10 2 MoleMass and MoleVolume Relationships The MoleMass











- Slides: 13

10. 2 Mole-Mass and Mole-Volume Relationships

The Mole-Mass Relationship • Use molar mass of an element or compound to convert between the mass of a substance and the moles of a substance. Example: • Let’s say you need 3. 00 mol of salt (Na. Cl) for a recipe. How do you measure this amount on a scale? 1 st Find Molar mass of Na. Cl = 58 g 2 nd Multiply by a conversion factor

• Suppose you need to obtain 5 moles of water to water your hydrangea plant. • How much is that in grams? Molar Mass of H 2 O= 18 g/mol

Do the same thing in reverse • • Rust is often a problem in agriculture because it weakens chains or other machinery made of iron. When iron is exposed to air, it corrodes to form red-brown rust. Rust is iron (III) oxide (Fe 2 O 3). How many moles of iron (III) oxide are contained in 92. 2 g of pure Fe 2 O 3 • Knowns -Mass 1 mol Fe 2 O 3= Unknowns # moles?

Try on your own • Boron is needed in alfalfa plants to prevent stunted growth. In corn, a boron deficiency results in twisted ears and barren stocks. Find the # moles in 3. 70 x 10^-1 g of boron.

Practice with essential plant nutrients! Your Challenge: You recently inherited 500 acres of corn from your father. It’s time to fertilize again but you have no idea where to start! Solve the problems below to figure out how much fertilizer you need to apply to your corn. Good Luck!

List of 10 1. How many particles is in 1 mole of Neon? 2. What are two examples of representative particles? 3. What is the molar mass of Potassium? 4. What’s the molar mass of KCl? 5. What’s the molar mass of Bromine? 6. What’s Avagadro’s number? 7. How do you convert grams to moles? 8. How do you convert moles to grams? 9. How many atoms is in 12 mol of H 2 O? 10. The atomic mass of ANY element is how many atoms?

Mole-Volume Relationship • Avagadro’s Hypothesis: Equal volumes of gases at the same temperature and pressure contain equal numbers of particles. – Particles of different gases are not the same size. – BUT particles in all gases are so far apart that larger particles don’t need much more space than the same # or smaller particles.

• Avagadro’s hypothesis Continued: 1 mol of gas at Standard Temperature and pressure (STP) occupies a volume of 22. 4 L. - 0° C, 101. 3 k. Pa In other words: • At STP; 1 mol (or 6. 02 x 10^23 representative particles, of any gas occupies a volume of 22. 4 L) Calculating Volume at STP: Suppose you have. 375 mol of oxygen gas and want to know what volume the gas will occupy at STP Volume of O 2 =. 375 mol x 22. 4 L = 8. 40 L 1 mol

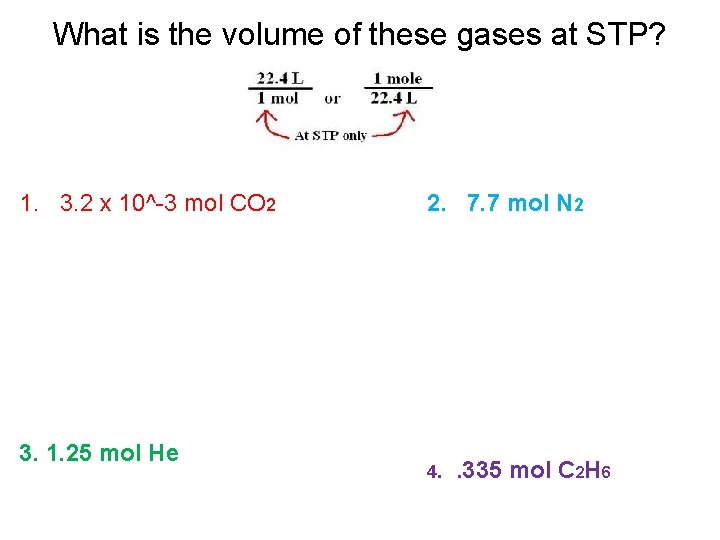
What is the volume of these gases at STP? 1. 3. 2 x 10^-3 mol CO 2 3. 1. 25 mol He 2. 7. 7 mol N 2 4. . 335 mol C 2 H 6

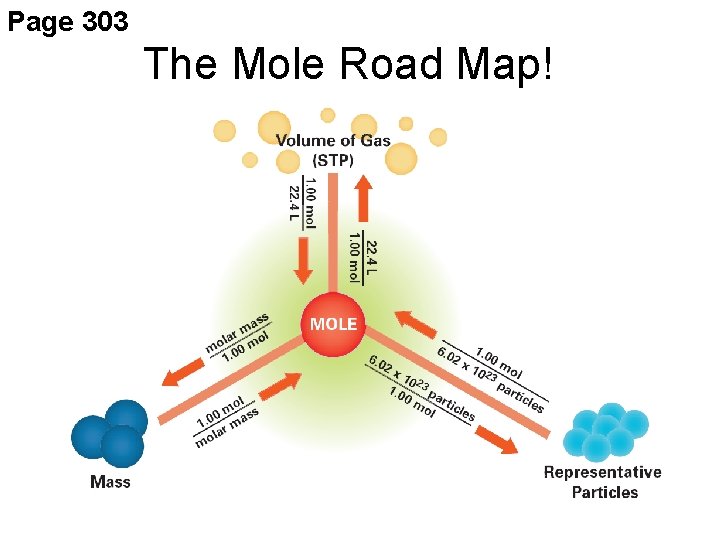
Page 303 The Mole Road Map!

Lower pressure Higher temperature Lower temperature Higher pressure

Brownie Challenge! Ingredients: 16. 38 grams of cocoa (CH 3 CH 2) 22. 3 grams softened butter (C 4 H 8 O 2). 024 grams salt (Na. Cl) 17. 65 grams flour (C 6 H 10 O 5) 120 grams of eggs (C 44 H 9109 NP)