The Chemistry of Life What are living creatures








- Slides: 19

The Chemistry of Life What are living creatures made of? Why do we have to eat?

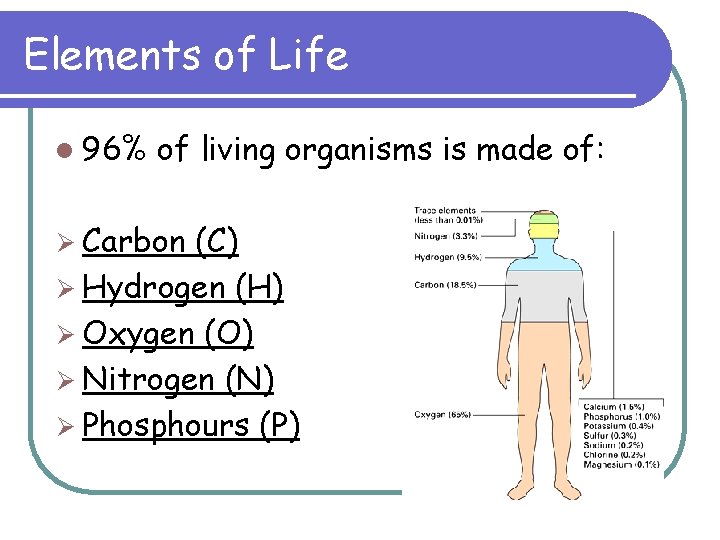
Elements of Life l 96% of living organisms is made of: Ø Carbon (C) Ø Hydrogen (H) Ø Oxygen (O) Ø Nitrogen (N) Ø Phosphours (P)

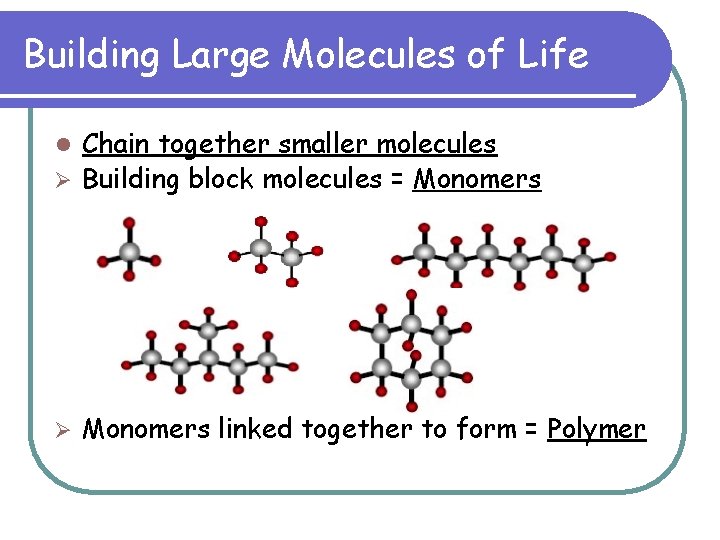
Building Large Molecules of Life Chain together smaller molecules Ø Building block molecules = Monomers l Ø Monomers linked together to form = Polymer

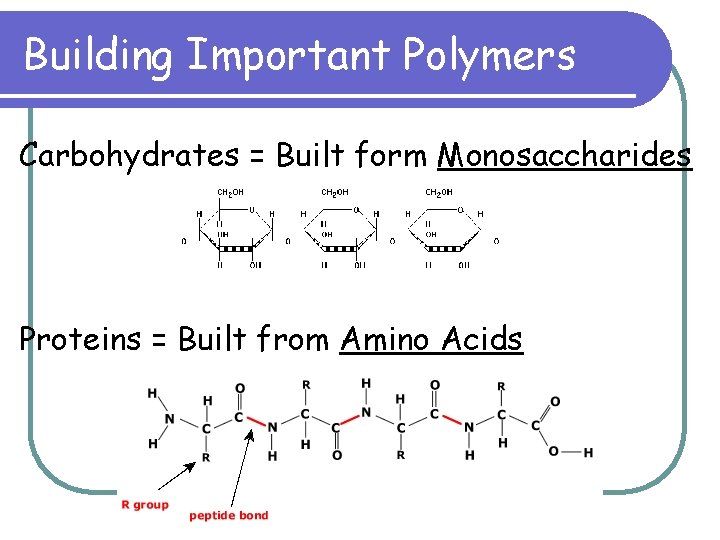
Building Important Polymers Carbohydrates = Built form Monosaccharides Proteins = Built from Amino Acids

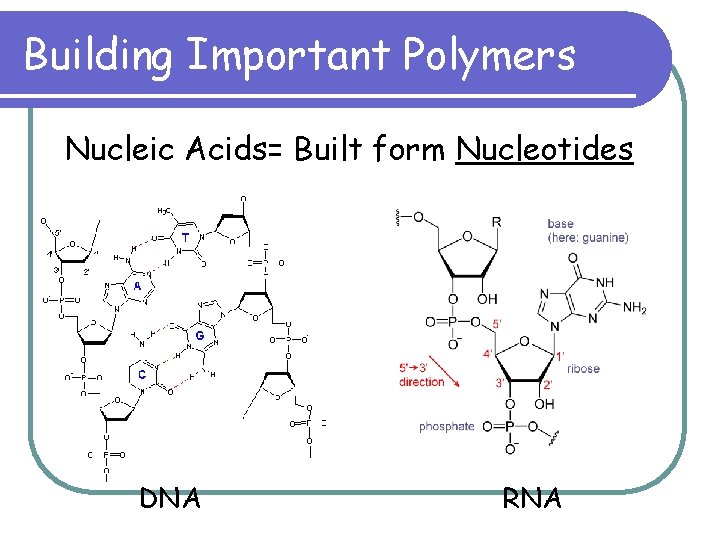
Building Important Polymers Nucleic Acids= Built form Nucleotides DNA RNA

Carbohydrates

General characteristics of Carbohydrates l Compounds composed of element C, H, and O l The ratio of carbon atoms to hydrogen atoms to oxygen atoms in every single carbohydrate is 1: 2: 1, the simplest carbohydrate has the molecular formula of CH 2 O l All sugars have the ending -ose e. g. glucose, galactose l carbohydrates are the most abundant compounds found in nature (E. g. cellulose: 100 billion tons annually)

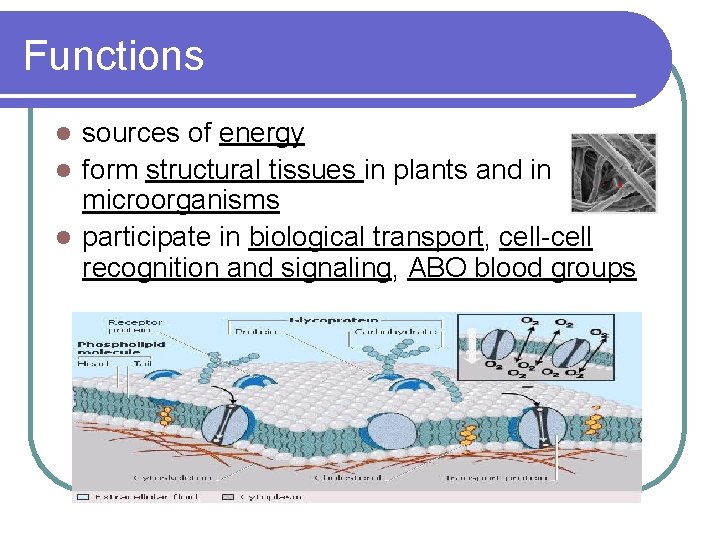
Functions sources of energy l form structural tissues in plants and in microorganisms l participate in biological transport, cell-cell recognition and signaling, ABO blood groups l

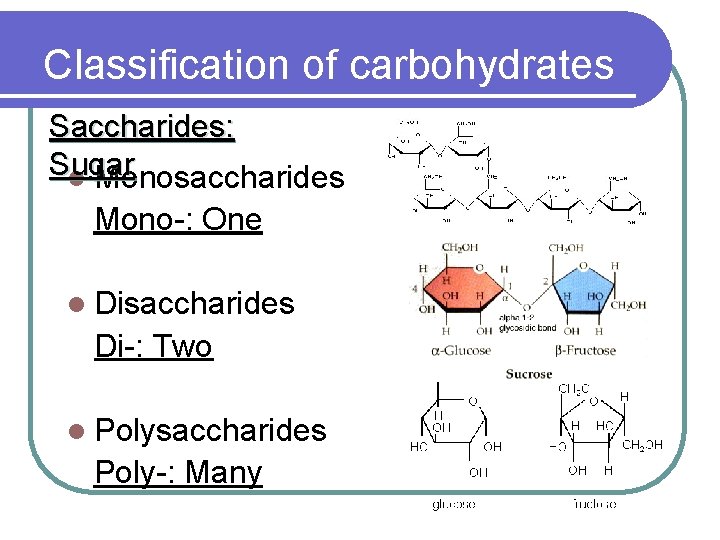
Classification of carbohydrates Saccharides: Sugar l Monosaccharides Mono-: One l Disaccharides Di-: Two l Polysaccharides Poly-: Many

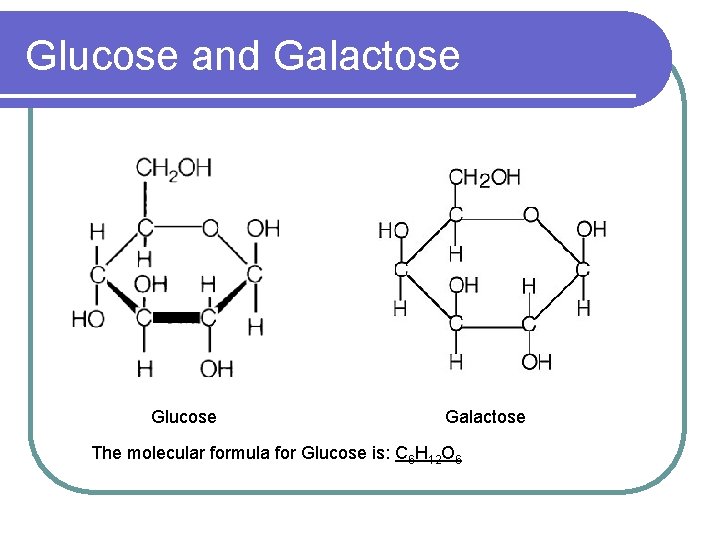
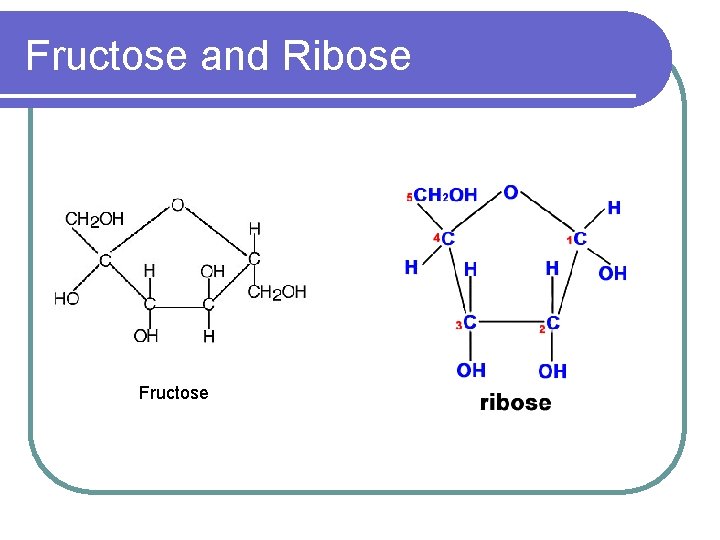
Monosaccharides l Also known as simple sugars l Basic unit of any carbohydrates l Example: Glucose, galactose, fructose, ribose

Glucose and Galactose Glucose Galactose The molecular formula for Glucose is: C 6 H 12 O 6

Fructose and Ribose Fructose

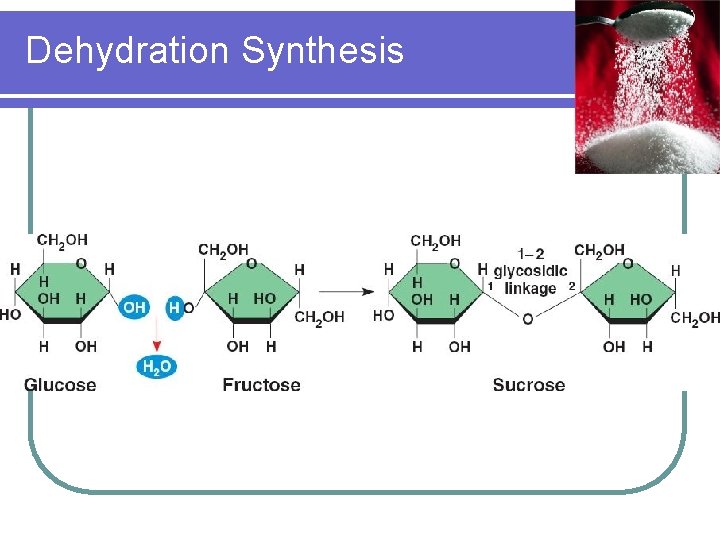
Disaccharides l Disaccharides are the products when two monosaccharides are chemically linked together with the loss of one water molecule l This chemical reaction is called dehydration synthesis l One example of disaccharides is sucrose, also known as table sugar

Dehydration Synthesis

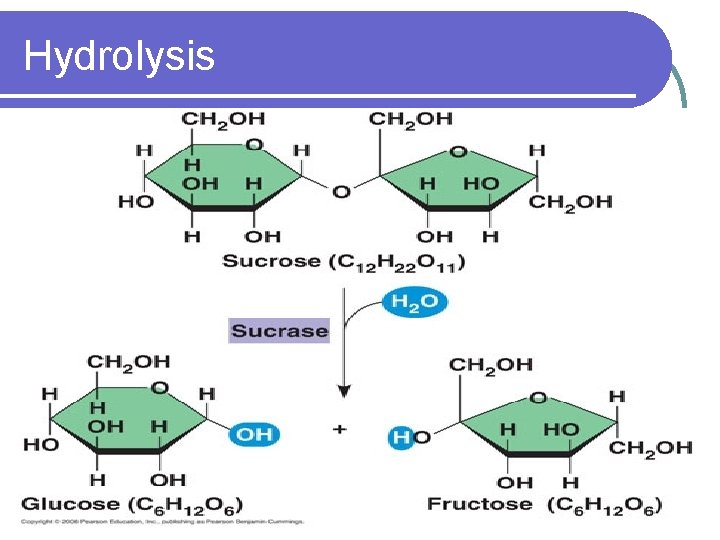
Hydrolysis l Monosaccharides can be linked through dehydration synthesis l Disaccharides and polysaccharides can be broken down through hydrolysis l Hydrolysis: The splitting of a molecule into two smaller molecules through the addition of water

Hydrolysis

Compare and Contrast l Dehydration Synthesis l Hydrolysis

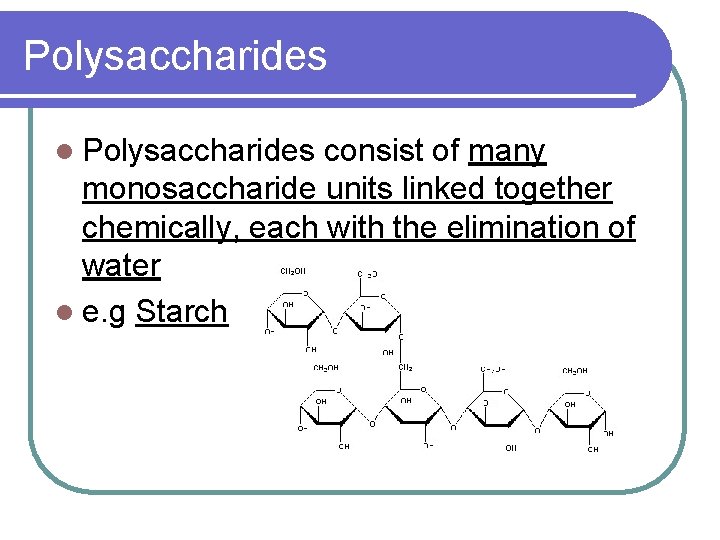
Polysaccharides l Polysaccharides consist of many monosaccharide units linked together chemically, each with the elimination of water l e. g Starch

Relative Sweetness l Fructose > Sucrose > Lactose l How do we know?