Enols and Enolates a Substitutions and Condensations of
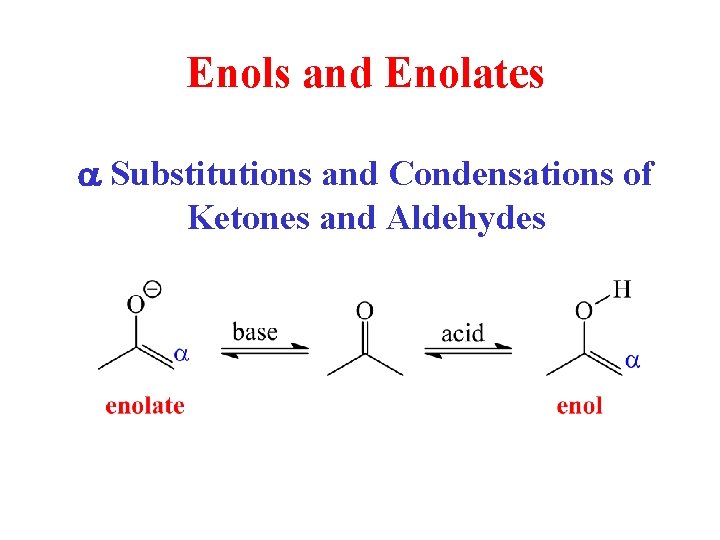
Enols and Enolates a Substitutions and Condensations of Ketones and Aldehydes
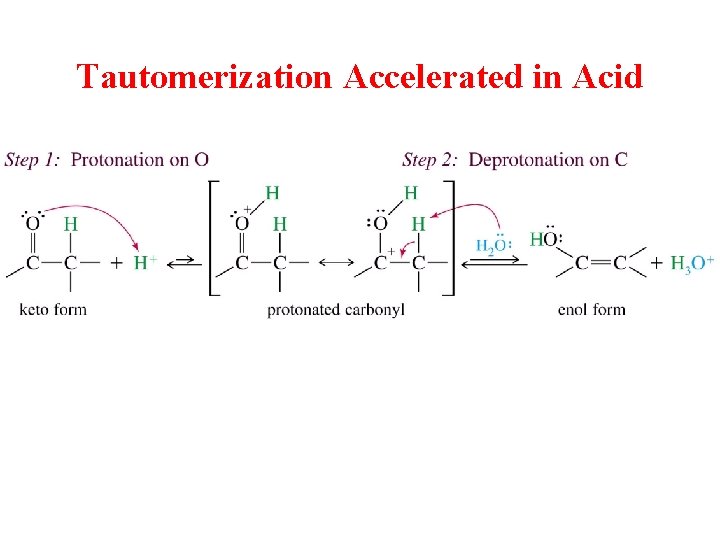
Tautomerization Accelerated in Acid
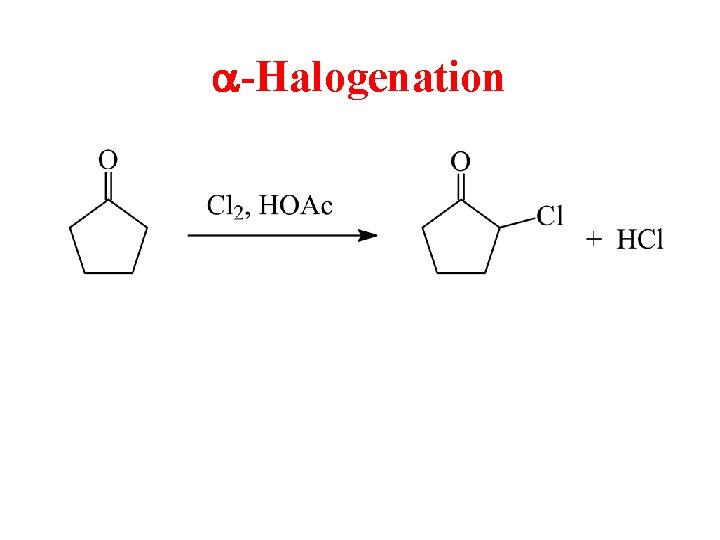
a-Halogenation
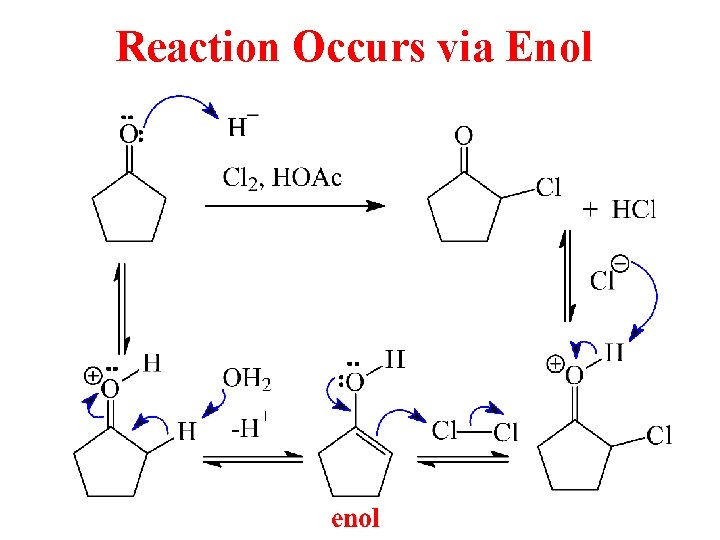
Reaction Occurs via Enol
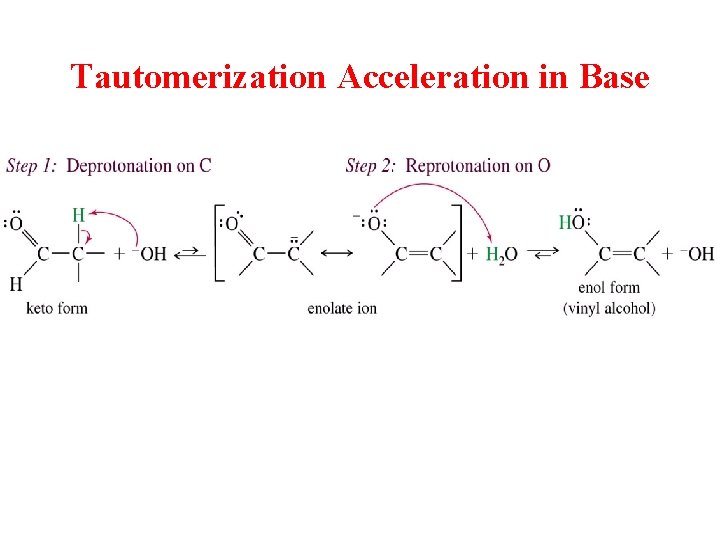
Tautomerization Acceleration in Base
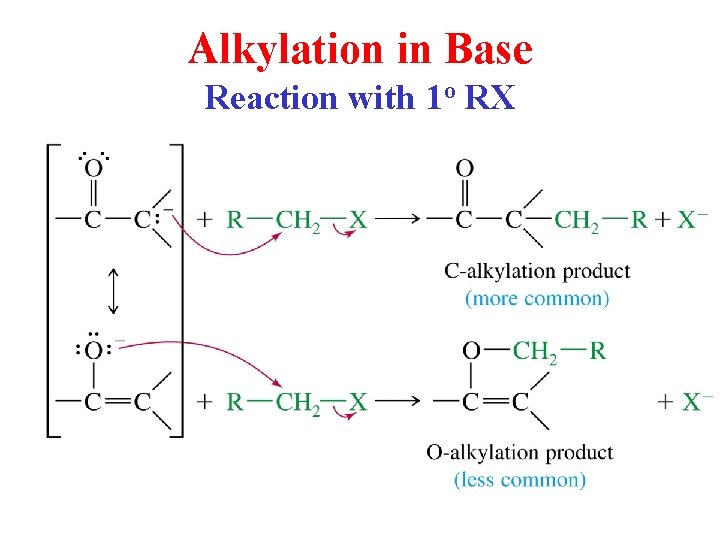
Alkylation in Base Reaction with 1 o RX
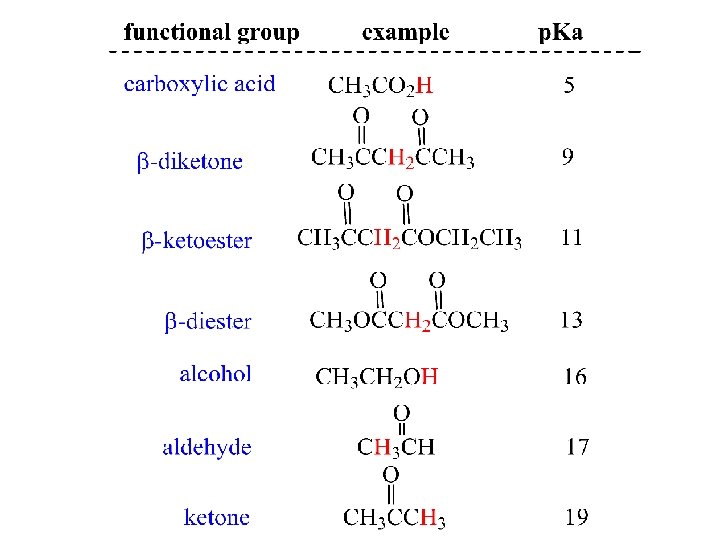
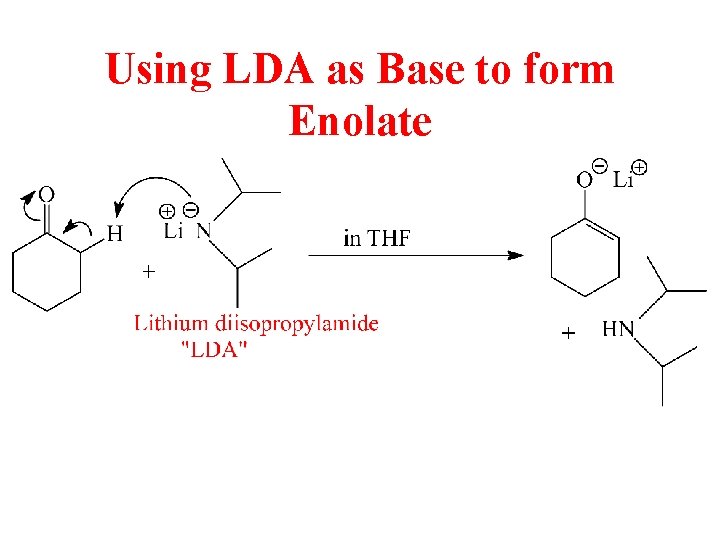
Using LDA as Base to form Enolate

Alkylation a to C=O C-alkylation preferred
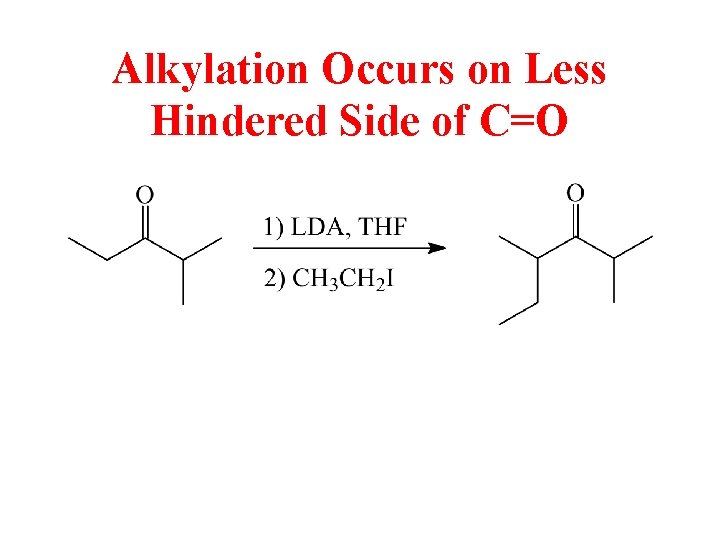
Alkylation Occurs on Less Hindered Side of C=O
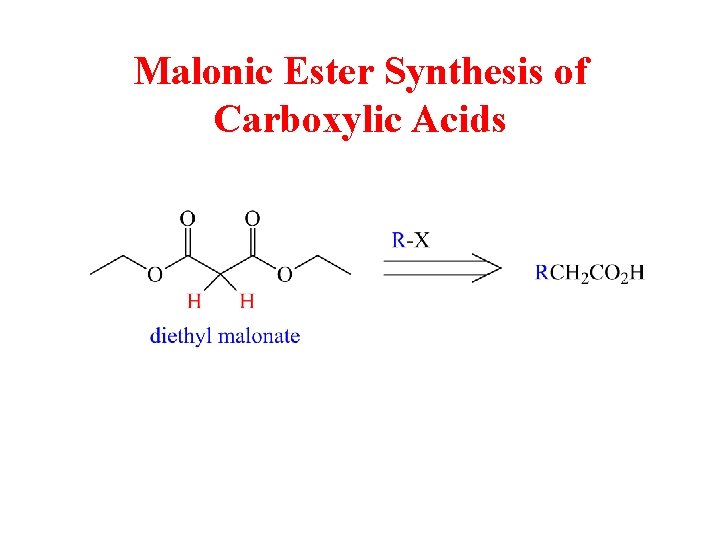
Malonic Ester Synthesis of Carboxylic Acids
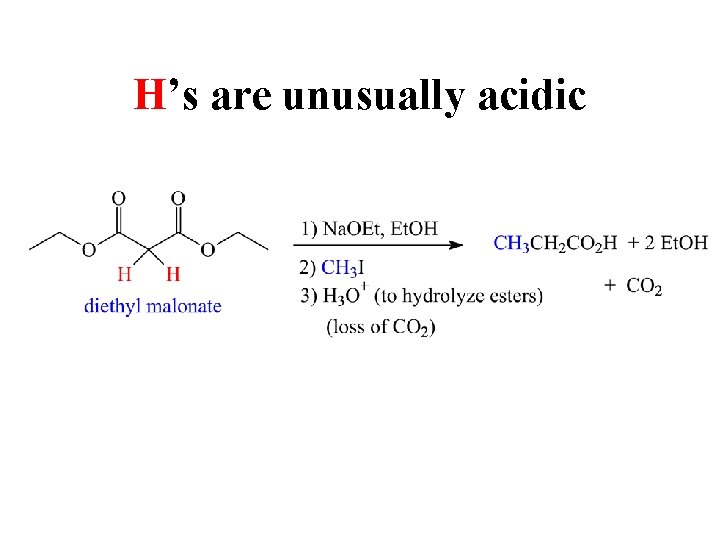
H’s are unusually acidic
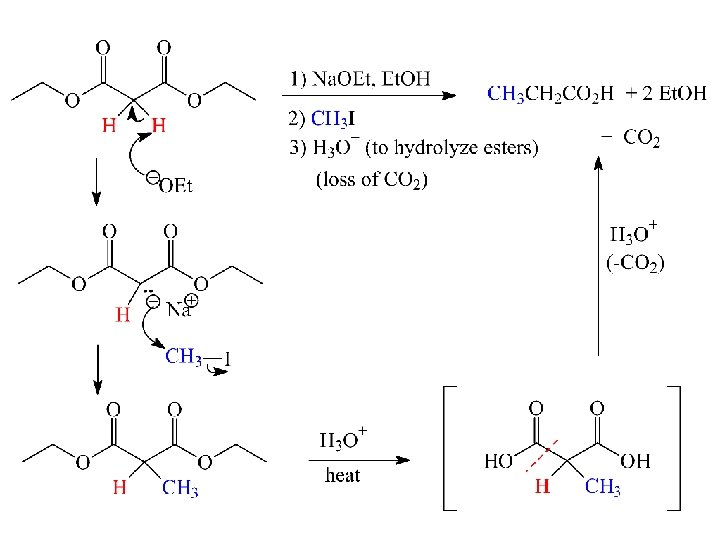
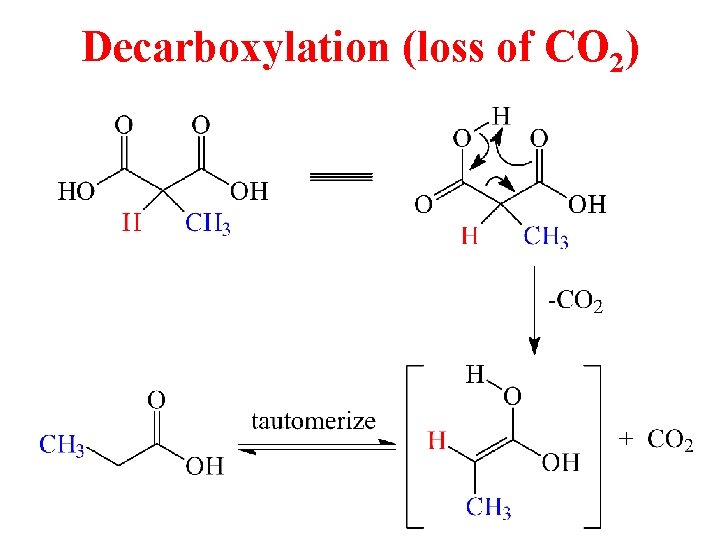
Decarboxylation (loss of CO 2)
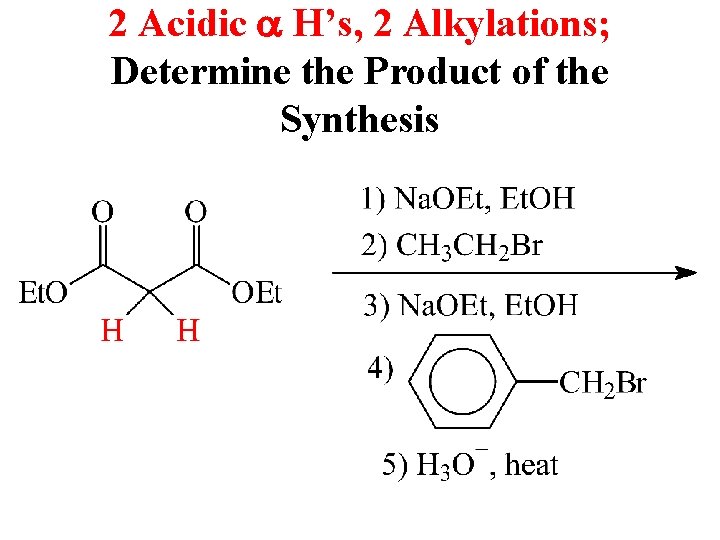
2 Acidic a H’s, 2 Alkylations; Determine the Product of the Synthesis
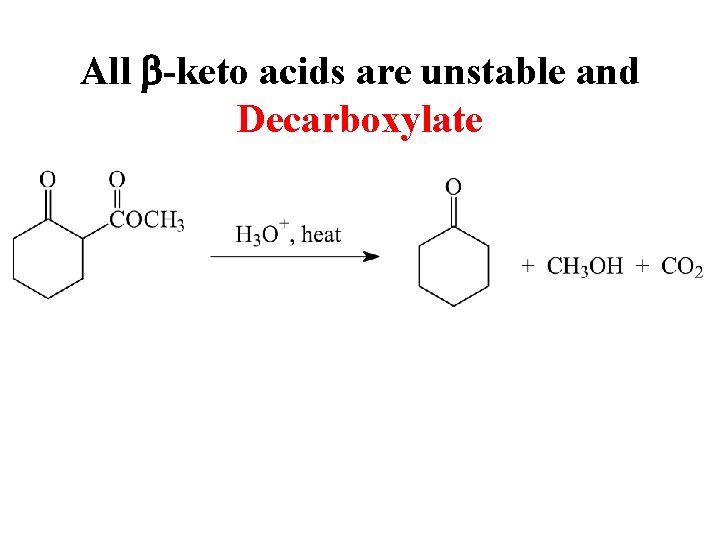
All b-keto acids are unstable and Decarboxylate
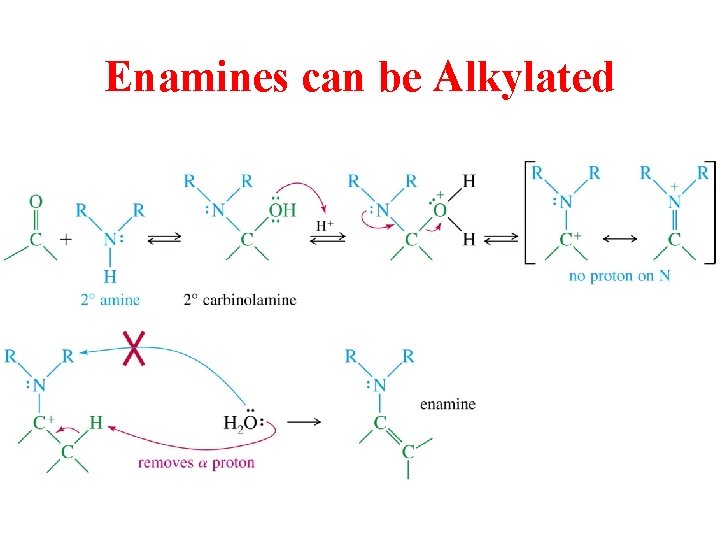
Enamines can be Alkylated
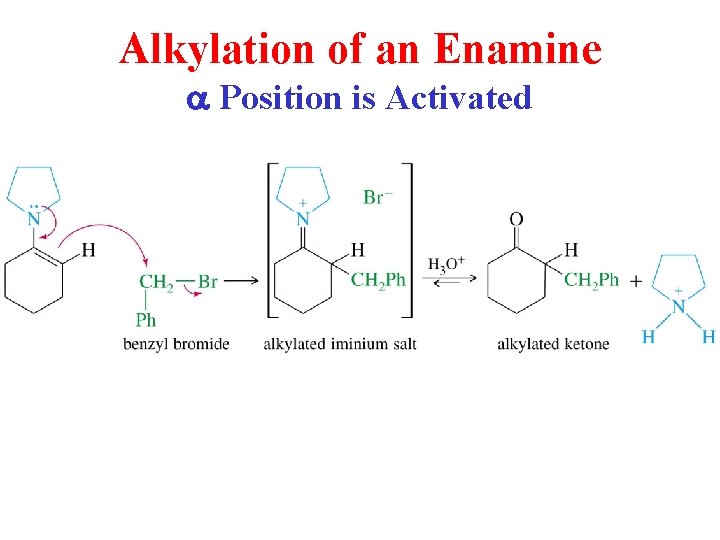
Alkylation of an Enamine a Position is Activated

Complements LDA Reaction

LDA vs. Enamine Reaction
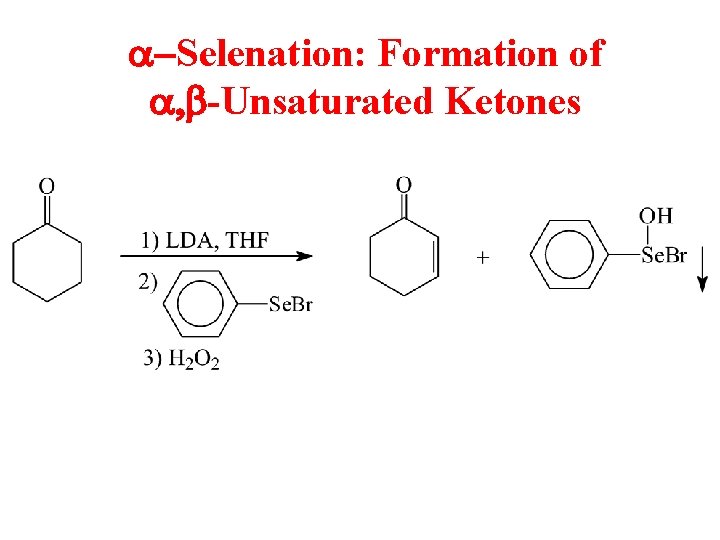
a-Selenation: Formation of a, b-Unsaturated Ketones

Mechanism
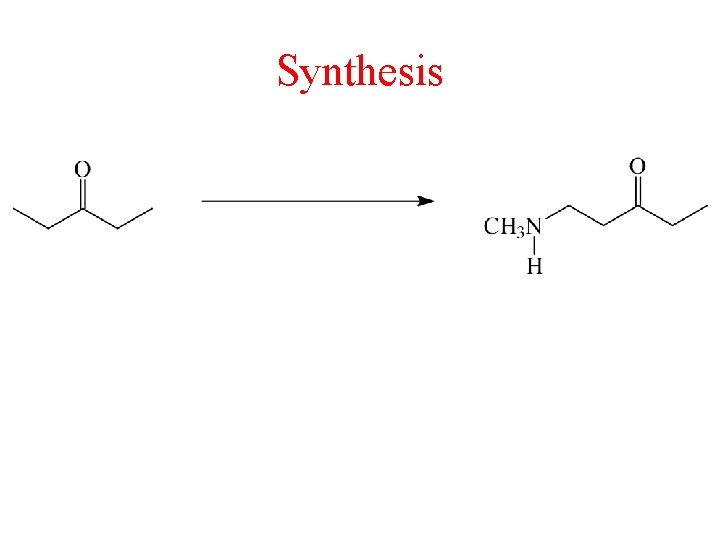
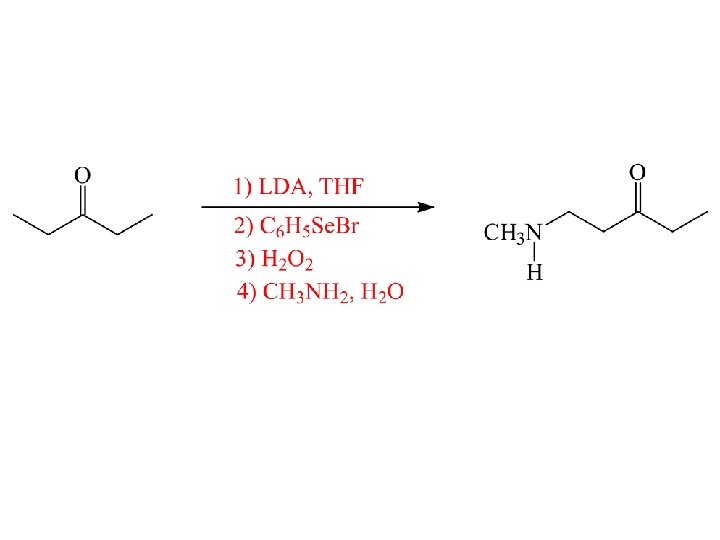
Synthesis
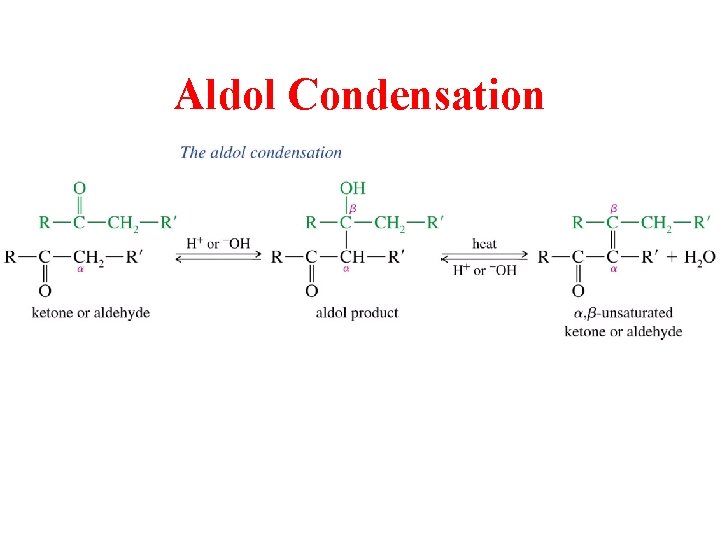
Aldol Condensation
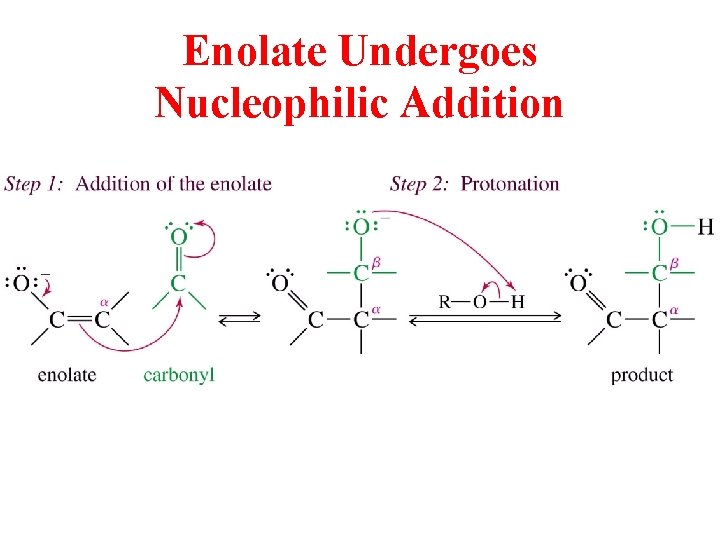
Enolate Undergoes Nucleophilic Addition
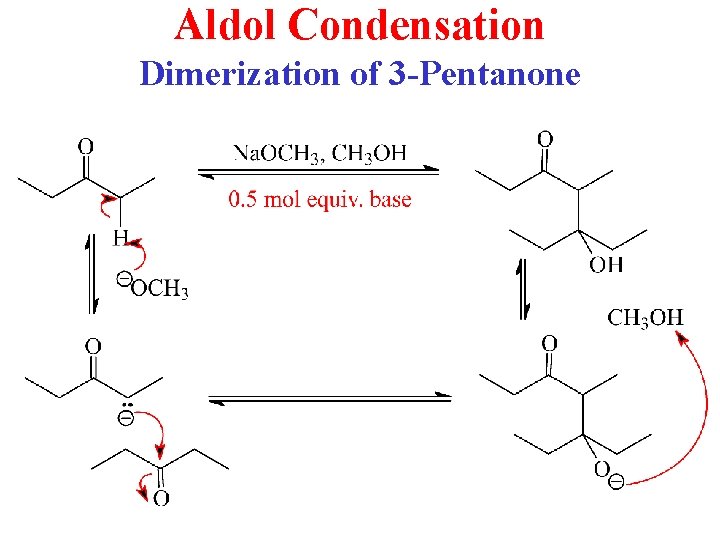
Aldol Condensation Dimerization of 3 -Pentanone
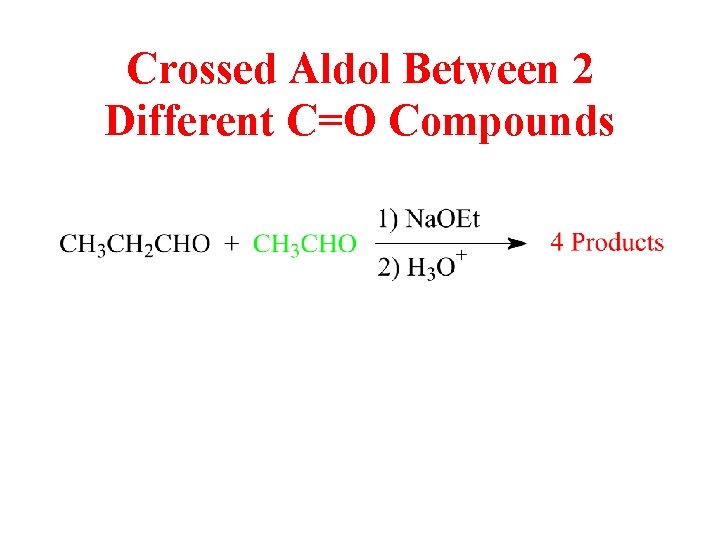
Crossed Aldol Between 2 Different C=O Compounds
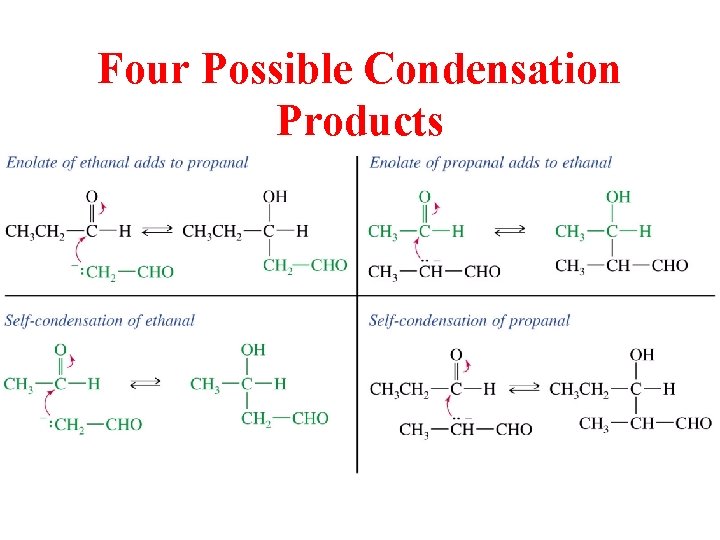
Four Possible Condensation Products
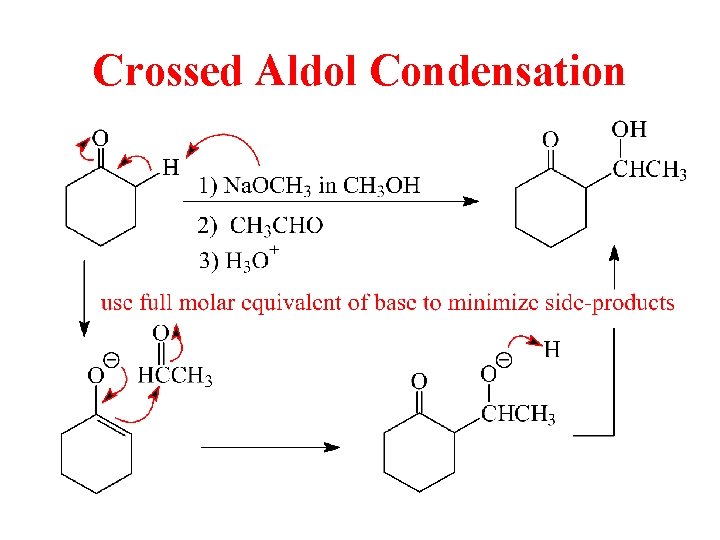
Crossed Aldol Condensation

Dehydration of Aldol Products
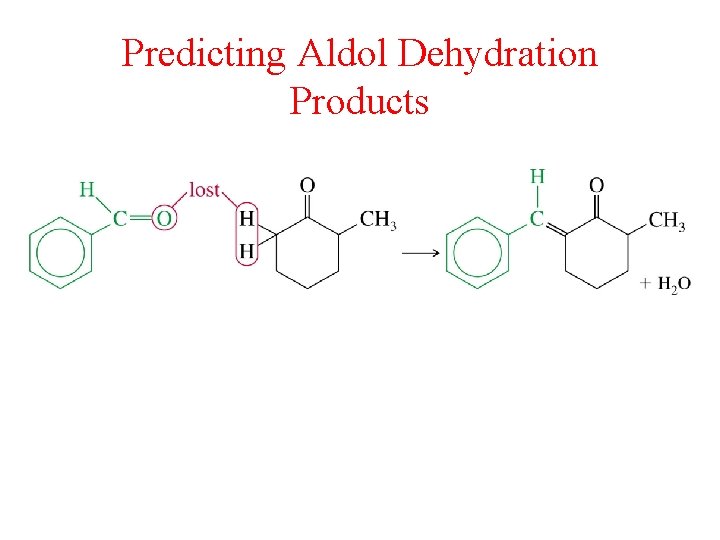
Predicting Aldol Dehydration Products
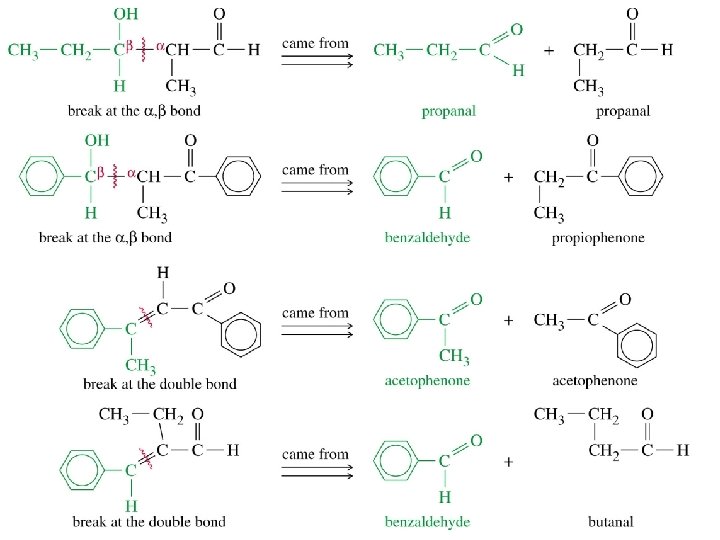
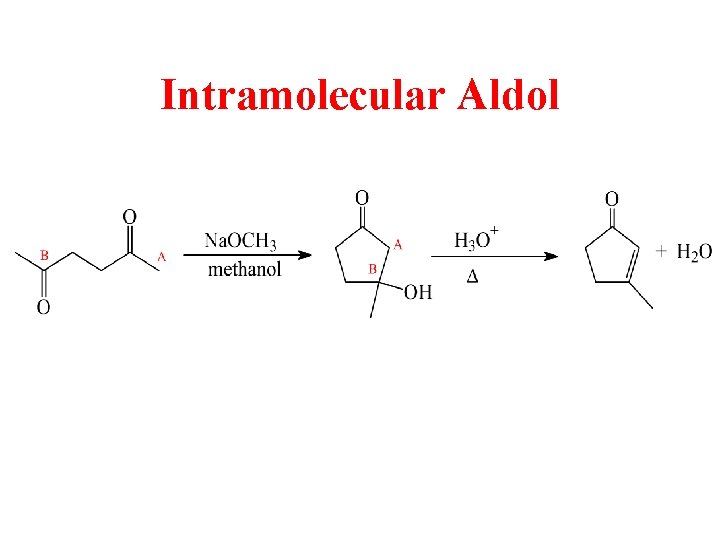
Intramolecular Aldol
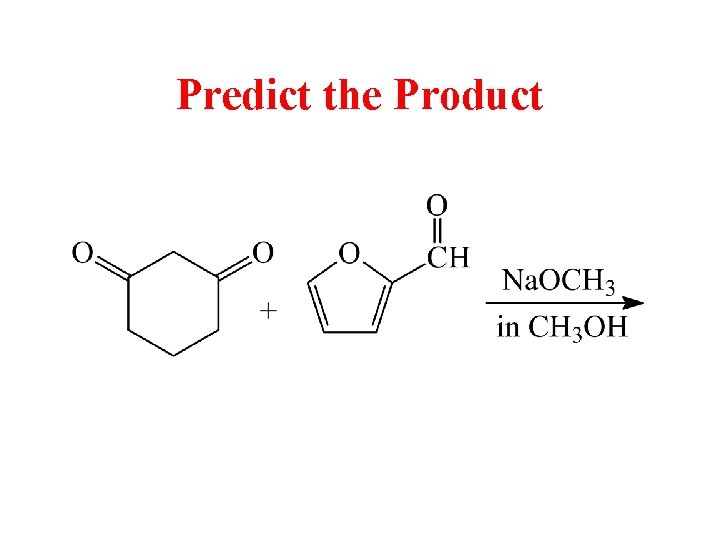
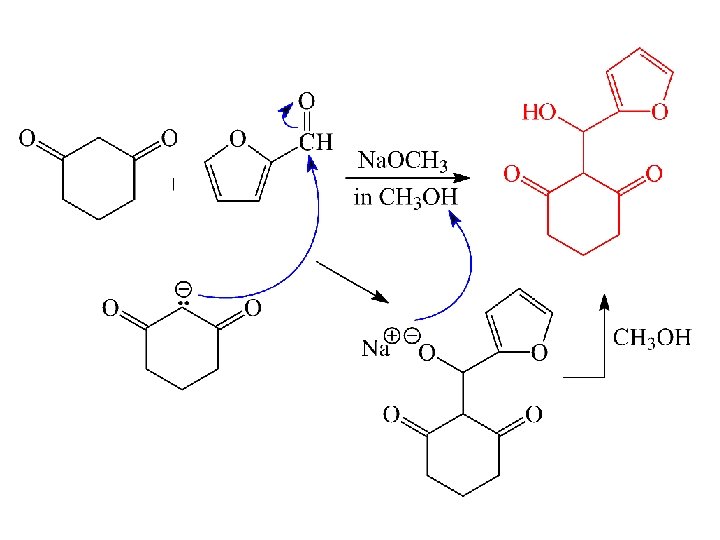
Predict the Product
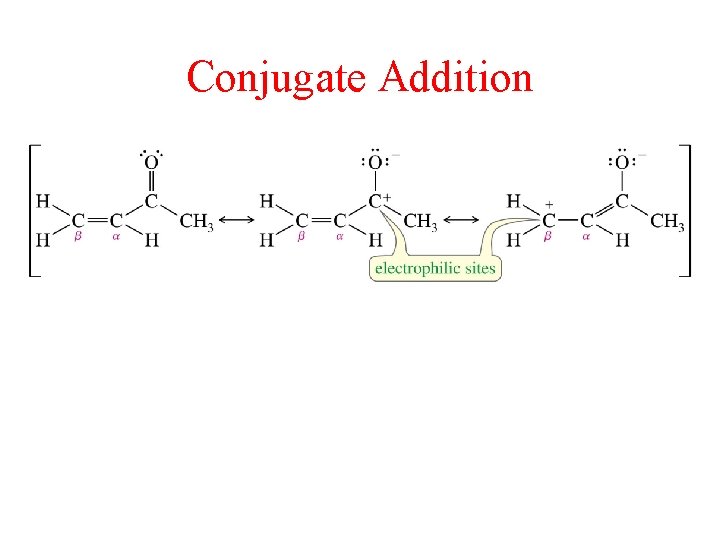
Conjugate Addition
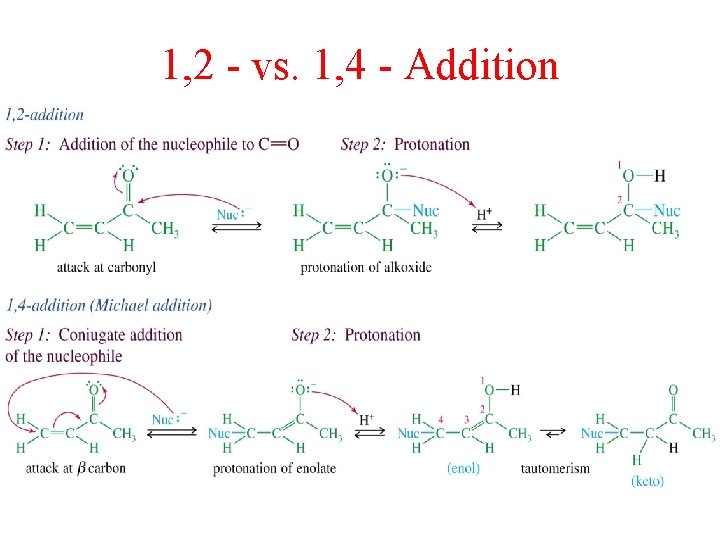
1, 2 - vs. 1, 4 - Addition
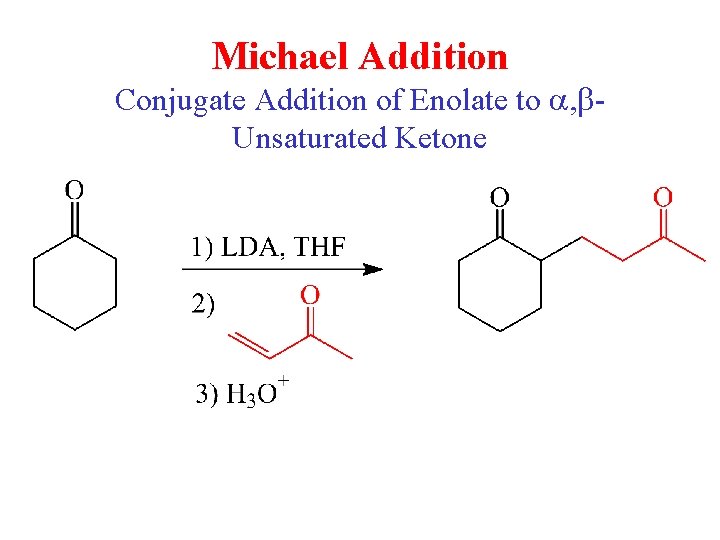
Michael Addition Conjugate Addition of Enolate to a, b. Unsaturated Ketone
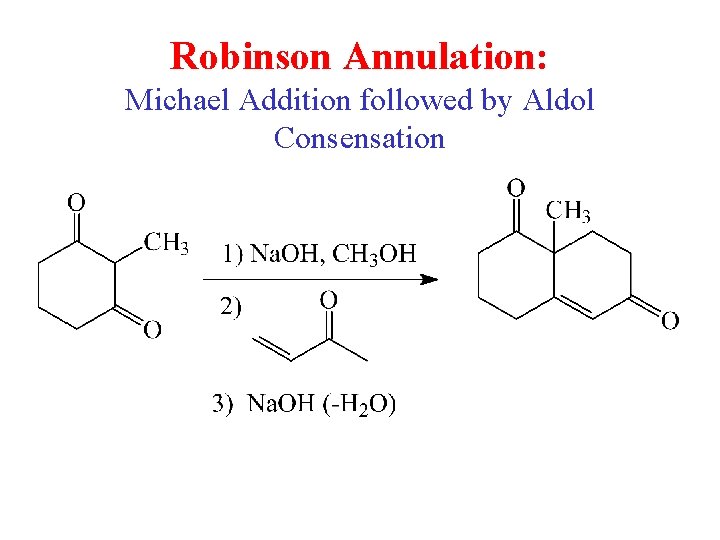
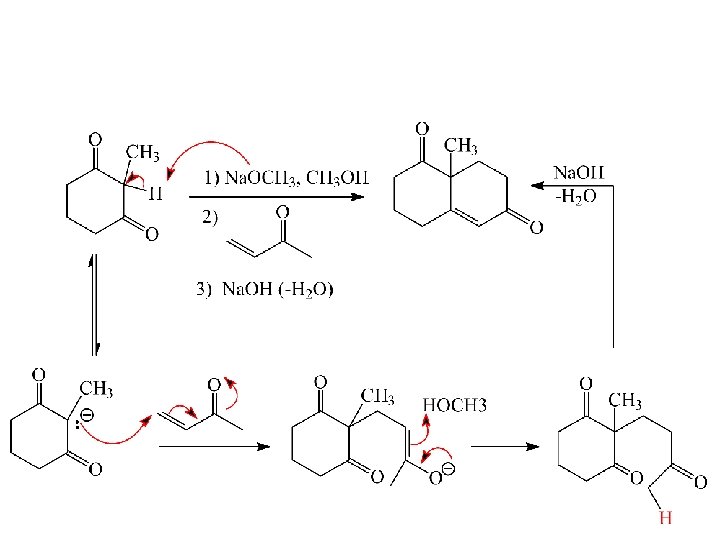
Robinson Annulation: Michael Addition followed by Aldol Consensation
- Slides: 41