What is limestone Limestone a biochemical sedimentary rock

What is limestone? Limestone = a biochemical sedimentary rock made up mostly of calcium carbonate Mineral Crystal system Formula Remarks Calcite Rhombohedral Ca. CO 3 Dominant limestone mineral, especially in rocks older than Cenozoic Aragonite Orthorhombic Ca. CO 3 Dominant mineral in Recent carbonate sediments; alters readily to calcite

How do limestones form? Most limestones are simply the cemented remains of marine shells Limestone “anatomy” n Grains w Skeletal particles, ooids, peloids n Lime mud w Microscopic crystals produced by calcareous algae and through abrasion of larger particles n Cement w Inorganically precipitated Ca. CO 3 crystals

Skeletal grains
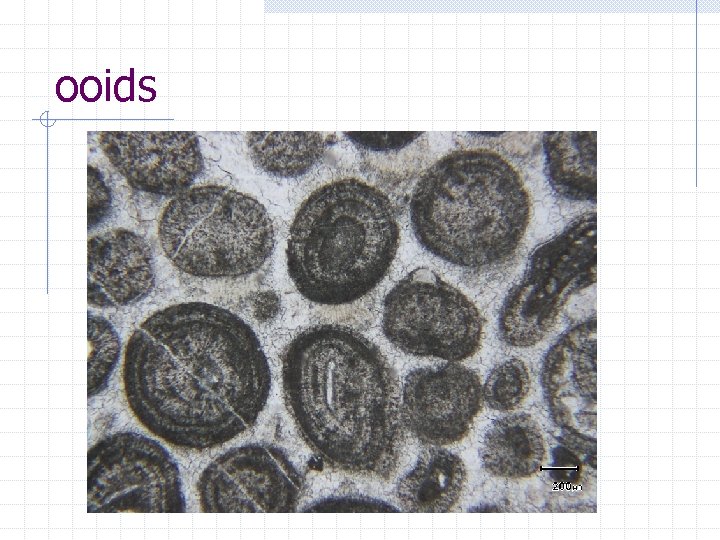
ooids
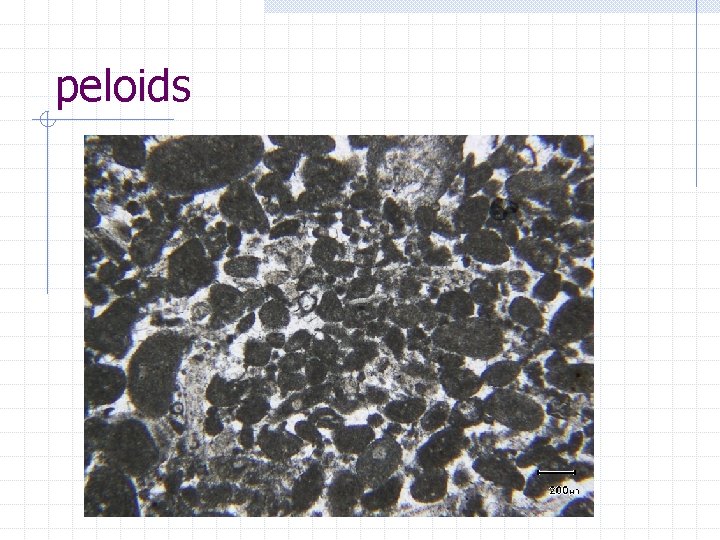
peloids

Lime mud
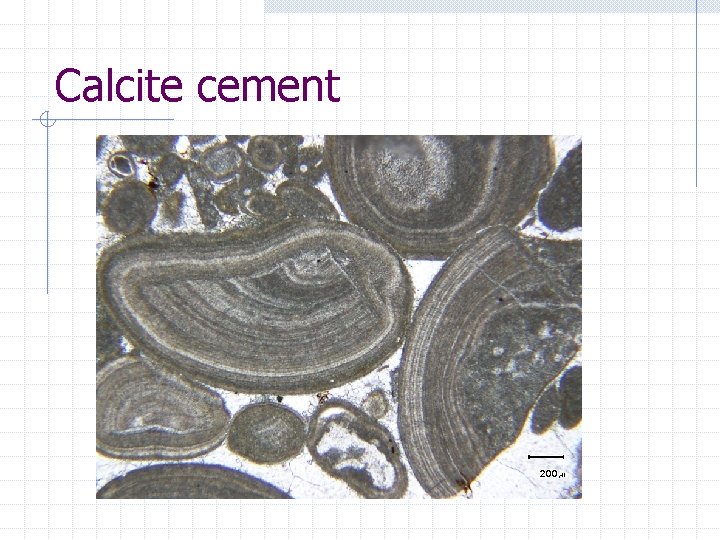
Calcite cement
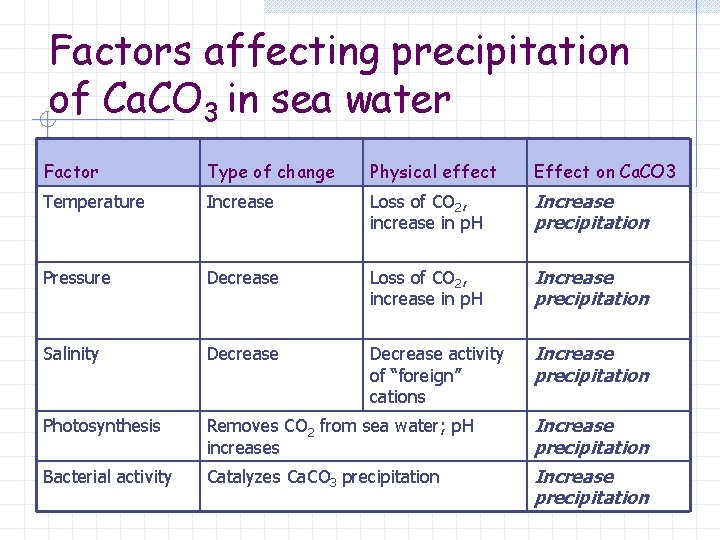
Factors affecting precipitation of Ca. CO 3 in sea water Factor Type of change Physical effect Effect on Ca. CO 3 Temperature Increase Loss of CO 2, increase in p. H Increase precipitation Pressure Decrease Loss of CO 2, increase in p. H Increase precipitation Salinity Decrease activity of “foreign” cations Increase precipitation Photosynthesis Removes CO 2 from sea water; p. H increases Increase precipitation Bacterial activity Catalyzes Ca. CO 3 precipitation Increase precipitation

Where do limestones form? Because Ca. CO 3 precipitates most readily in warm, well lit, agitated water of normal marine salinity…. . most limestones form in shallow, tropical depositional environments n e. g. , Bahamas, central America, Persian Gulf, NW shelf of Australia, Great Barrier Reef, Malaysia, Indonesia, etc.
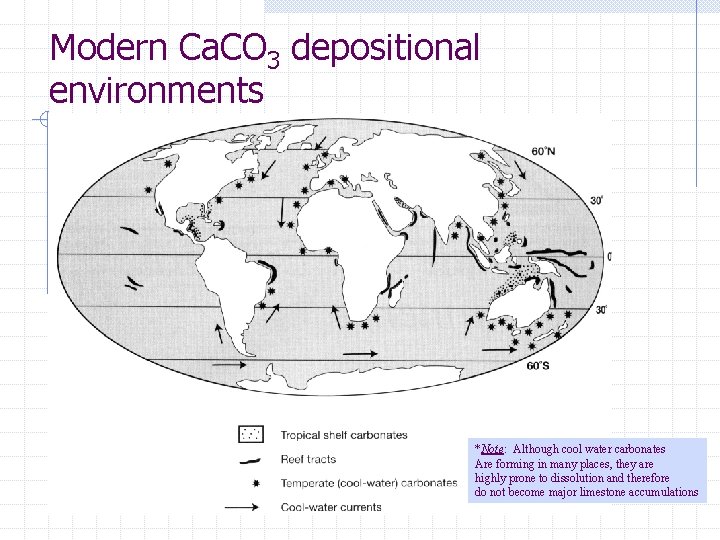
Modern Ca. CO 3 depositional environments *Note: Although cool water carbonates Are forming in many places, they are highly prone to dissolution and therefore do not become major limestone accumulations
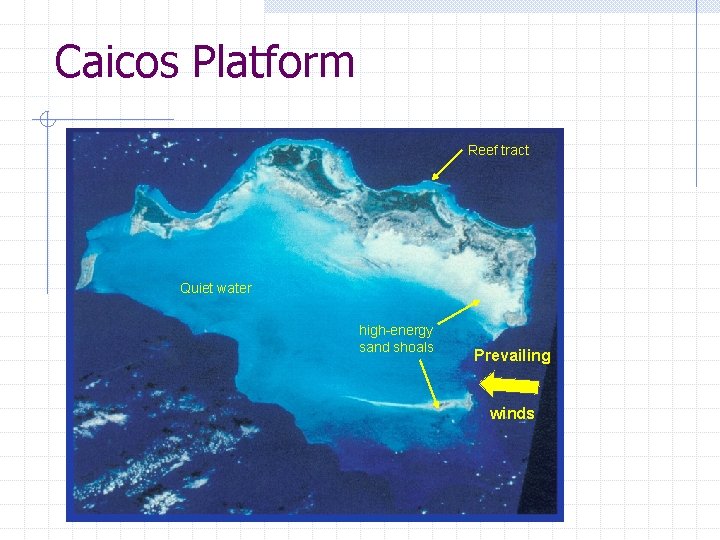
Caicos Platform Reef tract Quiet water high-energy sand shoals Prevailing winds
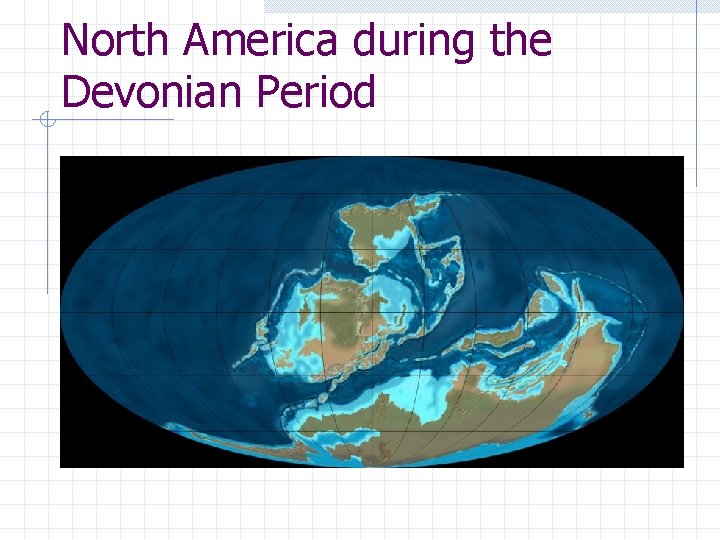
North America during the Devonian Period
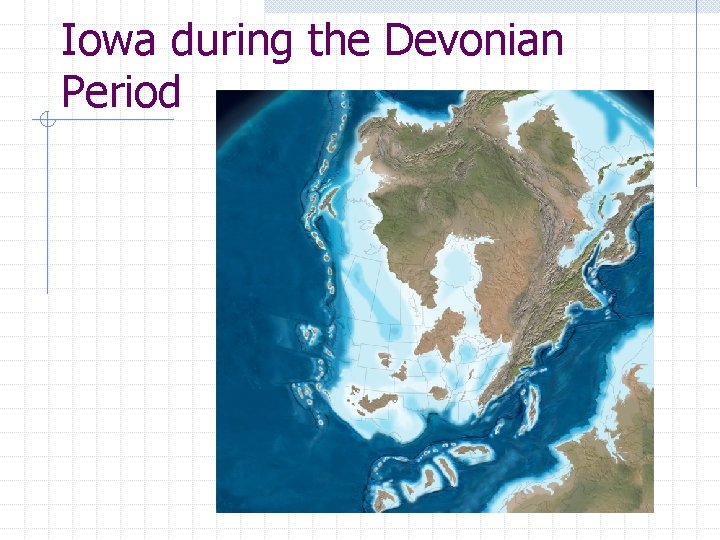
Iowa during the Devonian Period

What are limestone products? Whole rock n n Crushed limestone Dolomitic limestone Burned lime (calcium oxide) n n High calcium lime Dolomitic lime Hydrated lime (calcium hydroxide)

How is limestone used? Construction Soil stabilization Flue Gas Desulfurization Steel Production (flux for blast furnaces) Glass Production Water Treatment Waste Treatment Paper Production (filler) Chemical Production Masonry, Mortars and Other Building Materials
- Slides: 15