LIME Lime is obtained from limestone Pure limestone

LIME

Lime is obtained from limestone. Ø Pure limestone → Ca. CO 3 Ø Impurities → Mg. CO 3, Al 2 O 3, Fe 2 O 3, Si. O 2 Ø

PRODUCTION STEPS Excavation of limestone Ø Crushing Ø Grinding Ø Calcination → Quicklime Ø Pulverize Quicklime Ø Mixed with water under pressure → Slaked Lime Ø Drying of Slaked Lime Ø Pulverizing Ø Marketing in bags. Ø

CALCINATION Ø Ca. CO 3 → Ca. O + CO 2 (>900°C) Quick lime Calcination is carried out in kilns n Intermittent n Continuous n Rotary n Reactor Ø
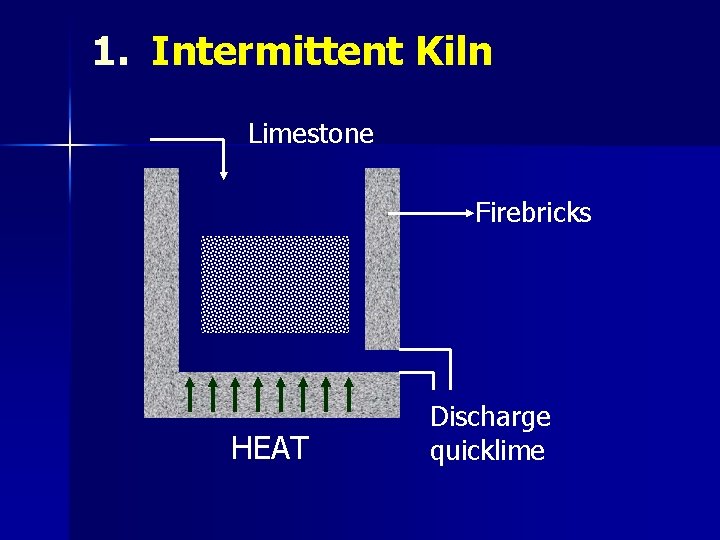
1. Intermittent Kiln Limestone Firebricks HEAT Discharge quicklime
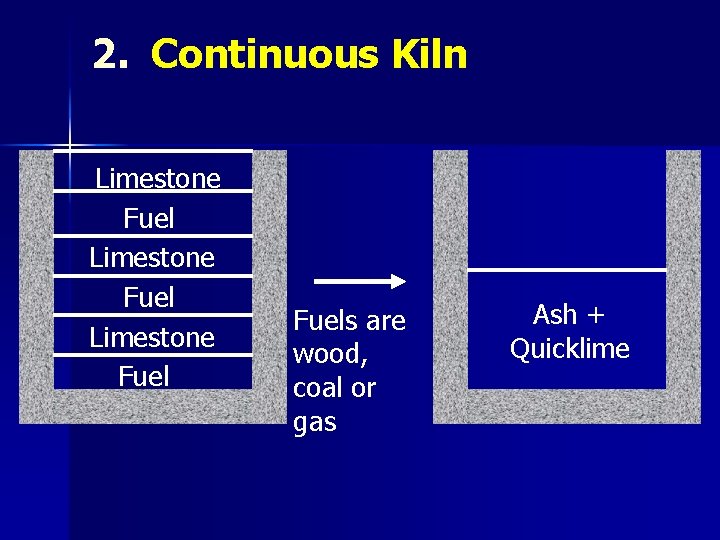
2. Continuous Kiln Limestone Fuels are wood, coal or gas Ash + Quicklime
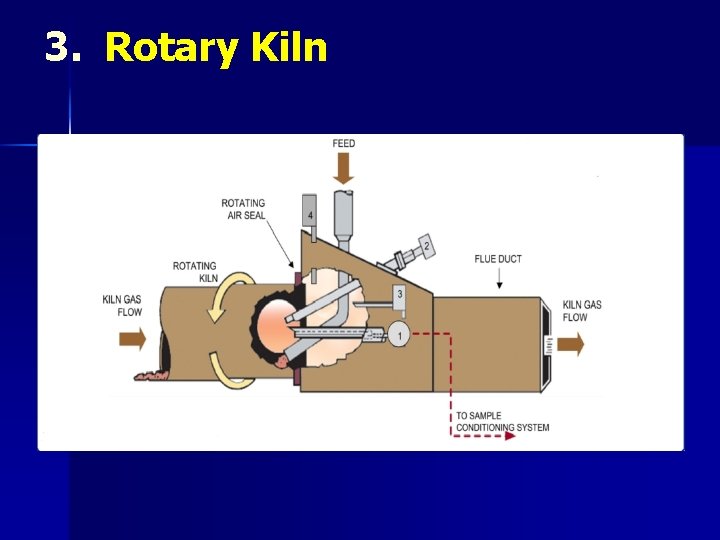
3. Rotary Kiln
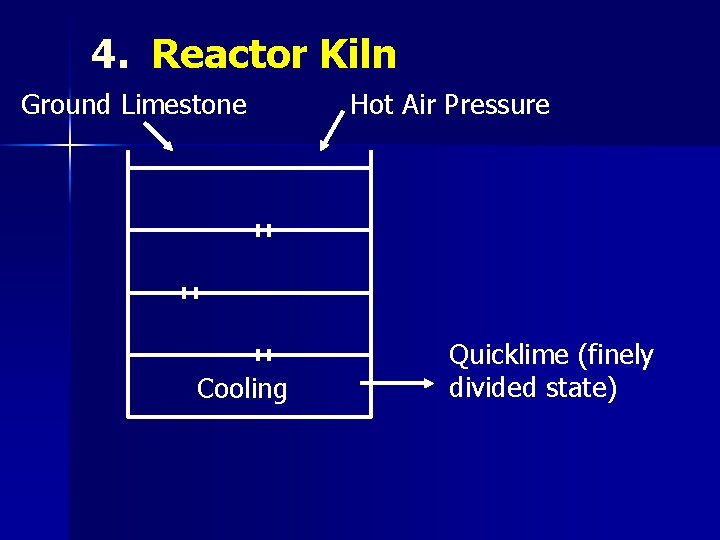
4. Reactor Kiln Ground Limestone Cooling Hot Air Pressure Quicklime (finely divided state)

CLASSIFICATION OF QUICKLIME 1. n n n According to Particle Size Lump Lime (10 -30 cm lumps) Pebble Lime (2 -5 cm) Granular Lime (~0. 5 cm) Crushed Lime (~5 -8 mm) Ground Lime (passes #10 sieve, by grinding crushed lime) Pulverized Lime (passes #100 sieve)

2. n n 3. n n According to Chemical Composition High-Calcium Quicklimes (~90% of Ca. O) Calcium Quicklime (75% of Ca. O) Magnesian Quicklime (at least 20% of Mg. O) Dolomitic Quicklime (at least 25% of Mg. O) According to Intended Use Mortar Lime Plaster Lime

SLAKING (HYDRATION) OF LIME Ø Ca. O + H 2 O → Ca(OH)2 + Heat Ø Ca. O is mixed with water in a slaking box until a “putty” has been formed. Ø The putty is then covered with sand to protect it from the action of the air & left for seasoning. Ø Time of seasoning → 1 week for mortar use 6 weeks for plaster use

Ø If Ca. O is not slaked well, it will absorb moisture from air & since the volume expands upto 2. 5 -3 times popouts will occur. Ø The slaked lime can also be bought from a factory. It is more homogeneous & economical but less plastic. Ø Seasoning provides a homogeneous mass & completion of chemical reactions Ø During slaking heat evolves & volume expands.

HARDENING OF SLAKED LIME Ø Ca (OH)2 + CO 2 → Ca. CO 3 + H 2 O AIR SLAKED LIME When quicklime (Ca. O) is left uncovered it picks up moisture and CO 2 from air & becomes partly Ca. CO 3. Ø Ca. O + H 2 O → Ca(OH)2 Ø Ca(OH)2 + CO 2 → Ca. CO 3 + H 2 O Ø

LIME POPS Ø If quicklime is not mixed completely with water, some Ca. O will be carried to construction stage. Ø In its final stage it will absorb water & CO 2 from air and will expand upto 2. 5 -3 times. Ø This will cause cracking & pop-ups in the structure.

PROPERTIES OF LIME MORTARS Ø By adding sand to lime, lime mortar is obtained. Ø Adjusting plasticity Ø Gaining economy Ø Decreasing shrinkage effects

STRENGTH OF LIME MORTARS Chemical composition of lime Magnesian Limes > Calcium Limes Ø Sand amount & properties Adding sand decreases strength Ø Amount of water Voids are formed after evaporation Ø Setting conditions Lower humidity & higher CO 2 higher strength Ø

PROPERTIES OF HIGH CALCIUM LIMES Ø Slakes faster Ø Hardens faster Ø Have greater sand carrying capacity

USES OF LIME Ø In producing masonry mortars Ø Plaster mortars Ø White-wash Ø In production of masonry blocks

HYDRAULIC LIME Ø A lime obtained by calcination of siliceous or clayey limestone at higher temperature Ø It differs from quicklime: n Burned at higher temperature n It contains lime silicates n It can set & harden under water
- Slides: 19