AP CHEMISTRY UNIT 2 ATOMS MOLECULES AND IONS










- Slides: 17
AP CHEMISTRY UNIT 2 – ATOMS, MOLECULES, AND, IONS Day 3 – Finish Analysis of Food Dyes and Mass Spectroscopy

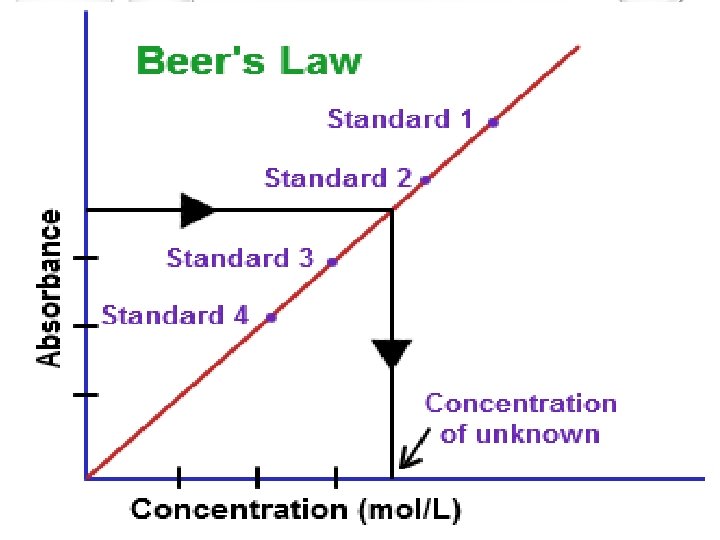
WARM UP SIT WITH LAB PARTNERS TAKE OUT: LAB NOTEBOOK MAKE: “ANALYSIS” SECTION BELOW DATA SECTION FIND: YOUR GRAPHS IN BLUE BIN FOLD, CUT, AND TAPE GRAPHS: INTO ANALYSIS SECTION ON YOUR GRAPHS: FIND EQUATIONS FOR ABSORBANCE AND %TRANSMITTANCE WRITE: EQUATIONS IN ANALYSIS SECTION TIME: 6 MINUTES

AP CHEMISTRY AGENDA • FINISH ANALYSIS OF FOOD DYES LAB • UNIT 2 NOTES (MASS SPECTROSCOPY) • GUIDED INQUIRY ASSIGNMENT NEXT CLASSES: • LAB NOTEBOOK DUE TUESDAY WITH ANALYSIS, CONCLUSION, SOURCES OF ERROR, AND POST-LAB QUESTIONS


FINISH ANALYSIS OF FOOD DYES FIND: LAB HANDOUT “GUIDED INQUIRY” SECTION READ: 1, 2, 4 TALK: WITH LAB PARTNERS TO DETERMINE AN APPROPRIATE PROCEDURE TO DETERMINE CONCENTRATION OF BLUE DYE TIME: 6 MINUTES

FINISH ANALYSIS OF FOOD DYES YOU WILL: OBSERVE HOW MS. MYRIAH OBTAINS THE ABSORBANCE OF THE GLACIER FREEZE GATORADE THEN: RECORD THE ABSORBANCE IN YOUR DATA TABLE IN ANALYSIS SECTION: CALCULATE THE NUMBER OF MILLIGRAMS OF THE GLACIER FREEZE GATORADE TIME: 6 MINUTES WHEN DONE: BE READY TO SHARE

REMEMBER! COMPLETE: THE FOLLOWING SECTIONS BY TUESDAY • ANALYSIS SECTION • CONCLUSION • SOURCES OF ERROR • POST LAB QUESTIONS IF YOU NEED HELP: REFER TO LAB COMPONENT FOR LAB NOTEBOOK, CLASS WEBSITE, THE INTERNET, OR MESSAGE ME (INCLUDE PICTURES IF

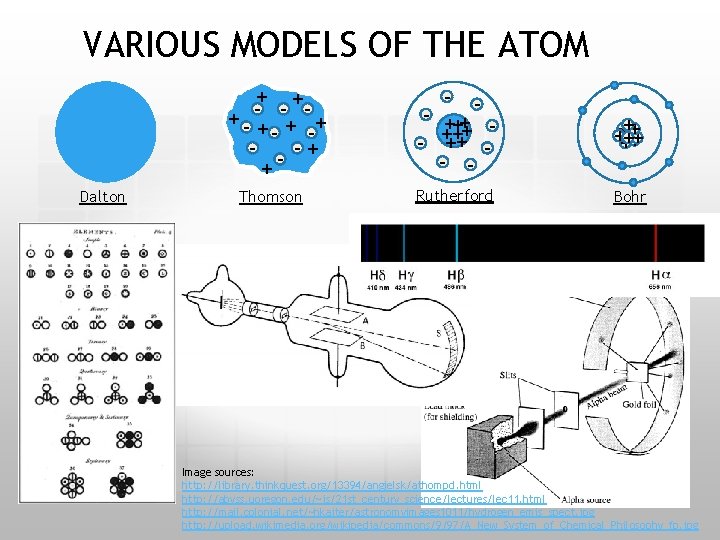
VARIOUS MODELS OF THE ATOM + + - - ++ - + -+ -+ + Dalton Thomson - - - + ++ ++ + - ++ - - + +++++ ++ Rutherford Bohr Image sources: http: //library. thinkquest. org/13394/angielsk/athompd. html http: //abyss. uoregon. edu/~js/21 st_century_science/lectures/lec 11. html http: //mail. colonial. net/~hkaiter/astronomyimages 1011/hydrogen_emis_spect. jpg http: //upload. wikimedia. org/wikipedia/commons/9/97/A_New_System_of_Chemical_Philosophy_fp. jpg

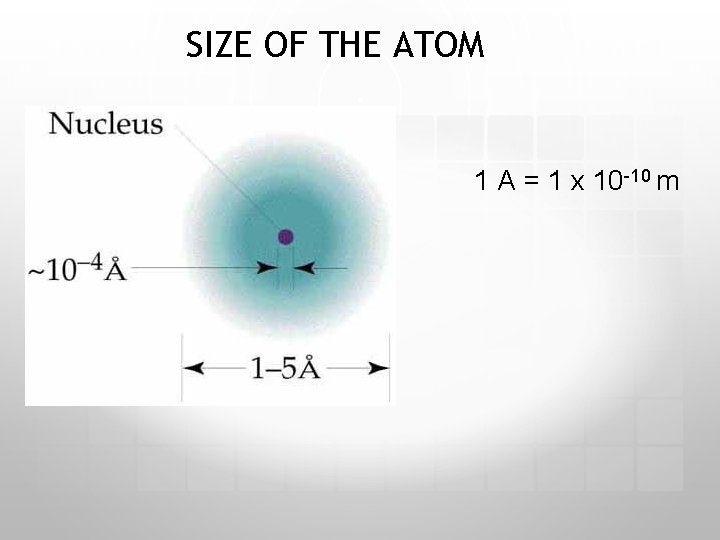
SIZE OF THE ATOM 1 A = 1 x 10 -10 m

ATOMIC NUMBERS, MASS NUMBERS, ISOTOPES


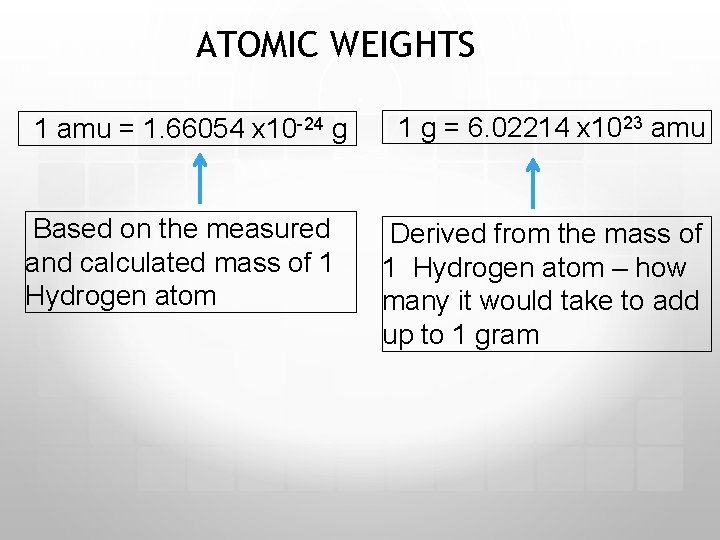
ATOMIC WEIGHTS 1 amu = 1. 66054 x 10 -24 g Based on the measured and calculated mass of 1 Hydrogen atom 1 g = 6. 02214 x 1023 amu Derived from the mass of 1 Hydrogen atom – how many it would take to add up to 1 gram

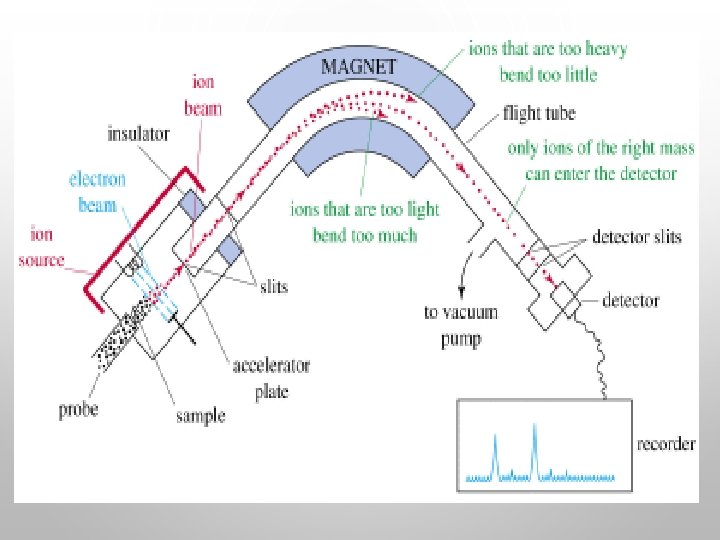
MASS SPECTROSCOPY VIDEO ON NOTEBOOK PAGE: Draw and write notes about Mass Spectroscopy from video MAKE SURE TO: Write Flow Chart

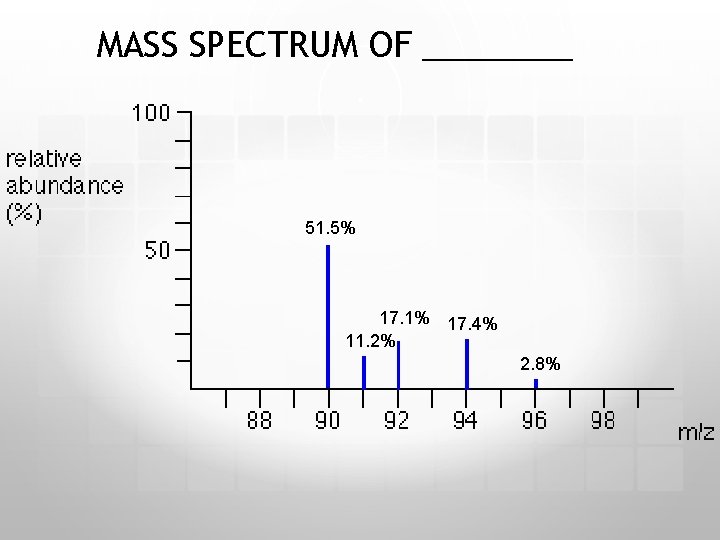
MASS SPECTRUM OF ____ 51. 5% 17. 1% 17. 4% 11. 2% 2. 8%


GUIDED INQUIRY: ATOMIC NUMBER AND MASS WITH THIS GROUP: WORK ON ATOMIC NUMBER AND ATOMIC MASS GUIDED INQUIRY • CTQ = CRITICAL THINKING QUESTIONS TIME: UNTIL END OF CLASS WHEN DONE: HAVE A GREAT WEEKEND AND SEND A MESSAGE IF YOU HAVE A

WHAT WILL YOU NEED TO KNOW FOR UNIT 2 WHAT DO YOU NEED TO KNOW? • CALCULATE ISOTOPES (GUIDED INQUIRY) • MASS SPECTROSCOPY • DALTON’S LAWS • EMPIRICAL FORMULAS • ELECTRON CONFIGURATIONS • HOW TO USE PLANCK’S EQUATION • HOW TO USE BEER’S LAW