I Carbonyl aSubstitution using Enols 2 Reactions of

I. Carbonyl a-Substitution using Enols 2. Reactions of Enols c. a-Halogenation of aldehydes and ketones d. The Hell-Volhard-Zolinskii reaction
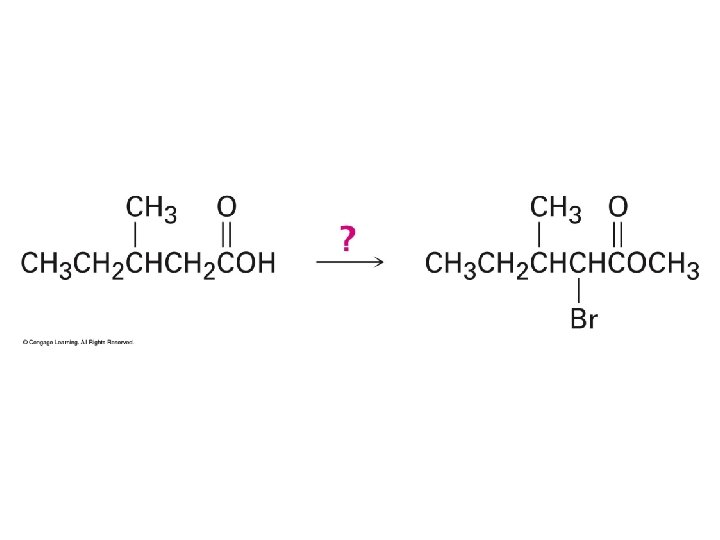

I. Carbonyl a-Substitution using Enols 3. Limitations of Enols a. Equilibrium

I. Carbonyl a-Substitution using Enols 3. Limitations of Enols a. Equilibrium b. Reactivity

II. Carbonyl a-Substitution using Enolates 1. Advantages of Enolates a. Reactivity
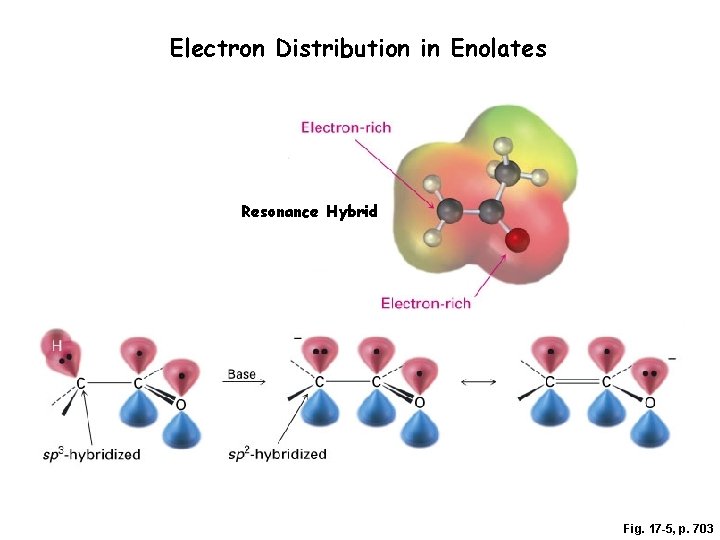
Electron Distribution in Enolates Resonance Hybrid Fig. 17 -5, p. 703

II. Carbonyl a-Substitution using Enolates 1. Advantages of Enolates a. Reactivity b. Quantitative formation

II. Carbonyl a-Substitution using Enolates 2. Preparation of Enolates a. p. Ka’s of important a-hydrogens
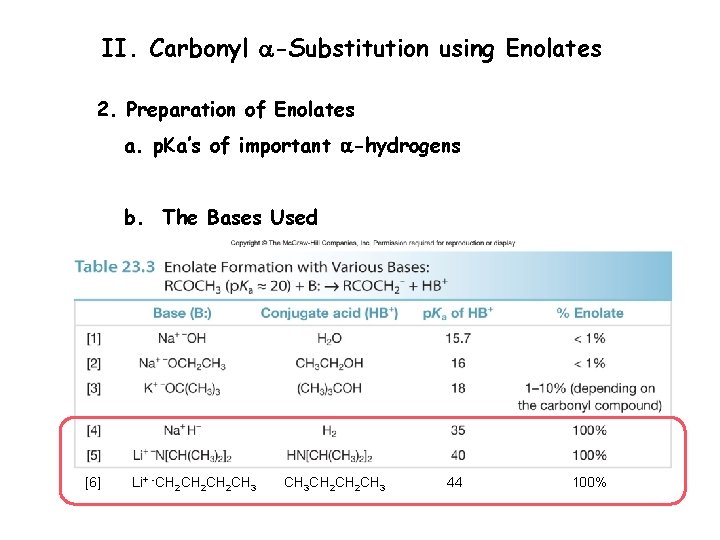
II. Carbonyl a-Substitution using Enolates 2. Preparation of Enolates a. p. Ka’s of important a-hydrogens b. The Bases Used [6] Li+ -CH 2 CH 2 CH 3 CH 2 CH 3 44 100%

II. Carbonyl a-Substitution using Enolates 3. Reactions of Enolates a. The reactive site

II. Carbonyl a-Substitution using Enolates 3. Reactions of Enolates a. The reactive site b. Reactions of ketones i. thermodynamic and kinetic enolates.

II. Carbonyl a-Substitution using Enolates 3. Reactions of Enolates a. The reactive site b. Reactions of ketones i. thermodynamic and kinetic enolates. c. Reactions of nitriles and esters

II. Carbonyl a-Substitution using Enolates 3. Reactions of Enolates d. Active methylene compounds i. Malonic ester synthesis
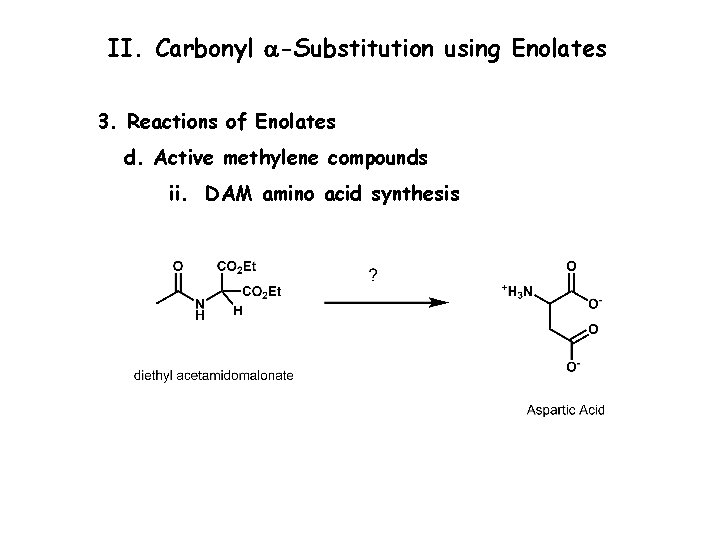
II. Carbonyl a-Substitution using Enolates 3. Reactions of Enolates d. Active methylene compounds ii. DAM amino acid synthesis
- Slides: 14