LIVING BY CHEMISTRY Unit 4 TOXINS Stoichiometry Solution

LIVING BY CHEMISTRY Unit 4: TOXINS Stoichiometry, Solution Chemistry, and Acids and Bases Chapter 15 “Toxins in Solution”

Lesson 81: Solutions Section 1: Properties of Solutions A. Solution Formation B. Rate of Dissolving
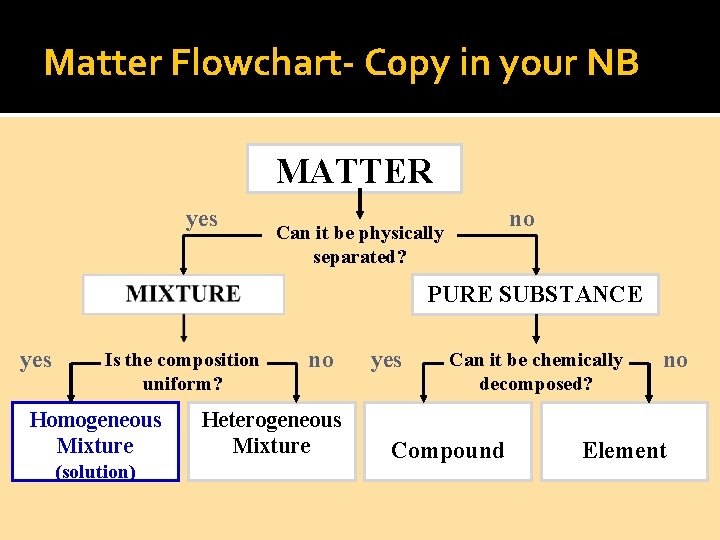
Matter Flowchart- C 0 py in your NB MATTER yes no Can it be physically separated? PURE SUBSTANCE yes Is the composition uniform? Homogeneous Mixture (solution) no Heterogeneous Mixture yes Can it be chemically decomposed? Compound no Element
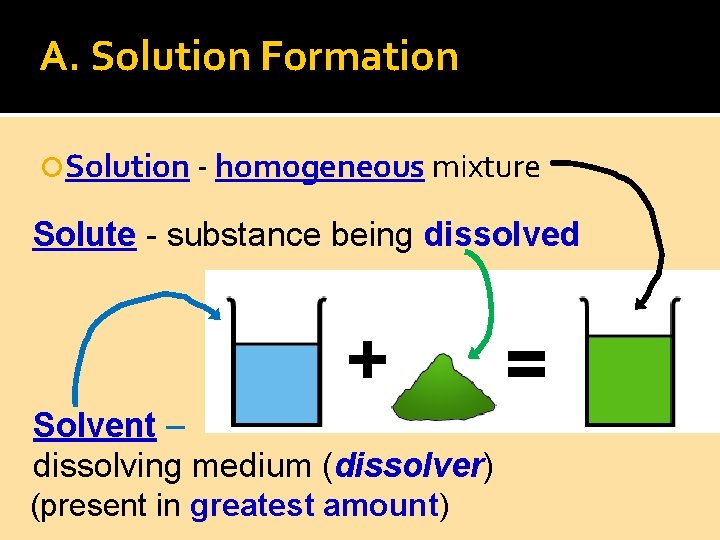
A. Solution Formation Solution - homogeneous mixture Solute - substance being dissolved Solvent – dissolving medium (dissolver) (present in greatest amount)

A. Solution Formation Types of Solutions: Solute Solvent Example Gas Air (O 2 & CO 2 in Nitrogen) Liquid Gas Water in Air Gas Liquid Soda Liquid Vinegar (acetic acid in water) Solid Liquid Salt in water Solid Stainless Steel (Cr & Ni in Iron)
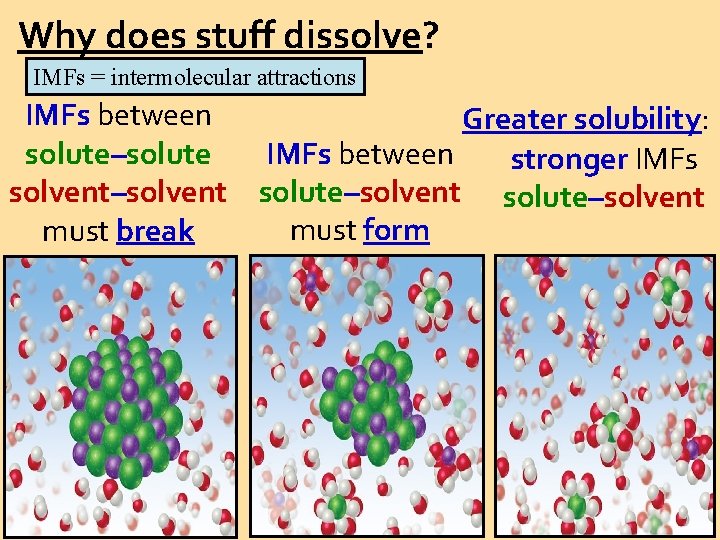
Why does stuff dissolve? IMFs = intermolecular attractions IMFs between Greater solubility: IMFs between solute–solute stronger IMFs solvent–solvent solute–solvent must form must break
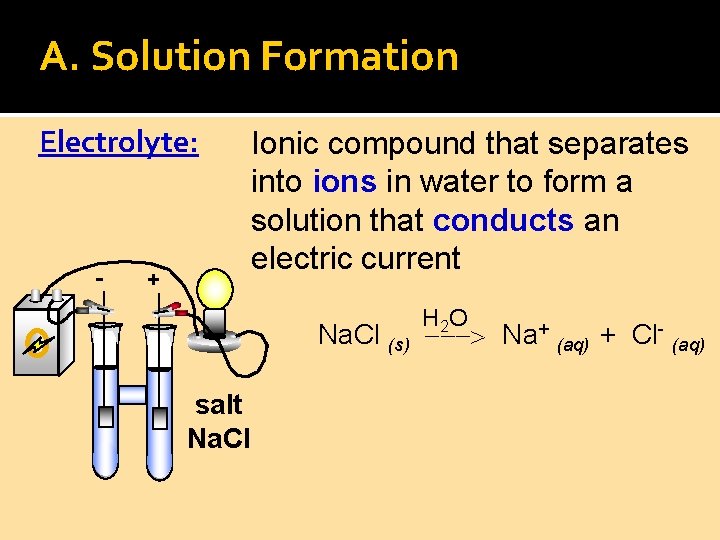
A. Solution Formation Electrolyte: - + Ionic compound that separates into ions in water to form a solution that conducts an electric current H 2 O Na. Cl (s) Na+ (aq) + Cl- (aq) salt Na. Cl
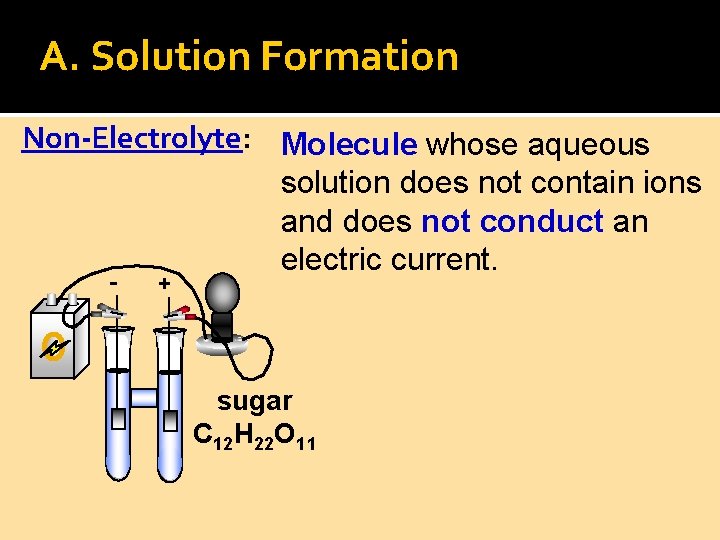
A. Solution Formation Non-Electrolyte: Molecule whose aqueous solution does not contain ions and does not conduct an electric current. - + sugar C 12 H 22 O 11
B. Rate of Dissolving Solids dissolve faster. . . more surface area (small particle size) 1 gram of sugar solute

B. Rate of Dissolving Solids dissolve faster. . . more surface area (small particle size) more stirring

B. Rate of Dissolving Solids dissolve faster. . . more surface area (small particle size) more stirring high temp.

B. Rate of Dissolving Gases dissolve faster. . . no stirring high pressure low temp.

Unit 12: Solutions Section 2: Properties of Solutions C. Solubility D. Factors Affecting Solubility
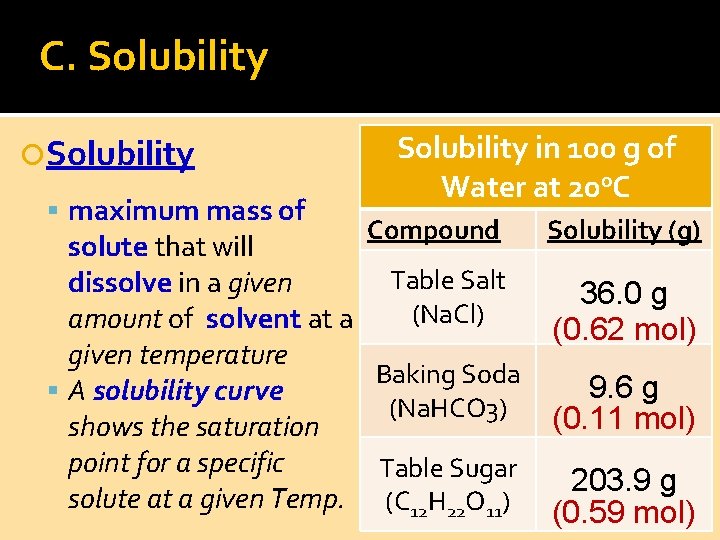
C. Solubility maximum mass of Solubility in 100 g of Water at 20 o. C Compound solute that will Table Salt dissolve in a given (Na. Cl) amount of solvent at a given temperature Baking Soda A solubility curve (Na. HCO 3) shows the saturation point for a specific Table Sugar solute at a given Temp. (C 12 H 22 O 11) Solubility (g) 36. 0 g (0. 62 mol) 9. 6 g (0. 11 mol) 203. 9 g (0. 59 mol)

Video Tutorials on Solubility Curves Video 1 Reading Solubility Curves 5: 52 min. https: //www. youtube. com/watch? v=XJm 3 t. FQG 2 FI Video 2 If you want more information 15: 09 min: https: //www. youtube. com/watch? v=6 s. J 6 k. WB_8 ag
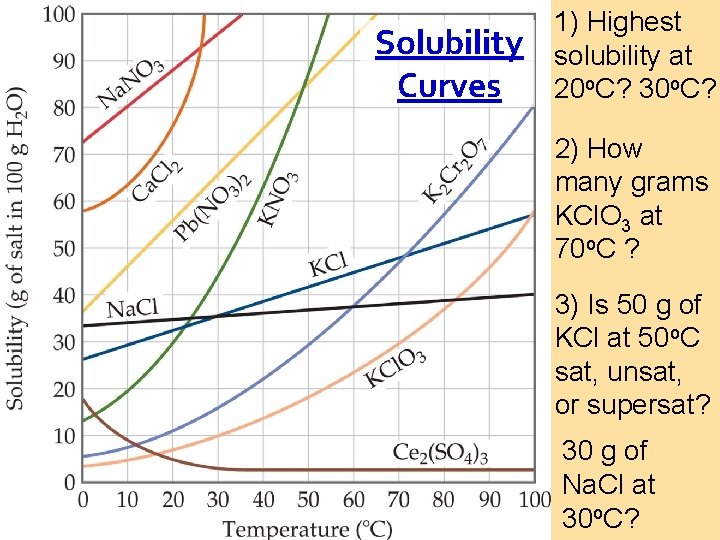
Solubility Curves 1) Highest solubility at 20 o. C? 30 o. C? 2) How many grams KCl. O 3 at 70 o. C ? 3) Is 50 g of KCl at 50 o. C sat, unsat, or supersat? 30 g of Na. Cl at 30 o. C?
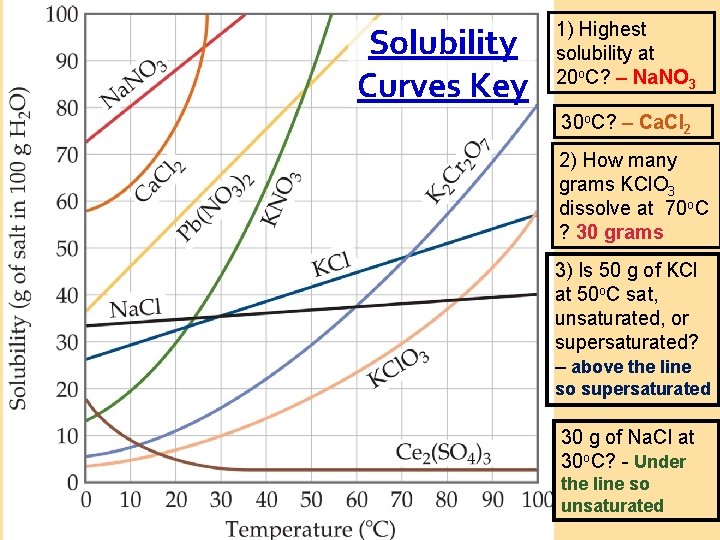
Solubility Curves Key 1) Highest solubility at 20 o. C? – Na. NO 3 30 o. C? – Ca. Cl 2 2) How many grams KCl. O 3 dissolve at 70 o. C ? 30 grams 3) Is 50 g of KCl at 50 o. C sat, unsaturated, or supersaturated? – above the line so supersaturated 30 g of Na. Cl at 30 o. C? - Under the line so unsaturated
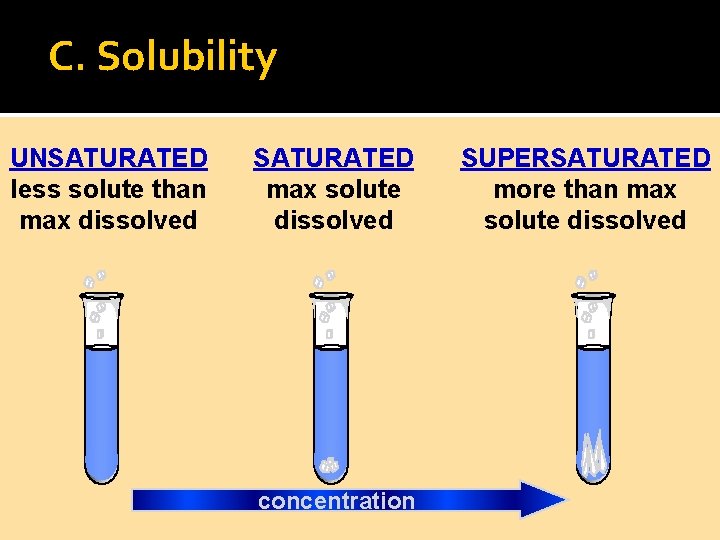
C. Solubility UNSATURATED less solute than max dissolved SATURATED max solute dissolved concentration SUPERSATURATED more than max solute dissolved

C. Solubility UNSATURATED less solute than max dissolved SATURATED max solute dissolved concentration SUPERSATURATED more than max solute dissolved Add a seed crystal

C. Solubility UNSATURATED less solute than max dissolved SATURATED max solute dissolved concentration SUPERSATURATED more than max solute dissolved

C. Solubility UNSATURATED less solute than max dissolved SATURATED max solute dissolved concentration SUPERSATURATED more than max solute dissolved Extra solute crystallizes
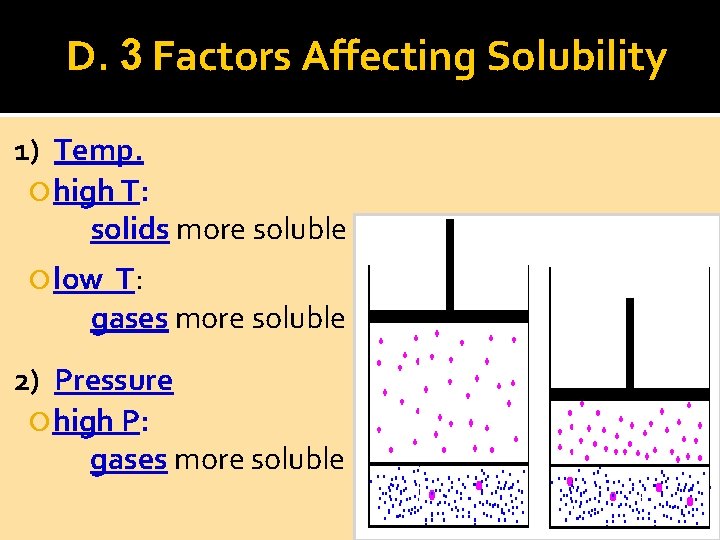
D. 3 Factors Affecting Solubility 1) Temp. high T: solids more soluble low T: gases more soluble 2) Pressure high P: gases more soluble
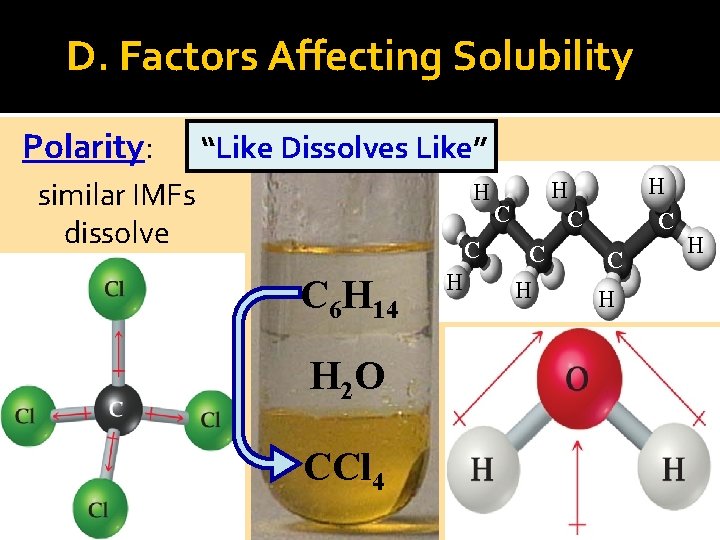
D. Factors Affecting Solubility Polarity: “Like Dissolves Like” similar IMFs dissolve H C C 6 H 14 H 2 O CCl 4 H H C C C H H

D. 3 Factors Affecting Solubility Polar –Polar –Nonpolar Immiscible Oil &Water Miscible Antifreeze & Water


Quick Quiz. 1) For a given substance, which of the following will NOT influence how fast it dissolves? A) temperature B) amount of agitation C) molar mass D) size of the crystals

Quick Quiz. 3) The solubility of a gas in a solvent is affected by? A) both temperature and pressure. B) only pressure C) only temperature D) your face

Quick Quiz. 4) Polar water molecules will NOT dissolve… A) polar alcohol molecules B) most ionic compounds C) nonpolar fat molecules D) partially ionized acetic acid molecules

Please complete WS Solutions & Solubility now in classroom
- Slides: 29