Water Life Civilization Why Water Water H 2














- Slides: 31

Water, Life, Civilization: Why Water?

Water: H 2 O

Water: H 2 O

Water and Similar Compounds H H O S Water H 2 O Hydrogen Sulfide H 2 S

Water: H 2 O Hydrogen Sulfide: H 2 S

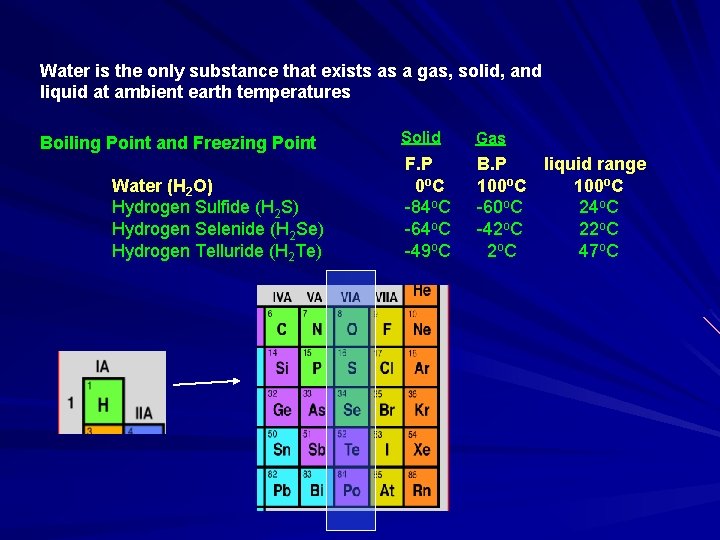
Water is the only substance that exists as a gas, solid, and liquid at ambient earth temperatures Boiling Point and Freezing Point Water (H 2 O) Hydrogen Sulfide (H 2 S) Hydrogen Selenide (H 2 Se) Hydrogen Telluride (H 2 Te) Solid Gas F. P 0 o. C -84 o. C -64 o. C -49 o. C B. P liquid range 100 o. C -60 o. C 24 o. C -42 o. C 2 o. C 47 o. C

H 2 O Two hydrogen atoms One Oxygen atom What makes water so unusual?

Hydrogen: 1 electron (-), 1 proton (+) Oxygen: 8 electrons (-), 8 protons (+) In water, the hydrogens shares their one electron with oxygen, which shares one of its electrons with each hydrogen. This sharing of electrons forms the bond between hydrogen and oxygen atoms to make the water molecule.

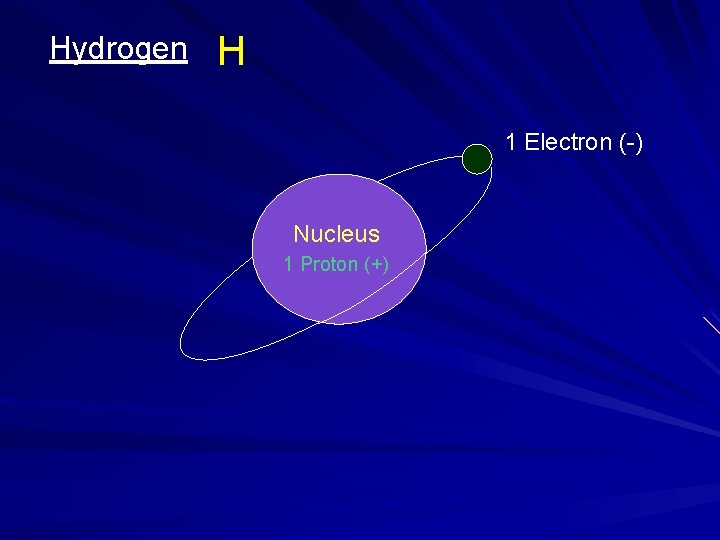
Hydrogen H 1 Electron (-) Nucleus 1 Proton (+)

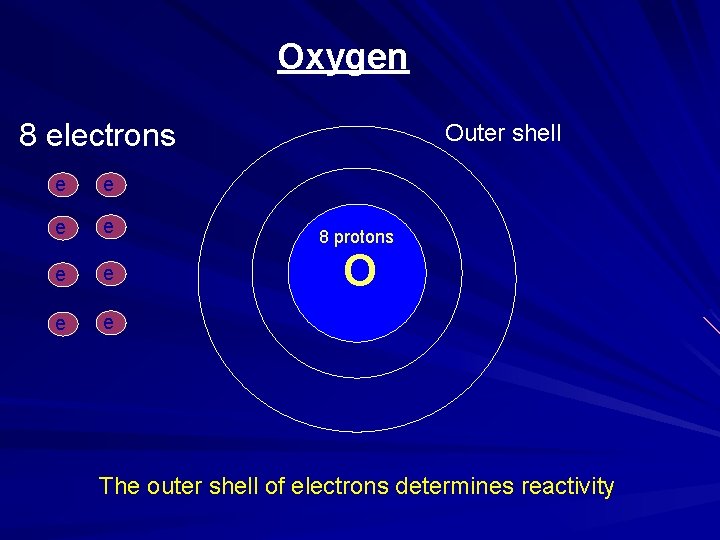
Oxygen 8 electrons e e e e - - Outer shell - 8 protons O - The outer shell of electrons determines reactivity

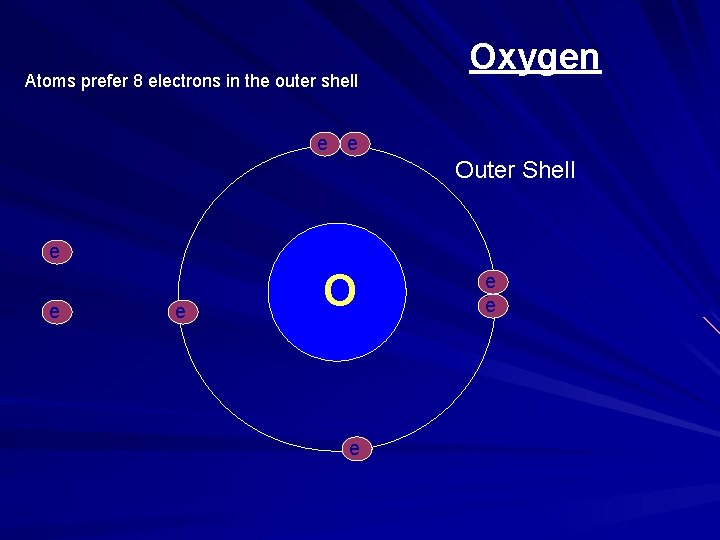
Atoms prefer 8 electrons in the outer shell Oxygen e e - - Outer Shell e - e O - e e -

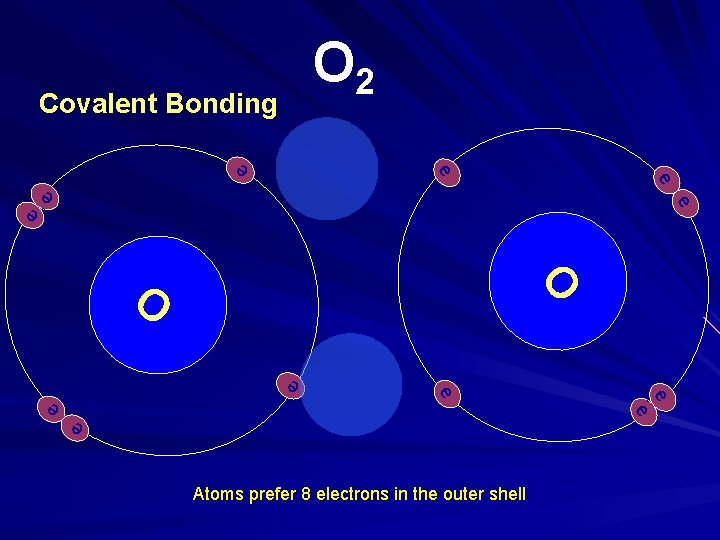
How does oxygen exist in the atmosphere? O 2

O 2 Covalent Bonding e- - e- e - e- O - ee - e- e - Atoms prefer 8 electrons in the outer shell e e e

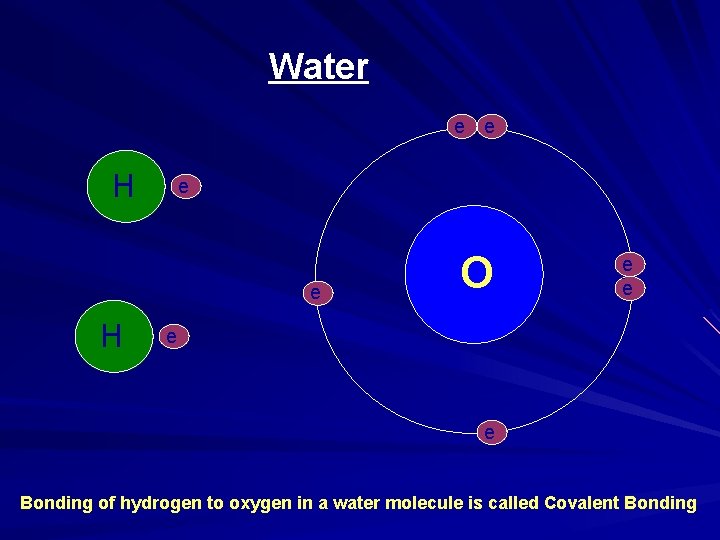
Water e e - HH - e O - H e e - e - Bonding of hydrogen to oxygen in a water molecule is called Covalent Bonding

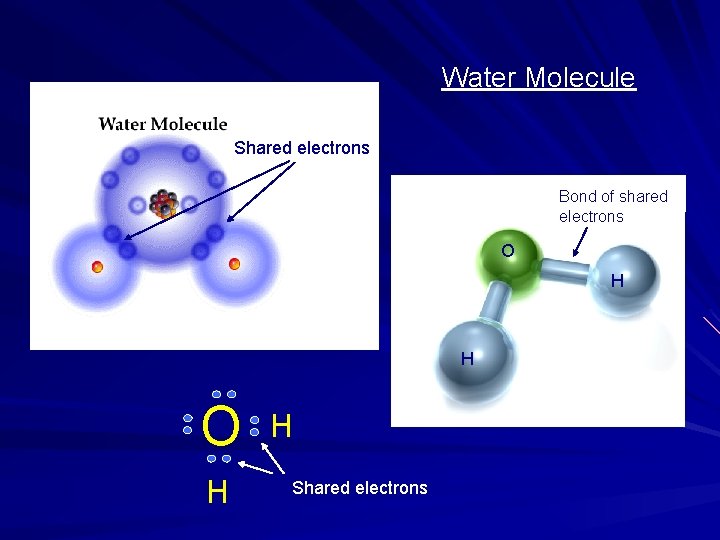
Water Molecule Shared electrons Bond of shared electrons O H H Shared electrons

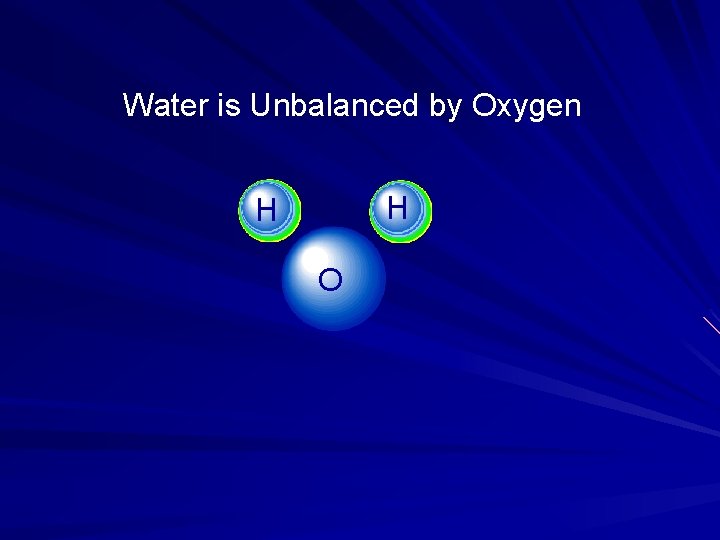
Water is Unbalanced by Oxygen H H O

Oxygen is “Electron Greedy” Oxygen attracts electrons very strongly e - H e - e - Oxygen’s pull on electrons is about 50% stronger than hydrogen

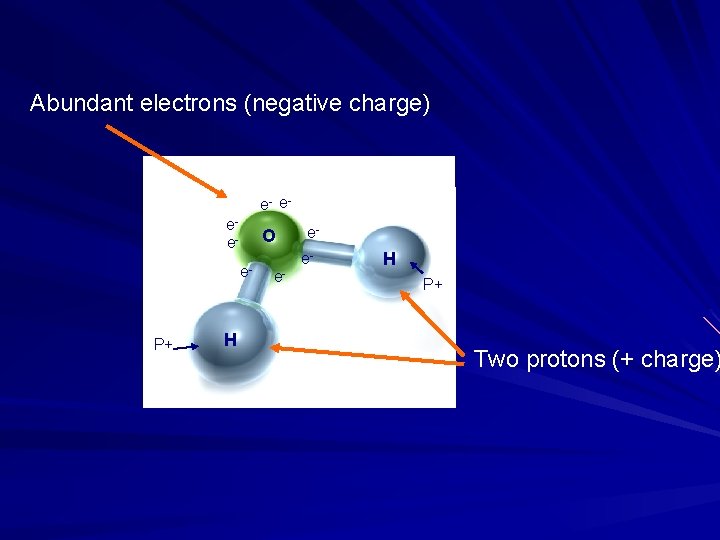
Electrons are negatively charged, protons positive Oxygen is “electron greedy” Oxygen pulls electrons toward itself and away from hydrogen e- eee- O e- e- ee- H P+ P+ P+ H P+ This pulling of electrons toward itself is called “electronegativity”

Abundant electrons (negative charge) e- eee- O e- P+ H e- ee- H P+ Two protons (+ charge)

More negative charges (-) Fewer negative charges

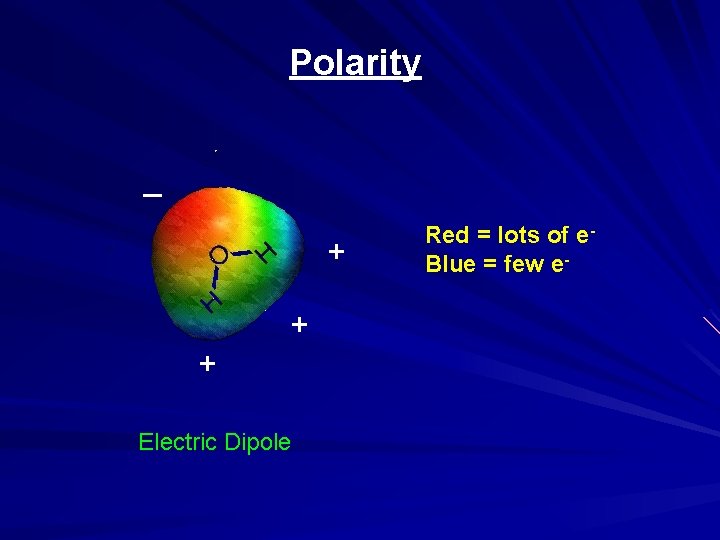
Polarity _ + + + Electric Dipole Red = lots of e. Blue = few e-

Consequences of Polarity

Magnets and Polarity N Magnetic Dipole S

Polarity N S Opposite Poles Attract

Same Poles Repel S N Polarity

Polarity N N S S http: //games. mochiads. com/c/g/polarityfreak-11 -Mochi-Sec. swf

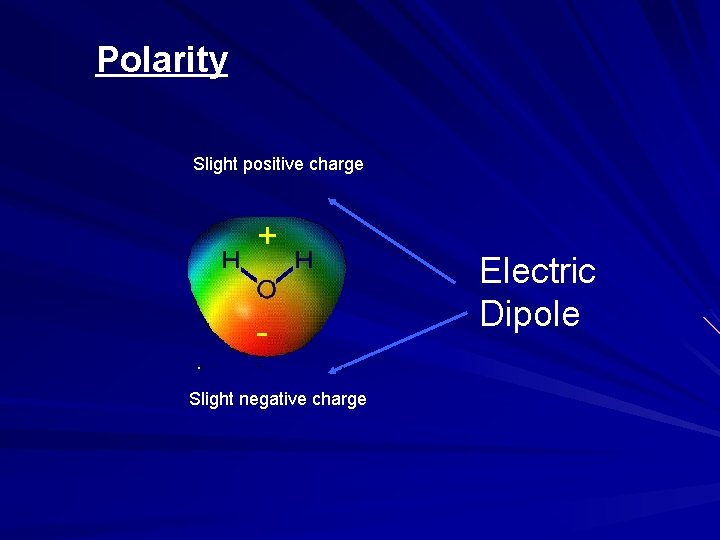
Polarity + Slight positive charge Slight negative charge Electric Dipole

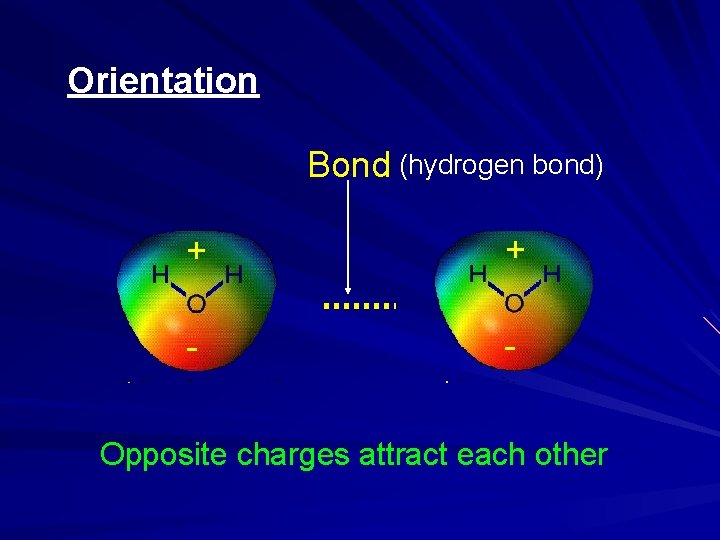
Orientation + + Bond (hydrogen bond) - - Opposite charges attract each other

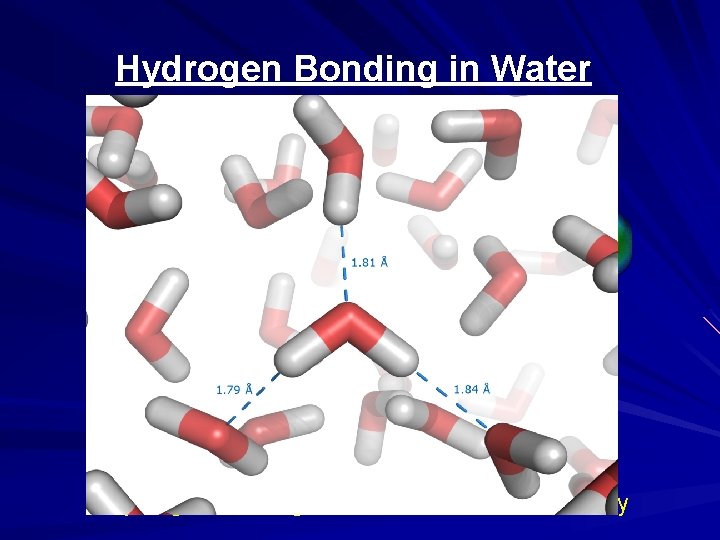
Hydrogen Bonding in Water Hydrogen Bonding Gives Water Unusual Stability

Summary Hydrogen and oxygen share electrons to form water The water molecule is electrically unbalanced Oxygen is electron greedy; it is highly electronegative Oxygen draws electrons toward itself and away from hydrogen This creates a slight negative charge near oxygen There is also a slight positive charge near hydrogen The result is a molecule that is polar (+ and – poles) This polarity accounts for electrostatic bonding between water molecules Bonding between water molecules gives water unusual stability.

Effect on Properties Extensive Hydrogen Bonding Allows Water to Exist as a Liquid at Normal Temperatures and across a wide range in temperatures High Boiling and Freezing Points Other Unusual Thermal Properties Unusual Density