Chapter 3 Water and Life WATER Objectives Importance

Chapter 3: Water and Life

WATER

Objectives • • Importance of H bonds to properties of water 4 unique properties of water Interpreting a p. H scale Importance of buffers in biological systems

Structure of Water • Structure key to special properties – Structure dictates function • Polarity • Hydrogen bonding – Each molecule can form max 4


4 Properties of Water • • Cohesion Moderation of temperature Ice floats on liquid water Water as a solvent

Properties of Water: Cohesive Behavior • Cohesion – Water linked to water – Responsible for surface tension – Partial charges make water “sticky” • Adhesion – Clinging of one substance to another • • Transpiration Water moves through xylem to open stomata What roles do cohesion and adhesion play ? How does polarity of water contribute to its cohesive and adhesive properties?
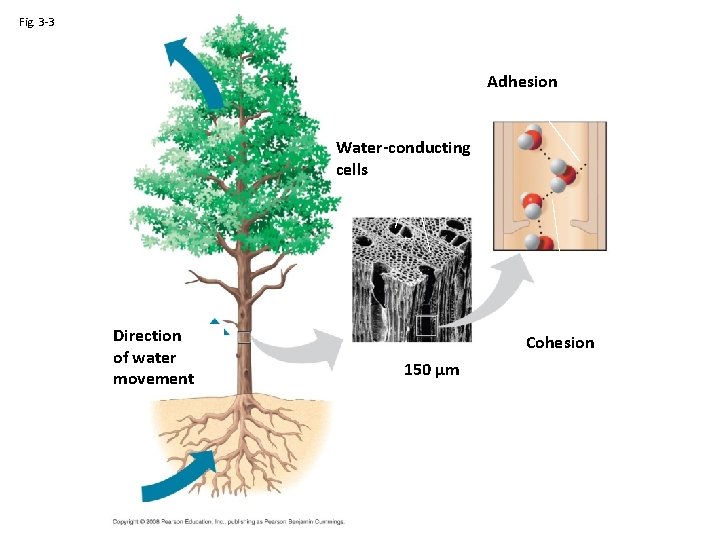
Fig. 3 -3 Adhesion Water-conducting cells Direction of water movement Cohesion 150 µm

Concept Check • How does polarity of water contribute to its cohesive and adhesive properties? • How might Rainex alter the properties of glass to allow it to shed water. • Discuss with a partner then we will discuss as a class.

Properties of Water: Ability to moderate Temperature • 2. Moderation of temperature – Specific heat: amount of heat required to change 1 g of substance 1 degree C – Water has very high specific heat – Implications to earths climate • Oceans • Organisms – Heat of vaporization: amount of heat to convert 1 g of liquid to gas • Water has high heat of vaporization

Evaporative cooling • Evaporation is transformation of a substance from liquid to gas • As a liquid evaporates, its remaining surface cools, a process called evaporative cooling • Evaporative cooling helps stabilize temperatures in organisms and bodies of water

Specific Heat • Water’s high specific heat can be traced to hydrogen bonding – Heat is absorbed when hydrogen bonds break – Heat is released when hydrogen bonds form • The high specific heat of water minimizes temperature fluctuations to within limits that permit life

Properties of Water: Expansion Upon Freezing • 3. Ice insulates large bodies of water – Ice floats – Moderates temps by not allowing large bodies of water to freeze solid – What are affects on pond life? – 104. 5 bond angle
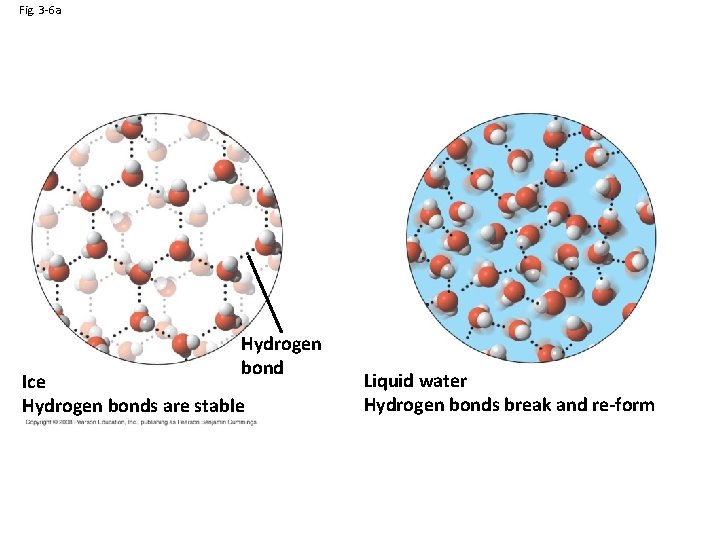
Fig. 3 -6 a Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form
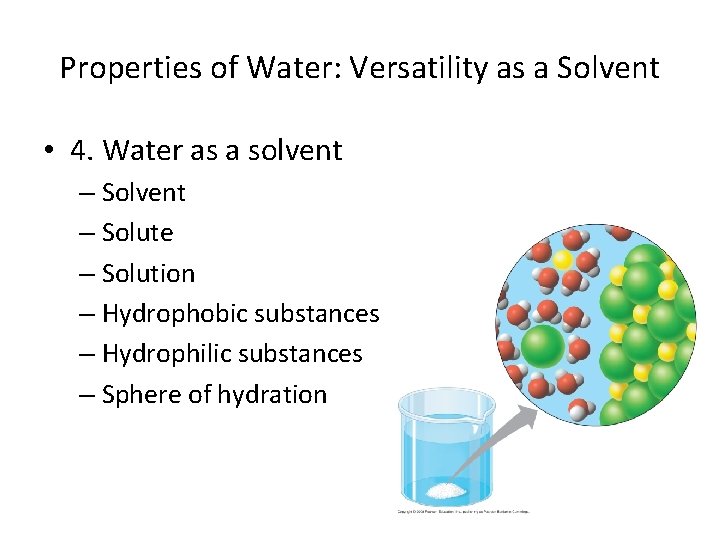
Properties of Water: Versatility as a Solvent • 4. Water as a solvent – Solute – Solution – Hydrophobic substances – Hydrophilic substances – Sphere of hydration
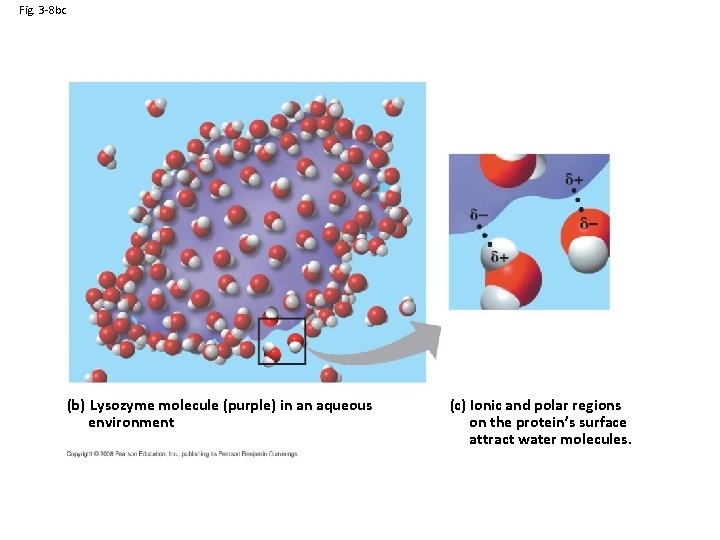
Fig. 3 -8 bc (b) Lysozyme molecule (purple) in an aqueous environment (c) Ionic and polar regions on the protein’s surface attract water molecules.

Deriving p. H equation • In any aqueous solution at 25°C the product of H+ and OH– is constant and can be written as [H+][OH–] = 10– 14 • The p. H of a solution is defined by the negative logarithm of H+ concentration, written as p. H = –log [H+] • For a neutral aqueous solution [H+] is 10– 7 = –(– 7) = 7
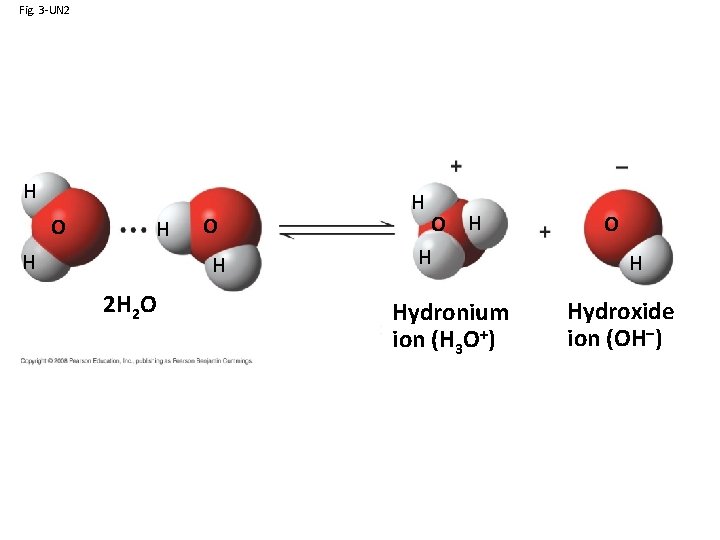
Fig. 3 -UN 2 H O H 2 H 2 O H H Hydronium ion (H 3 O+) O H Hydroxide ion (OH–)
![Acids & Bases • Acids and bases alter [H+] • p. H of pure Acids & Bases • Acids and bases alter [H+] • p. H of pure](http://slidetodoc.com/presentation_image_h/084fe833f0b8158cc705dc9b9458a1ae/image-19.jpg)
Acids & Bases • Acids and bases alter [H+] • p. H of pure water is 7 • How does HCl increase the acidity ? – Increase [H] • How does Na. OH decrease acidity ? – Decrease [H] • direct bases NH 3 gas • Indirect Na. OH
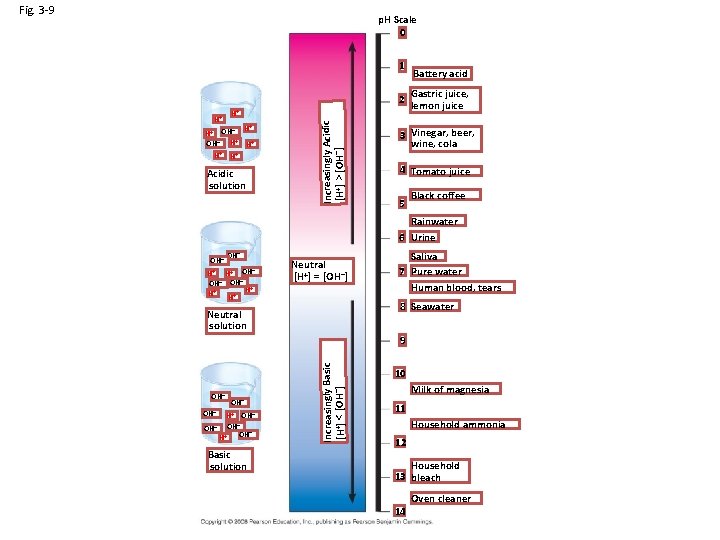
Fig. 3 -9 p. H Scale 0 1 Gastric juice, 2 lemon juice H+ – H+ OH OH– H+ H+ H+ Acidic solution Increasingly Acidic [H+] > [OH–] H+ Battery acid 3 Vinegar, beer, wine, cola 4 Tomato juice 5 Black coffee Rainwater 6 Urine OH– H+ H+ OH– OH– + H H+ H+ Neutral [H+] = [OH–] Saliva 7 Pure water Human blood, tears 8 Seawater Neutral solution OH– OH– H+ OH– – OH OH– + H Basic solution Increasingly Basic [H+] < [OH–] 9 10 Milk of magnesia 11 Household ammonia 12 Household 13 bleach 14 Oven cleaner

Importance of Buffers to Biological Systems • Carbonic acid and hyperventilation – Rapid breathing – Blood CO 2 concentration decreases – Blood carbonic acid levels decrease – Blood p. H changes from normal – Adverse physiological effects • • • Dizziness Visual impairment Fainting Seizures Death
![Acids and Bases • p. H scale – p. H = –log [H+] – Acids and Bases • p. H scale – p. H = –log [H+] –](http://slidetodoc.com/presentation_image_h/084fe833f0b8158cc705dc9b9458a1ae/image-22.jpg)
Acids and Bases • p. H scale – p. H = –log [H+] – 0 -14 – Lower more acidic < 7 – Higher more basic > 7 • Acids: Excess H+ ions • Bases: Excess OH • Pure water is neutral • Buffers: minimize changes in p. H – Can accept or give H+ ions when needed – Carbonic Acid (H 2 CO 3) moderates p. H changes in blood plasma and in the ocean

Summary and Review 1. List and explain the four properties of water that emerge as a result of its ability to form hydrogen bonds 2. Distinguish between the following sets of terms: hydrophobic and hydrophilic substances; a solute, a solvent, and a solution 3. Define acid, base, and p. H 4. Explain how buffers work
- Slides: 23