Types of Solids Three general types 1 Amorphous



![Examples of Lattice Planes in Cubic Lattices (100) Plane with normal direction [100] (110) Examples of Lattice Planes in Cubic Lattices (100) Plane with normal direction [100] (110)](https://slidetodoc.com/presentation_image_h/31df50358d0cd7af017628b3b04573da/image-8.jpg)


- Slides: 15

Types of Solids Three general types 1. Amorphous ― with order only within a few atomonic and molecular dimensions (Fig. (a)) 2. Polycrystalline ― with multiple sing-crystal regions (called grains) separated by grain boundary (Fig. (b)) 3. Single crystal ― with geometric periodicity throughout the entire material (Fig. (c)) (a) (b) (c) 1

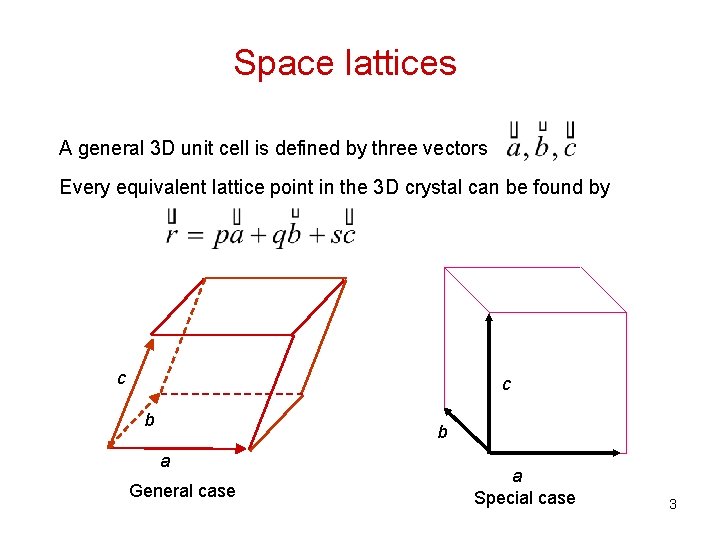
Geometric Description of Single-Crystal — Space Lattices Lattice: lattice: the periodic arrangement of atoms in the crystal Unit cell: unit cell: a small volume that can be used to repeat and form the entire crystal. Unit cells are not necessary unique. 2

Space lattices A general 3 D unit cell is defined by three vectors Every equivalent lattice point in the 3 D crystal can be found by c c b b a General case a Special case 3

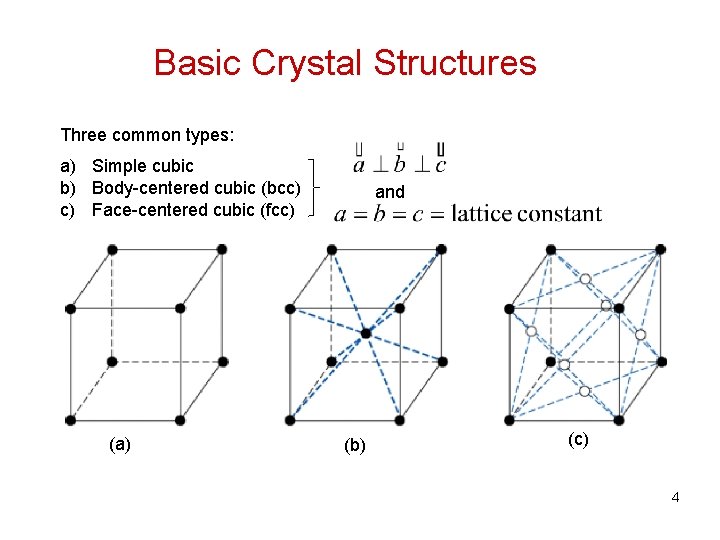
Basic Crystal Structures Three common types: a) Simple cubic b) Body-centered cubic (bcc) c) Face-centered cubic (fcc) (a) and (b) (c) 4

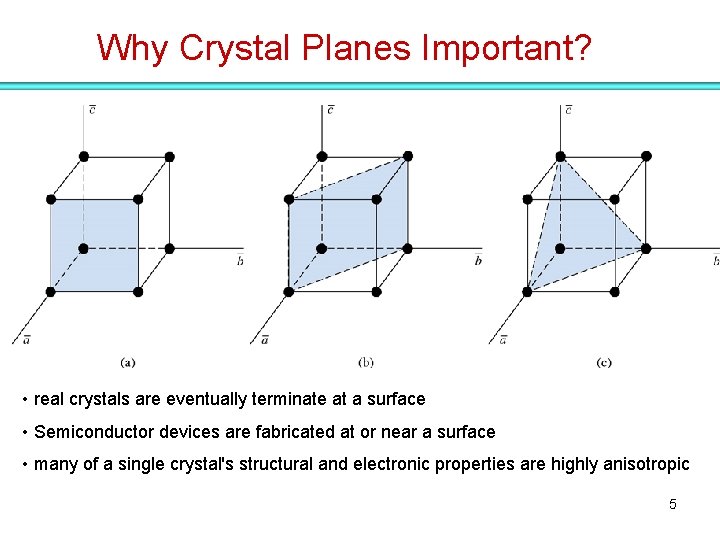
Why Crystal Planes Important? • real crystals are eventually terminate at a surface • Semiconductor devices are fabricated at or near a surface • many of a single crystal's structural and electronic properties are highly anisotropic 5

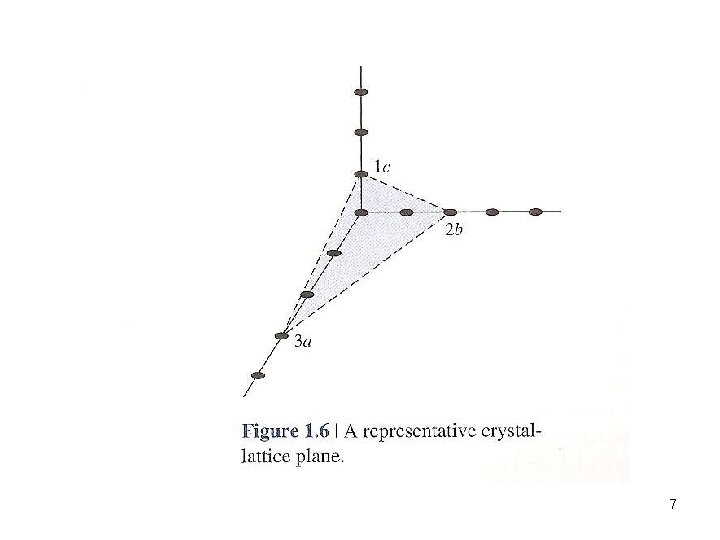
1. Find the intercept on the x, y, and z 2. Reduce to an integer. i. e. lowest common denominator 3. Take the reciprocal and reduce to the smallest set of integers (h, k, l) These are called the Miller Indices 6

7
![Examples of Lattice Planes in Cubic Lattices 100 Plane with normal direction 100 110 Examples of Lattice Planes in Cubic Lattices (100) Plane with normal direction [100] (110)](https://slidetodoc.com/presentation_image_h/31df50358d0cd7af017628b3b04573da/image-8.jpg)
Examples of Lattice Planes in Cubic Lattices (100) Plane with normal direction [100] (110) Plane with normal direction [110] (111) Plane with normal direction [111] 8

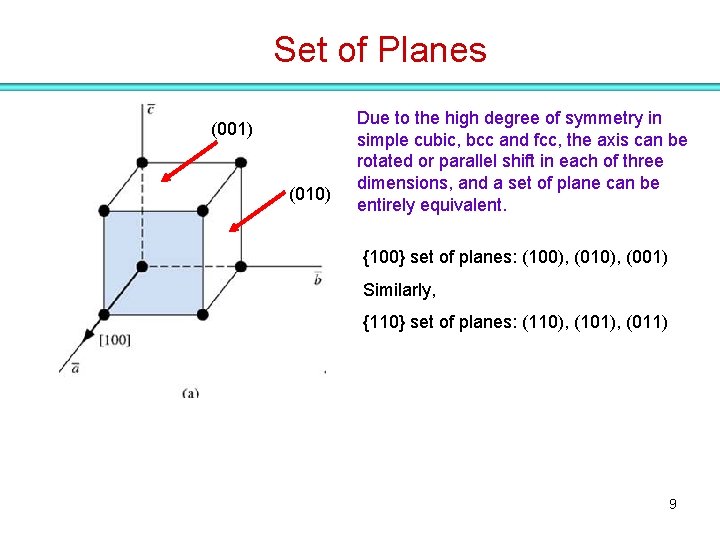
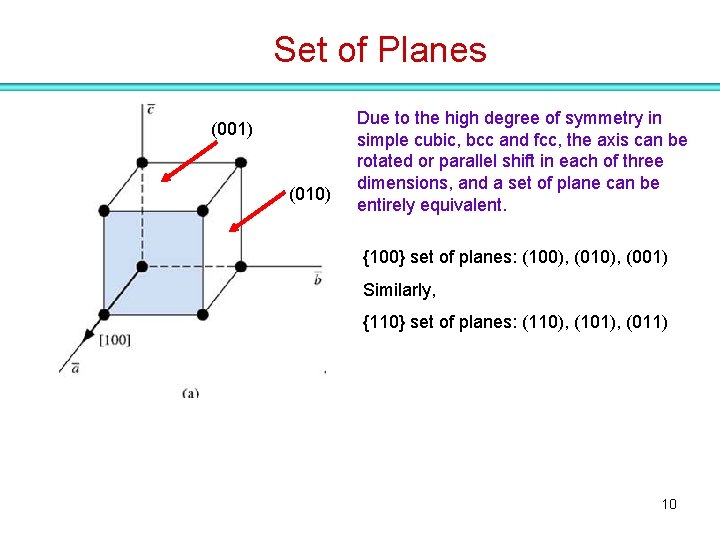
Set of Planes (001) (010) Due to the high degree of symmetry in simple cubic, bcc and fcc, the axis can be rotated or parallel shift in each of three dimensions, and a set of plane can be entirely equivalent. {100} set of planes: (100), (010), (001) Similarly, {110} set of planes: (110), (101), (011) 9

Set of Planes (001) (010) Due to the high degree of symmetry in simple cubic, bcc and fcc, the axis can be rotated or parallel shift in each of three dimensions, and a set of plane can be entirely equivalent. {100} set of planes: (100), (010), (001) Similarly, {110} set of planes: (110), (101), (011) 10

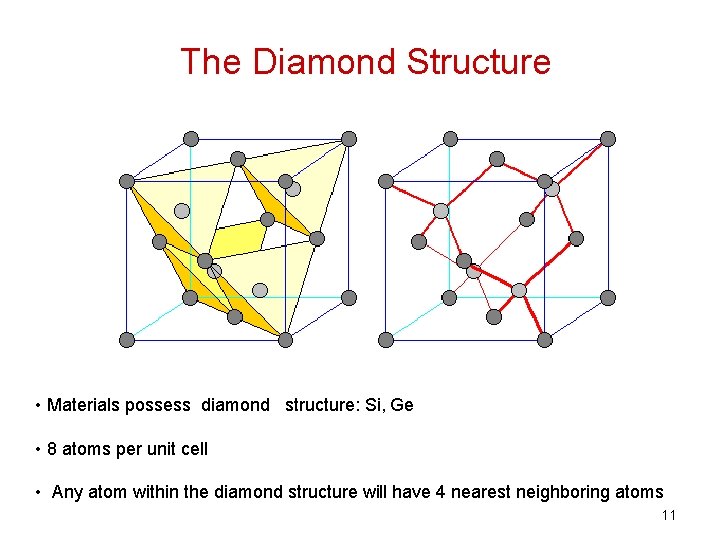
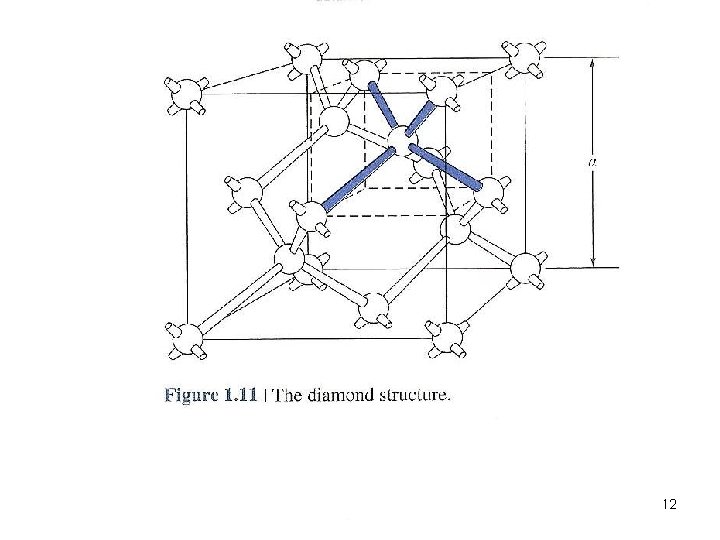
The Diamond Structure • Materials possess diamond structure: Si, Ge • 8 atoms per unit cell • Any atom within the diamond structure will have 4 nearest neighboring atoms 11

12

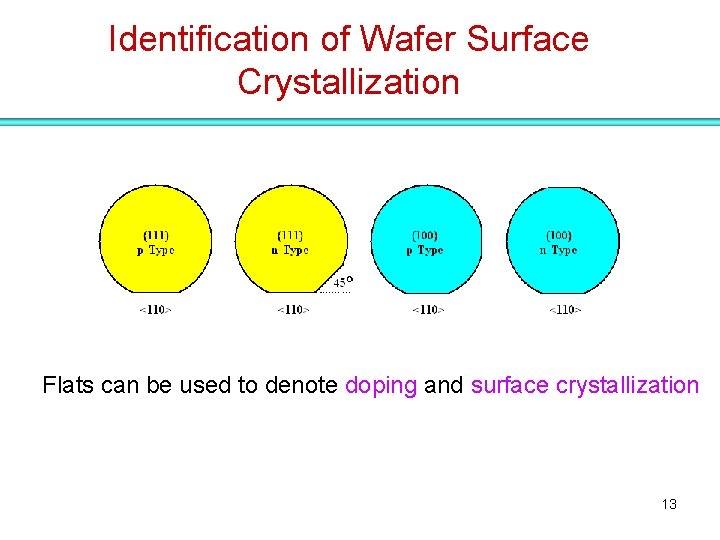
Identification of Wafer Surface Crystallization Flats can be used to denote doping and surface crystallization 13

Volume Density of Atoms • Volume density Number of atoms per unit volume = Total number of atoms / volume occupied by these atoms = number of atoms per unit cell/volume of the unit cell Unit: m-3 or (cm)-3 • Example For Silicon -8 a= 5. 43 Å = 5. 43 x 10 cm Volume density = 14

Procedure of Silicon Wafer Production Raw material ― Polysilicon nuggets purified from sand Si crystal ingot Crystal pulling A silicon wafer fabricated with microelectronic circuits Final wafer product after polishing, cleaning and inspection Slicing into Si wafers using a diamond saw 15