Water Chapter 3 Water n n n Life












![Fig. 3 -UN 5 0 Acidic [H+] > [OH–] Neutral [H+] = [OH–] Basic Fig. 3 -UN 5 0 Acidic [H+] > [OH–] Neutral [H+] = [OH–] Basic](https://slidetodoc.com/presentation_image_h2/5ea7c1a686bb5acfdfd0245f726b08f1/image-20.jpg)




- Slides: 27

Water Chapter 3

Water n n n Life began in water 2/3’s of an organisms body Organisms grow or reproduce in a water-rich environment.

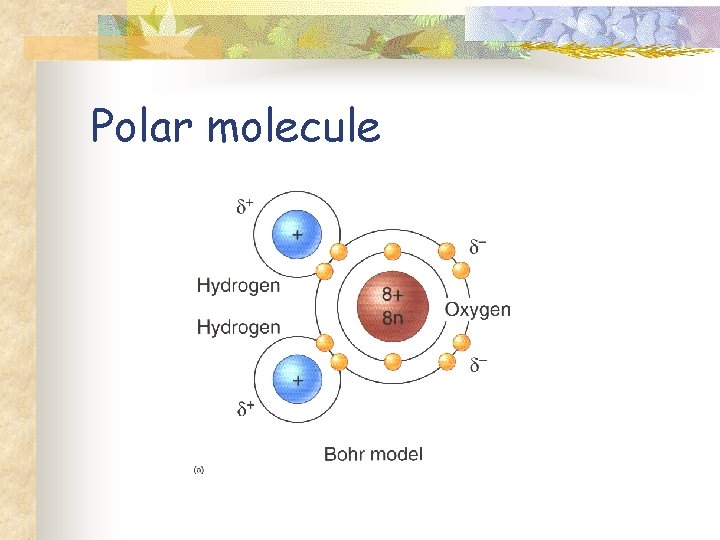
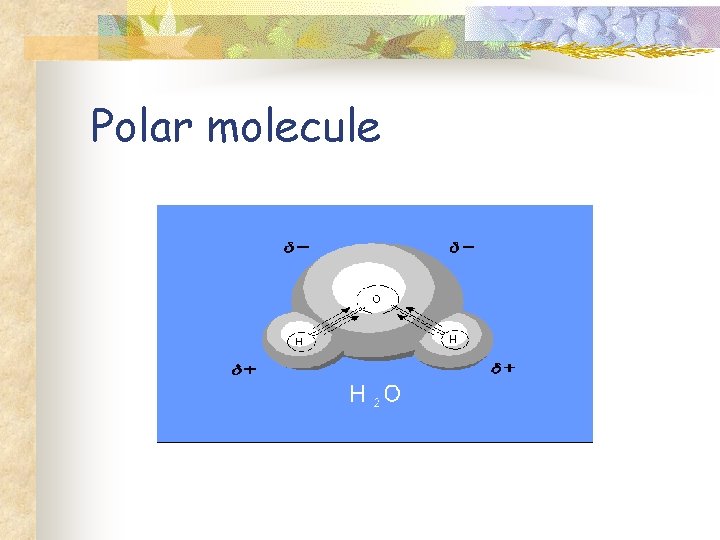
Water Molecule n n n Covalent bonding Oxygen is more electronegative Polar molecule Polarity of water underlies its chemistry Chemistry of life.

Polar molecule

Polar molecule

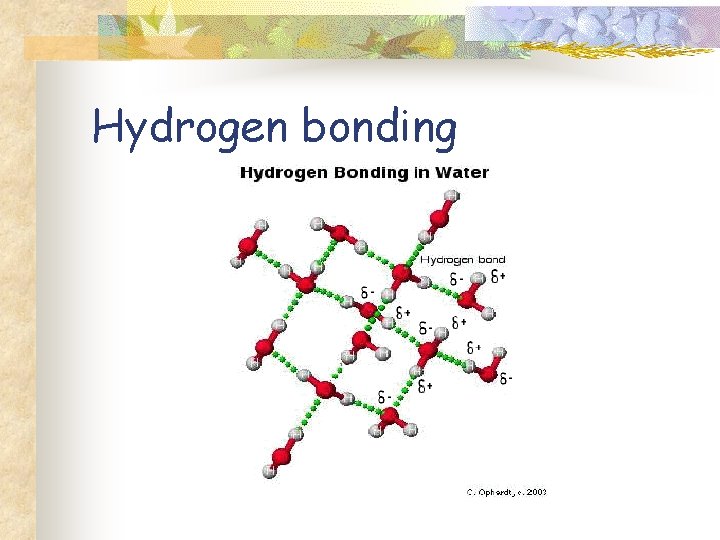
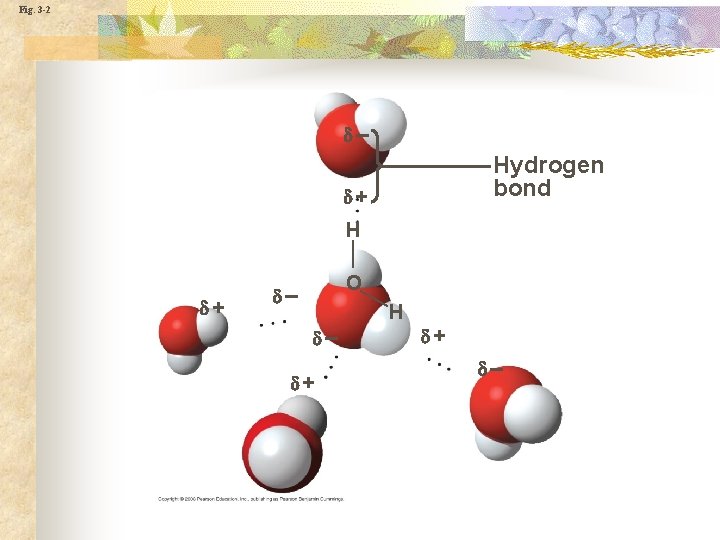
Hydrogen bonding


Properties of water • • • Cohesion: Attraction between water molecules Responsible for surface tension of water

Properties of water n n n Adhesion: Attraction of water between other molecules Capillary action

Properties of water n n n Moderation of water temperature Water is a liquid at moderate temperatures Specific heat: Amount of heat needed to a raise 1 gram of a substance 10 Celsius Water’s specific heat is 1 calorie/gram/0 C (4. 18 Joules)

Properties of water n n Evaporative cooling Heat of vaporization: Amount of heat needed to change 1 gram of a substance from a liquid to a gas. 586 Calories (2260 Joules)

Properties of water n n Ice floats Less dense

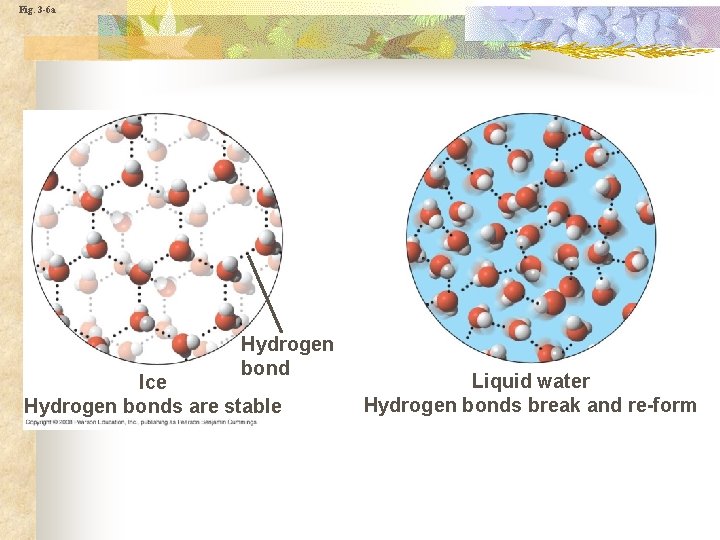
Fig. 3 -6 a Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form

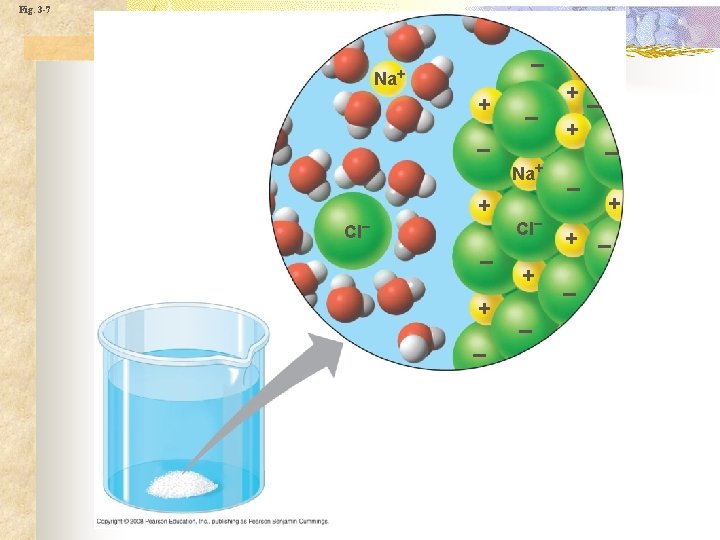
Properties of water n n n Solvent Water surrounds ionic & polar molecules Table salt or sugar Aqueous solution Molarity


Hydrophobic n n “fear” of water Common in non-polar molecules Non-polar molecules tend to aggregate in water Hydrophobic exclusion

Hydrophilic n n “water-loving” Common in polar molecules

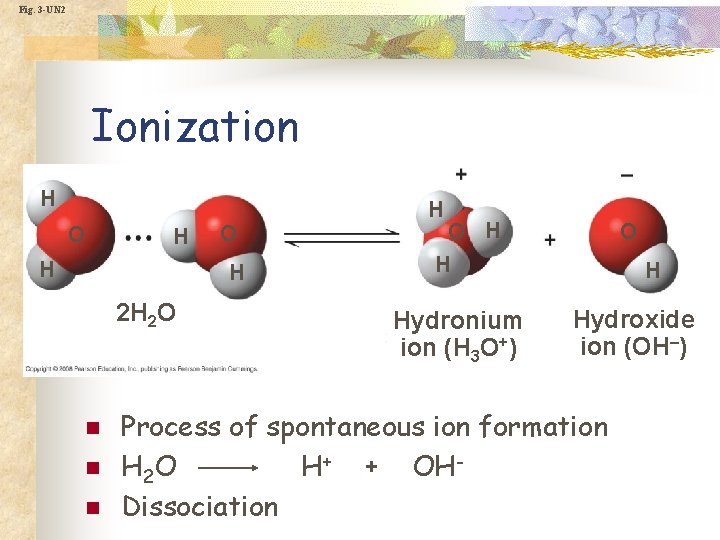
Fig. 3 -UN 2 Ionization H O H 2 H 2 O n n n H O H H Hydronium ion (H 3 O+) O H Hydroxide ion (OH–) Process of spontaneous ion formation H 2 O H+ + OHDissociation

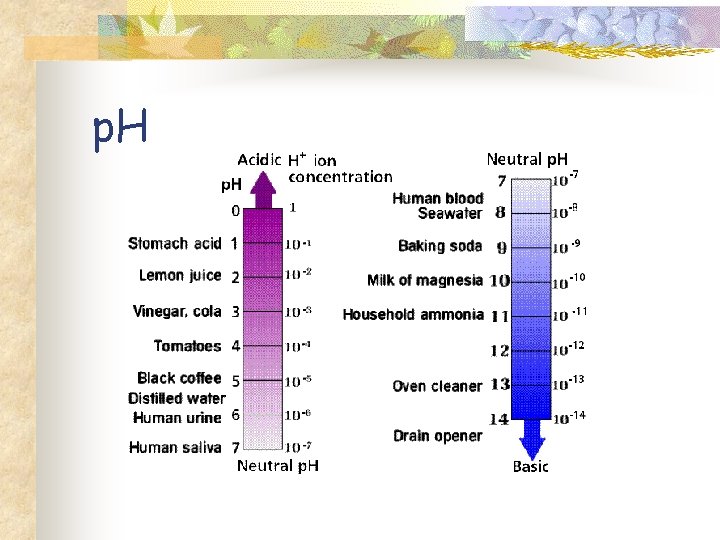
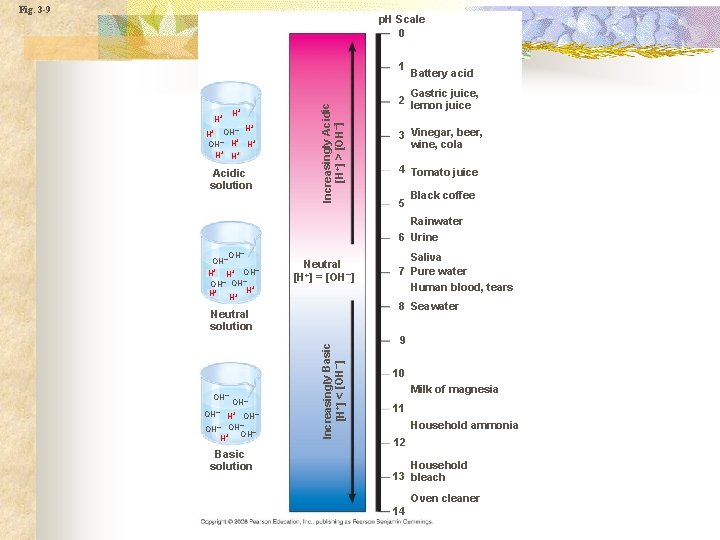
p. H n n n Measurement of the concentration of hydrogen ion. p. H = -log[H+] Acid: Increase concentration of H+ ions or a lower p. H. Base: Decrease in concentration of H+ions or a higher p. H.
![Fig 3 UN 5 0 Acidic H OH Neutral H OH Basic Fig. 3 -UN 5 0 Acidic [H+] > [OH–] Neutral [H+] = [OH–] Basic](https://slidetodoc.com/presentation_image_h2/5ea7c1a686bb5acfdfd0245f726b08f1/image-20.jpg)
Fig. 3 -UN 5 0 Acidic [H+] > [OH–] Neutral [H+] = [OH–] Basic [H+] < [OH–] Acids donate H+ in aqueous solutions 7 Bases donate OH– or accept H+ in aqueous solutions 14

p. H

Fig. 3 -9 p. H Scale 0 H+ H+ + – H H+ OH– H H+ H+ H+ Acidic solution Increasingly Acidic [H+] > [OH–] 1 Battery acid Gastric juice, 2 lemon juice 3 Vinegar, beer, wine, cola 4 Tomato juice 5 Black coffee Rainwater 6 Urine OH– H+ OH– OH– + H+ H+ H Neutral [H+] = [OH–] 8 Seawater OH– H+ OH– – OH OH– + H Basic solution Increasingly Basic [H+] < [OH–] Neutral solution OH– Saliva 7 Pure water Human blood, tears 9 10 Milk of magnesia 11 Household ammonia 12 Household 13 bleach Oven cleaner 14

Buffer n n n Substance helps maintain a balanced p. H Accepts H+ ions when excess Donates when there are too few.

Buffer n n Blood p. H is approximately 7. 4. Bicarbonate ion helps maintain the p. H of the blood. Blood acidosis: p. H drops 0. 2 to 0. 4 points on the p. H scale Blood alkalosis: p. H goes up 0. 2 to 0. 4 points on the p. H scale

Buffer n n Bicarbonate ion H 2 CO 3 ⇌ HCO 3 - + H+

Acid rain n n Sulfuric acid Nitric acid

Acid rain