright 2012 John Wiley & Sons, Inc. 22 -22

22. 3 Aldol Reactions • The elimination of water can be promoted under acidic or under basic conditions • Give a reasonable mechanism for each Copyright 2012 John Wiley & Sons, Inc. 22 -23
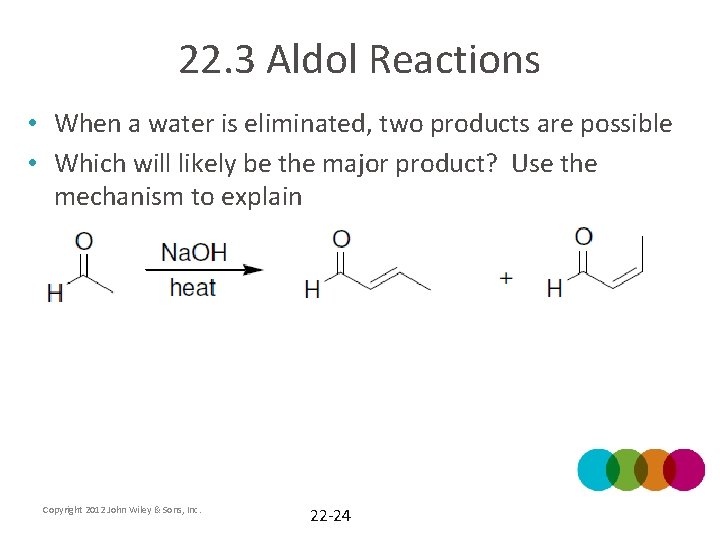
22. 3 Aldol Reactions • When a water is eliminated, two products are possible • Which will likely be the major product? Use the mechanism to explain Copyright 2012 John Wiley & Sons, Inc. 22 -24
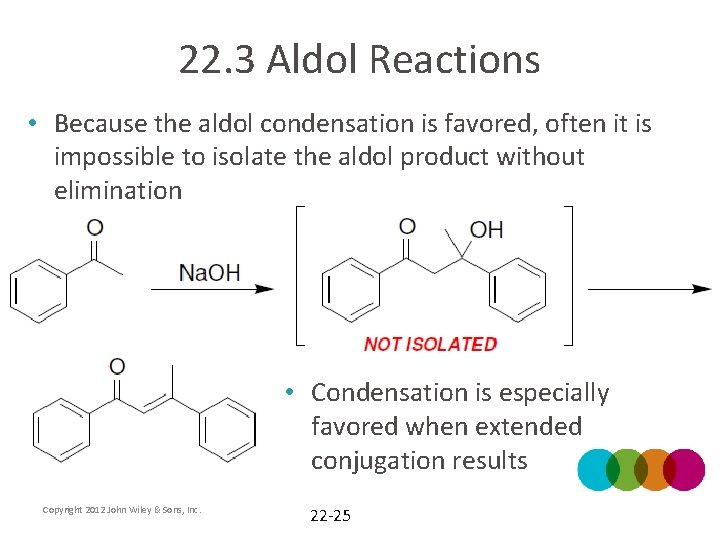
22. 3 Aldol Reactions • Because the aldol condensation is favored, often it is impossible to isolate the aldol product without elimination • Condensation is especially favored when extended conjugation results Copyright 2012 John Wiley & Sons, Inc. 22 -25
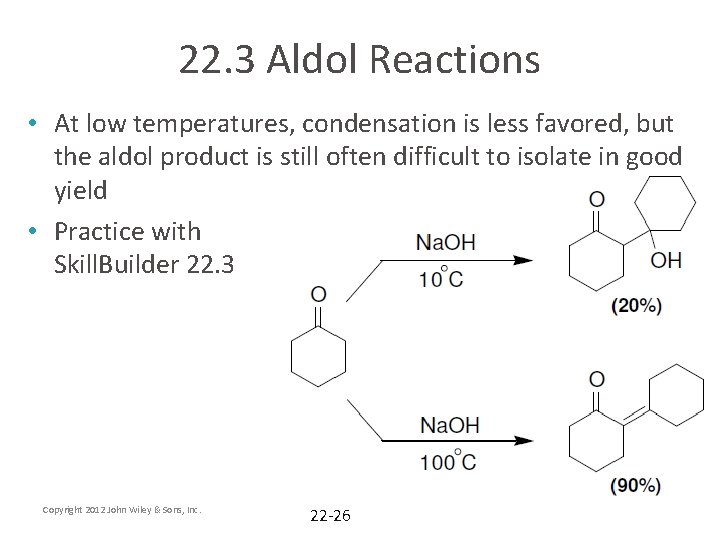
22. 3 Aldol Reactions • At low temperatures, condensation is less favored, but the aldol product is still often difficult to isolate in good yield • Practice with Skill. Builder 22. 3 Copyright 2012 John Wiley & Sons, Inc. 22 -26
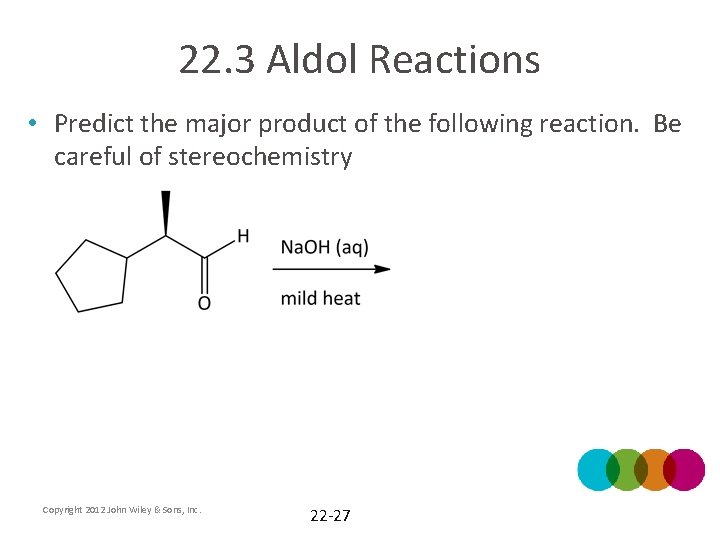
22. 3 Aldol Reactions • Predict the major product of the following reaction. Be careful of stereochemistry Copyright 2012 John Wiley & Sons, Inc. 22 -27

22. 3 Aldol Reactions • Substrates can react in a crossed aldol or mixed aldol reaction. Predict the 4 possible products in the reaction below • Such a complicated mixture of products is not very synthetically practical. WHY? Copyright 2012 John Wiley & Sons, Inc. 22 -28
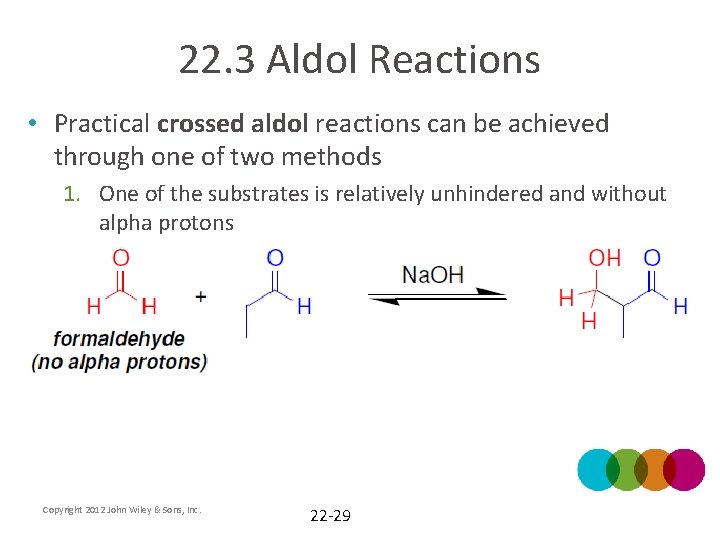
22. 3 Aldol Reactions • Practical crossed aldol reactions can be achieved through one of two methods 1. One of the substrates is relatively unhindered and without alpha protons Copyright 2012 John Wiley & Sons, Inc. 22 -29
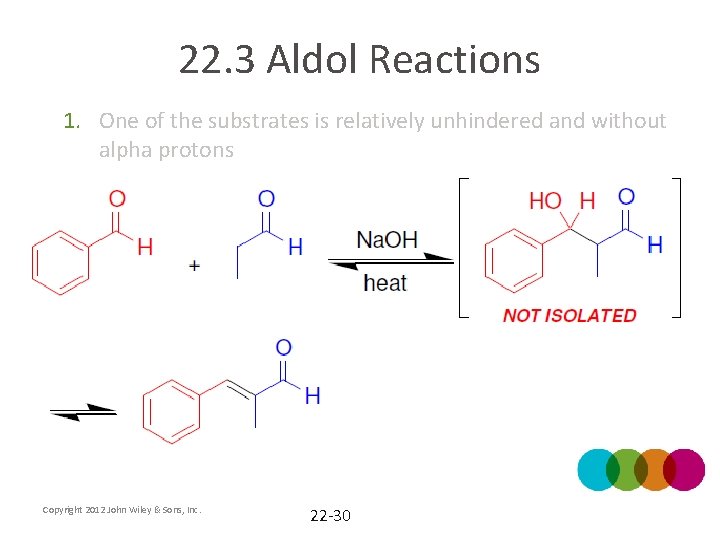
22. 3 Aldol Reactions 1. One of the substrates is relatively unhindered and without alpha protons Copyright 2012 John Wiley & Sons, Inc. 22 -30
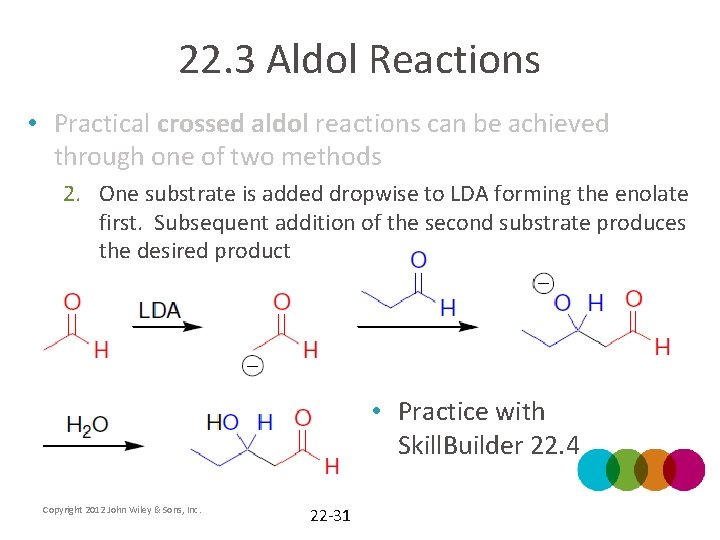
22. 3 Aldol Reactions • Practical crossed aldol reactions can be achieved through one of two methods 2. One substrate is added dropwise to LDA forming the enolate first. Subsequent addition of the second substrate produces the desired product • Practice with Skill. Builder 22. 4 Copyright 2012 John Wiley & Sons, Inc. 22 -31
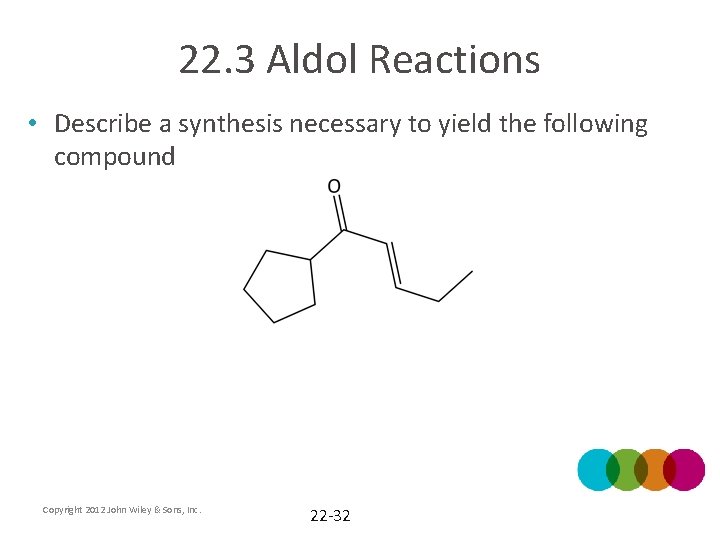
22. 3 Aldol Reactions • Describe a synthesis necessary to yield the following compound Copyright 2012 John Wiley & Sons, Inc. 22 -32
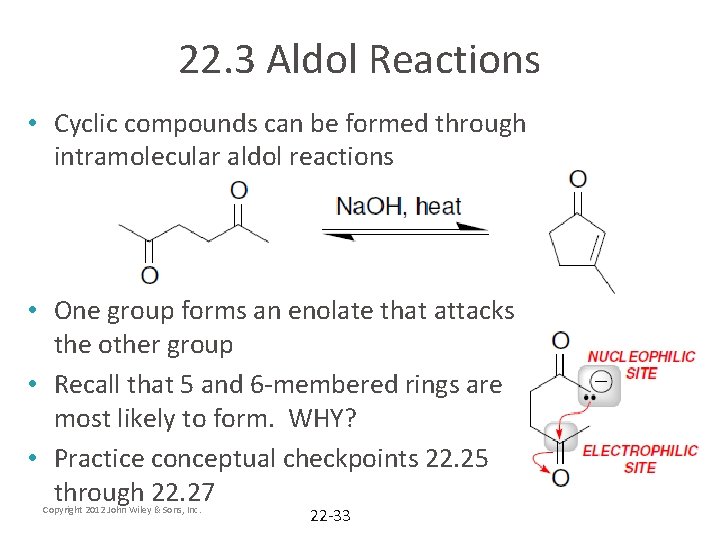
22. 3 Aldol Reactions • Cyclic compounds can be formed through intramolecular aldol reactions • One group forms an enolate that attacks the other group • Recall that 5 and 6 -membered rings are most likely to form. WHY? • Practice conceptual checkpoints 22. 25 through 22. 27 Copyright 2012 John Wiley & Sons, Inc. 22 -33

Study Guide for Sections 22. 1 -22. 3 DAY 24, Terms to know: Sections 22. 1 -22. 3 α, β-unsaturated ketone, LDA, haloform, aldol reaction, retro-aldol reaction, aldol condensation, crossed or mixed aldol DAY 24, Specific outcomes and skills that may be tested on exam 4: Sections 22. 1 -22. 3 • Based on given p. Ka values, be able to predict which bases will give enolate products in small quantity in equilibrium versus which will give nearly complete enolate production • Be able to predict products and give a complete mechanism for the two step process where ketones are converted into α, β-unsaturated ketones • Be able to describe ideal reaction conditions for the formation of a α, β-unsaturated ketone from a ketone • Be able to predict products and give a complete mechanism for the haloform reaction • Given a reactant and product, be able to describe ideal reaction conditions for the haloform reaction • Be able to predict products and give a complete mechanism for the aldol and retro-aldo reaction including subsequent aldol condensation if appropriate • Given a reactant and product, be able to describe ideal reaction conditions and reagents for the aldol reaction including subsequent aldol condensation if appropriate

Extra Practice Problems for Sections 22. 1 -22. 3 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 22. 6 22. 7 22. 58 22. 59 22. 61 22. 63 22. 65 22. 66 22. 9 22. 10 22. 13 22. 14 22. 15 22. 17 22. 18 22. 19 22. 20 22. 21 22. 23 22. 25 22. 26 22. 67 22. 69 22. 70 22. 71 22. 73

Prep for Day 25 Must Watch videos: https: //www. youtube. com/watch? v=4 bg. Xy. JE-o. NA (aldol, FLC) start at 15 minute mark https: //www. youtube. com/watch? v=y. E 3 e. A 50 r 3 m. Q (alpha alkylation, FLC) https: //www. youtube. com/watch? v=s. R_8 g. Nhds. Z 0 (conjugate addition, FLC) Other helpful videos: https: //www. youtube. com/watch? v=oa. Bz 6 bd. DHUU (Claisen, UC-Irvine) https: //www. youtube. com/watch? v=45 j. L 2 huo. L_I (Claisen and conjugate addition, UC-Irvine) https: //www. youtube. com/watch? v=CBLx 2 vch. Ui. A and https: //www. youtube. com/watch? v=Z 31 v_1 h. Z 42 k (conjugate addition, Dave) https: //www. youtube. com/watch? v=R 3 i 2 E 77 sf. Io (enolate chemistry, UC-Irvine) Read Sections 22. 4 -22. 7
- Slides: 36