Solutions Dr Ron Rusay Spring 2003 Limestone Caves
















- Slides: 22

Solutions Dr. Ron Rusay Spring 2003

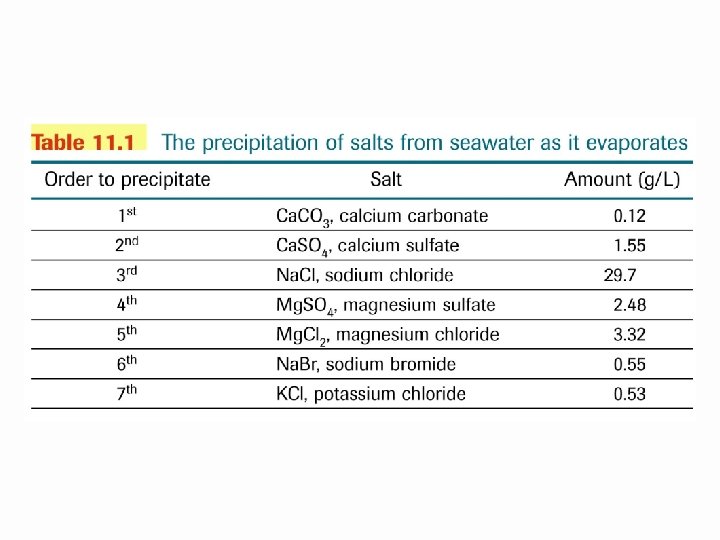
Limestone Caves: Solubility of Ca. CO 3


Solutions Substances can mix together to form homogeneous mixtures (solutions). The material present in the larger amount is the solvent and the other(s) is (are) the solute(s). Together they form a solution. The most common solutions are liquids. The solute can be a solid, liquid or gas which is dissolved in a liquid solvent. The most common solvent is water. © Copyright 1995 -2001 R. J. Rusay

DHMO, dihydromonoxide : “The Universal” Solvent http: //www. dhmo. org

Water as a Solvent The oil (nonpolar) and water (polar) mixture don’t mix and are immiscible. If liquids form a homogeneous mixture, they are miscible. Generally, likes dissolve likes, i. e. polar-polar and nonpolar-nonpolar. If polar and nonpolar mix , eg. oil and water:

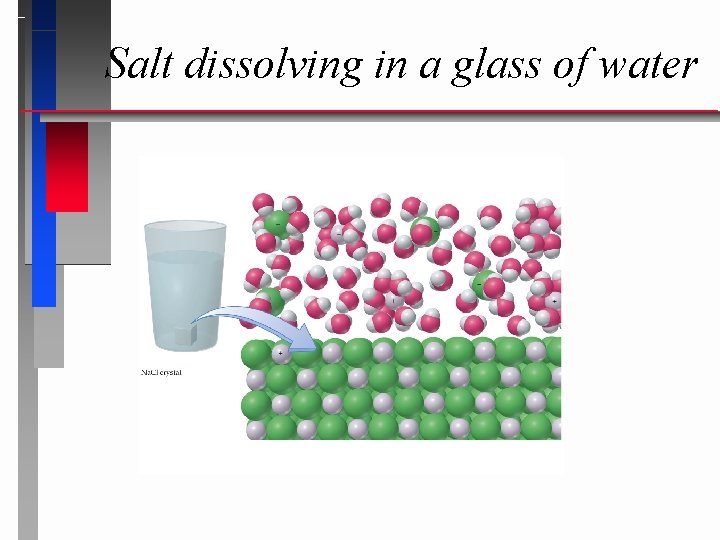
Salt dissolving in a glass of water


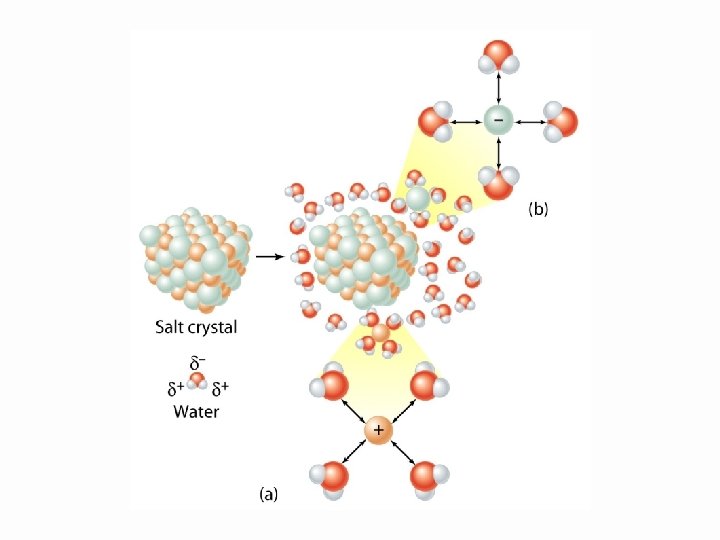
Water dissolving an ionic solid

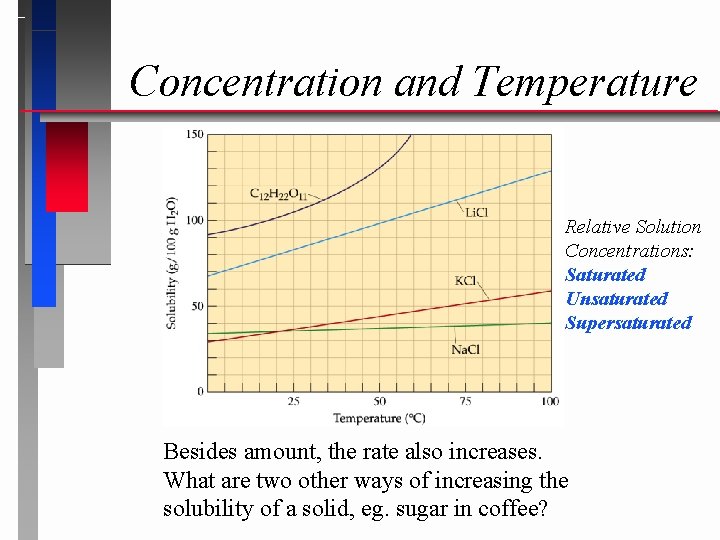
Concentration and Temperature Relative Solution Concentrations: Saturated Unsaturated Supersaturated Besides amount, the rate also increases. What are two other ways of increasing the solubility of a solid, eg. sugar in coffee?

Solution Types Solutions with less solute dissolved than is physically possible are referred to as “unsaturated”. Those with a maximum amount of solute are “saturated”. Occasionally there are extraordinary solutions that are “supersaturated” with more solute than normal.

A Giant, Single Crystal and Nuclear Energy

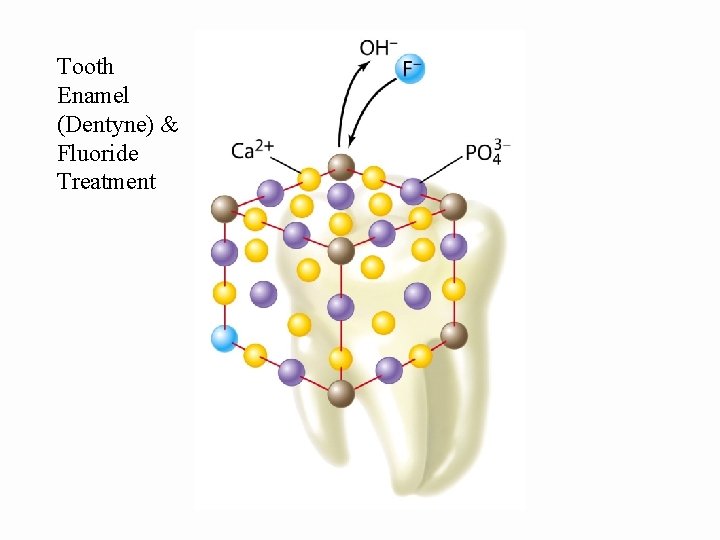
Tooth Enamel (Dentyne) & Fluoride Treatment


Gas Solubility @ P 2 = Solubility @ P 1 x [P 2 / P 1 ] P is the partial pressure of the gas vapor. Solubility units (Concentration) are usually: g / 100 ml

Preparation of Solutions

Solution Concentration A solution’s concentration is the measure of the amount of solute dissolved. Concentration is expressed in several ways. One way is mass percent. Mass % = Mass solute / [Mass solute + Mass solvent ] x 100 What is the mass % of 65. 0 g of glucose dissolved in 135 g of water? Mass % = 65. 0 g / [65. 0 + 135]g x 100 = 32. 5 % © Copyright 1995 -2001 R. J. Rusay

Solution Concentration is expressed more importantly as molarity (M). Molarity (M) = Moles solute / Liter Solution An important relationship is M x Vsolution= mol This relationship can be used directly in mass calculations of chemical reactions. What is the molarity of a solution of 1. 00 g KCl in 75. 0 m. L of solution? M = 1. 00 g KCl x 1 mol KCl / 74. 55 g KCl x 1/ 75 m. L x 1000 m. L / L = 0. 18 mol / L © Copyright 1995 -2001 R. J. Rusay

Acid-Base Titration

Solution Applications Neutralization-Titration 15. 50 m. L of vinegar, a solution of acetic acid (aa), required 20. 00 m. L of a 0. 3000 M (mol/L) solution of a sodium hydroxide solution to react completely. Maa = ? HC 2 H 3 O 2 (aq) + Na. OH(aq) ? + ? HC 2 H 3 O 2 (aq) + Na. OH(aq) 1 Na. C 2 H 3 O 2 (aq) + 1 H 2 O (l) ? Maa = [MNa. OHx ? Maa = ? Maa VNa. OH / VHC 2 H 3 O 2 ] [? mol. HC 2 H 3 O 2 / ? mol. Na. OH] x 0. 02000 LNa. OH x 1 mol. HC 2 H 3 O 2 LNa. OH x 0. 01550 LHC 2 H 3 O 2 x 1 mol. Na. OH 0. 3000 mol. Na. OH = 0. 3870 mol. HC 2 H 3 O 2 / LHC 2 H 3 O 2 = 0. 3870 MHC 2 H 3 O 2

Solution Applications http: //ep. llnl. gov/msds/Chem 106/vinegar. html What is the weight percent of the acetic acid (aa)? (Density of vinegar = 1. 006 g/m. L; Molar Mass (aa) = MW (aa) in grams/ mole aa = 60. 056 g aa/mole aa) � (Molarity aa x Volume aa) x (60. 056 g aa /mole aa) � = mass aa = 0. 360 gaa [(mass of Acetic Acid) / (mass of vinegar) ] * 100% = % Acetic Acid � mass of vinegar = density x sample volume = 15. 59 g � % = 0. 360 gaa / 15. 59 g * 100 = 2. 3%

Solution Dilution