Analytical quality control AQC for pesticide residues analysis























- Slides: 24

Analytical quality control (AQC) for pesticide residues analysis Alan Hill Central Science Laboratory, York, UK Rome, 26 -28 November, 2001

Why is AQC necessary? • Analysis is complex – many things can go wrong – interpretation of results can be difficult. • Provides evidence to support results – supports the credibility of data.

SANCO/3013/2000 • Covers all aspects of AQC. • Objectives: – avoid false positives and false negatives; – acceptable accuracy and precision; – EU harmonisation of fit-for-purpose AQC, to provide uniform level of confidence in reported data.

General issues relating to topics that other speakers will consider… • Accreditation, record keeping, traceability, etc. – important to document the supporting evidence. • Sampling and sub-sampling – important because these procedures determine the actual residue levels present, irrespective of the quality of the analysis.

Sampling • MRL applies to the sample, not a subsample or the lot. • Sample should be representative of the lot – impossible to prove but it should have a high probability of being representative; – few validation data available to show the probability of an incorrect conclusion. • Mixed lots are a problem.

What is a uniform lot?

Sampling • MRL applies to sample, not a sub-sample or the lot. • Sample should be representative of the lot – impossible to prove but it should have a high probability of being representative; – few validation data available to indicate probability of incorrect conclusion. • Mixed lots are a problem. • Sample transport and storage.

Sub-sampling • Sub-sample (analytical portion) should be representative of the sample -

Sub-sampling

Sub-sampling • Sub-sample (analytical portion) should be representative of the sample – replicate analysis provides evidence of homogeneity (precision of sub-sampling), – few validation data available to prove analyte stability (accuracy of sub-sampling), – few validation data available to prove the minimum portion size giving acceptable precision, – volatile analytes are problematic - very few validation data available.

Calibration standards • Accuracy is dependent on good standards. • Standards occasionally incorrectly identified. • Purity can be difficult to determine, hence expiry dates. • Cross-checking “new” and “old” can help to eliminate gross errors.

Extraction & concentration • Very few validation data for extraction efficiency – not usually a problem with fruit and vegetables but may be with dry cereal products. • Could be an issue where a common moiety is determined. • Precision requirements may restrict the use of small extract volumes. • Internal standards may have advantages.

Contamination and interference • Can occur in the best laboratories. • Leads to poor accuracy, wrong identification, no results and/or higher costs. • Potential contamination with the analyte can be detected by use of mixed standards. • Interference can be indirect (“matrix effects”). • Natural presence of the analyte - a problem.

Calibration • Shape of detector response curve must be known. • Changes in detector response must be tracked. • Number of points and frequency of repeat injections dependent upon stability of detector response. • Extrapolation of the calibration curve beyond LCL and HCL requires caution.

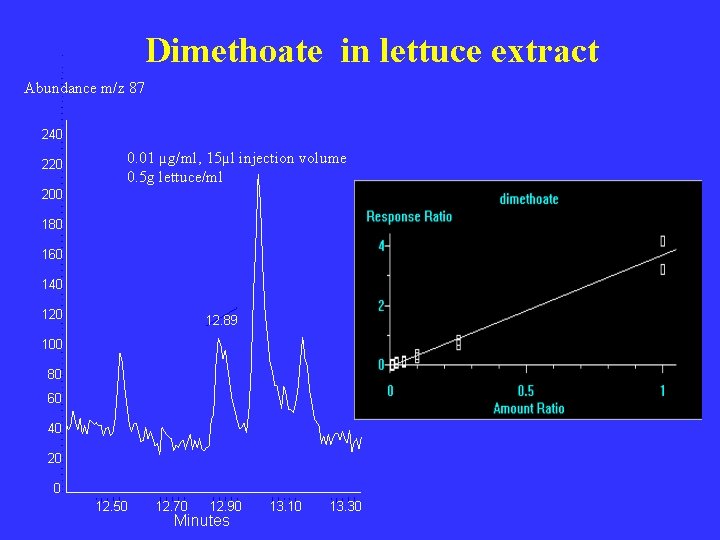
Dimethoate in lettuce extract Abundance m/z 87 240 220 0. 01 µg/ml, 15µl injection volume 0. 5 g lettuce/ml 200 180 160 140 12. 89 100 80 60 40 20 0 12. 50 12. 70 12. 90 Minutes 13. 10 13. 30

Calibration • “Matrix effects” should be taken into account. • “Matrix effects” are variable: the only way to be sure of compensating for them is by “standard addition”. • “Representative analytes” may be used but data must be interpreted with caution.

Analytical performance • Methods capable of 70 -110% recovery. • Routine performance within 60 -140% for recovery at 1 to 10 times the LCL. • Recovery for MRL exceedance, 70 -110%. • “Representative analytes” and “representative matrices” may be used…but data must be interpreted with caution. • Recovery measurement not possible? – rely on calibration data only.

Analytical performance • Participate in proficiency tests. • Investigate unacceptable scores and rectify problems. • Analyse in-house reference materials – mainly suitable for single-analyte methods.

Confirmation • Negative results require acceptable LCL and recovery. • Positive results require acceptable calibration and recovery and… – other requirements decided case-by-case… – should be qualitative and quantitative; – use different detectors, columns etc; – MS techniques particularly important.

Reporting multi-component results for example, endosulfan Result Report options = <0. 05 SO 4 = <0. 1 (i) <0. 2 (ii) 0, <0. 2 (iii) <0. 1 = 0. 05 = <0. 05 SO 4 = <0. 1 (i) <0. 2 (ii) 0. 05, <0. 2 (iii) 0. 05 = 0. 05 SO 4 = <0. 1 (i) <0. 2 (ii) 0. 1, <0. 2 (iii) 0. 1 = <0. 05 SO 4 = 0. 1 (i) <0. 2 (ii) 0. 05, <0. 2 (iii) 0. 1

Uncertainty • Required by ISO 17025. • “Typical uncertainty”, not specific uncertainty. • Most easily measured from AQC data, precision and accuracy required. • Doubts remain over the best methods for estimation. • Users of data may not understand the implications.

In future…. . ? • • Cost reduction is important…. . AQC for 0. 01 -0. 05 mg/kg residues? AQC for rapid or large-scale screening? In both cases: – must avoid major bias and – must have correct identification but… – what precision is acceptable? what range for routine recovery? – what limits for ion ratios for confirmation?

Identification points? MS technique* points per ion** LR-MS 1. 0 LR-MSn transition product ions 1. 0 HR-MS 2. 0 HR-MSn transition product ions 2. 5 Other techniques points for each or all LC-DAD, LC-FL or 1. 0 2 -D TLC + UV spectrum * EI and CI counted separately ** Each ion counted once only

Summary • AQC provides the evidence to support validity of results. • More rigorous AQC = more support, • Less rigorous AQC = less support. • Results and AQC must be fit for purpose. • The analyst or the “customer” must decide fitness for purpose but… • …if a result is proven to be wrong, the analyst will blamed!