Bonding Molecular Shapes Dr Ron Rusay Spring 2003

Bonding & Molecular Shapes Dr. Ron Rusay Spring 2003 © Copyright 2003 R. J. Rusay

Chemical Bonds Definition: Attractive forces which hold atoms together and provide a particular molecular arrangement of atoms with new chemical properties.
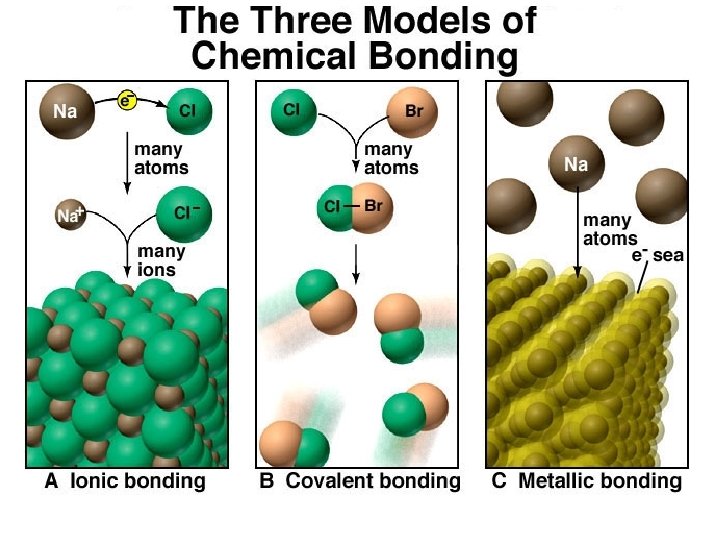
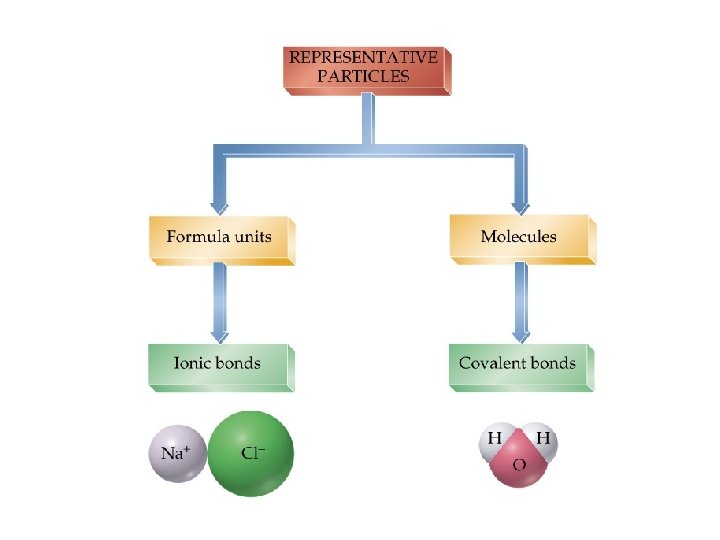

Ionic vs. Covalent Bonding

Electron Configurations Noble Gases and The Rule of Eight ÛA nonmetal and a metal react to form an ionic compound: Valence electrons of the metal are lost and the nonmetal gains these electrons. (Ionic Bonding) Û When two nonmetals react: They share electrons to achieve a Noble Gas Configuration. (Covalent Bonding)

Octet Rule: General Comments 2 nd row elements C, N, O, F observe the octet rule. 2 nd row elements B and Be often have fewer than 8 electrons around themselves - they are very reactive. 3 rd row and heavier elements CAN exceed the octet rule using empty valence d orbitals. When writing Lewis structures, satisfy octets first, then place electrons around elements having available d orbitals.

Lewis Electron-Dot Symbols for Elements in Periods 2 & 3
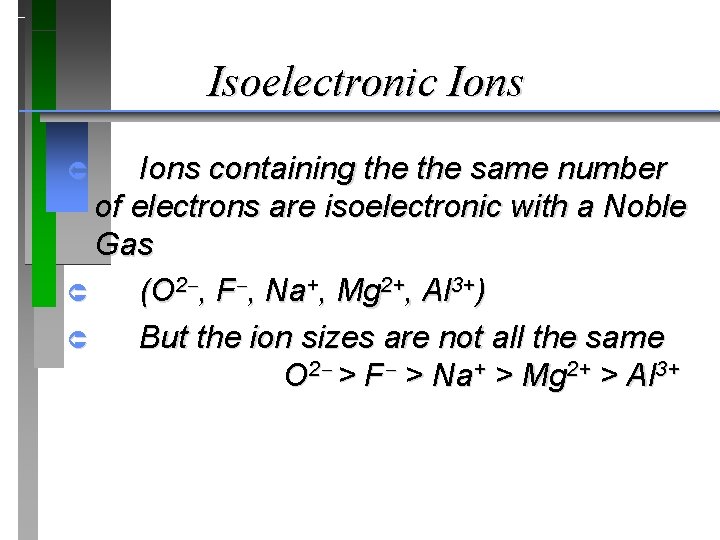
Isoelectronic Ions containing the same number of electrons are isoelectronic with a Noble Gas Û (O 2 , F , Na+, Mg 2+, Al 3+) Û But the ion sizes are not all the same O 2 > F > Na+ > Mg 2+ > Al 3+ Û

Ionic Bonds Result from electrostatic attractions of closely packed, oppositely charged ions. Form when an atom which can easily lose electrons reacts with one which has a high electron affinity, that is, it can easily gain electrons. Mg and Cl; K and O

Covalent Bond Lengths Interatomic distance. Û It is the distance where the bond energy is at a minimum value, and which is the most stable atomic form. Û

Covalent Bonding & Bond Length
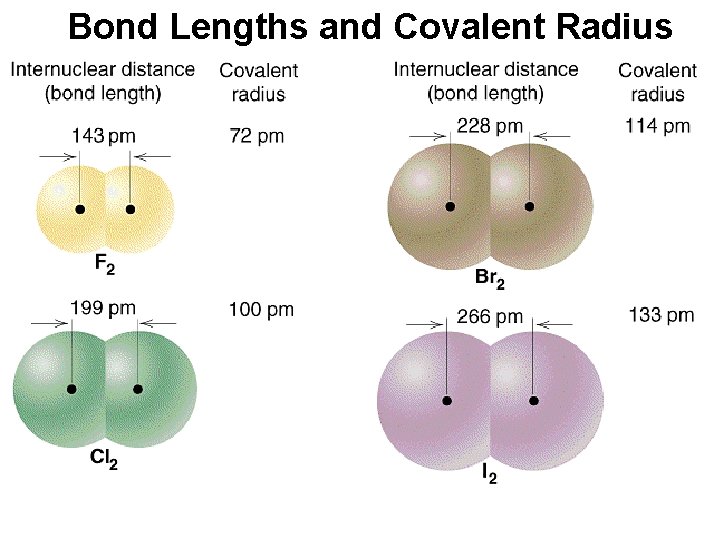
Bond Lengths and Covalent Radius

The Periodic Table Bond Lengths : Bond Strengths


Actual vs. Expected Bond Length Actual = 0. 127 nm Expected = 0. 136 nm

Electronegativity

Electronegativity Û The ability of an atom in a molecule to attract shared electrons to itself. = (H X)actual (H X)expected
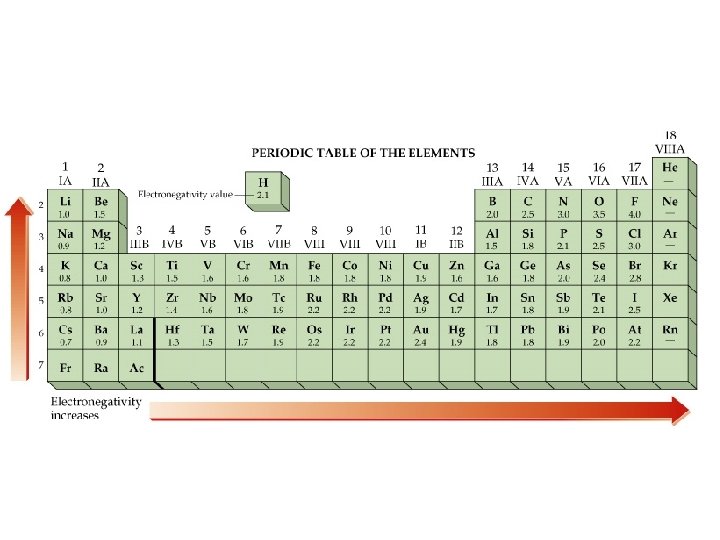

Periodic Trend

Electronegativity Differences & Polar Covalent Bonds Û A molecule with a relatively high difference in electronegativities, such as HF, has a center of positive charge and a center of negative charge. It is polar, having an experimentally measureable dipole moment.
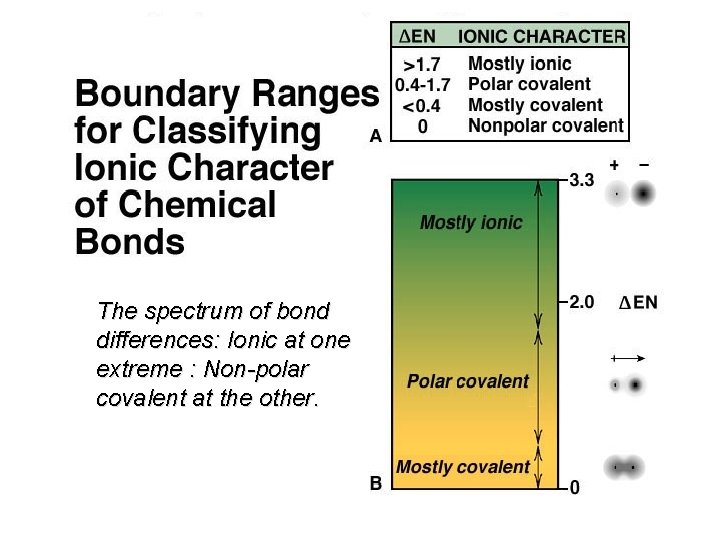
The spectrum of bond differences: Ionic at one extreme : Non-polar covalent at the other.

Bond Shapes

Lewis Structure Shows how valence electrons are arranged among atoms in a molecule. Reflects central idea that stability of a compound relates to noble gas electron configuration.
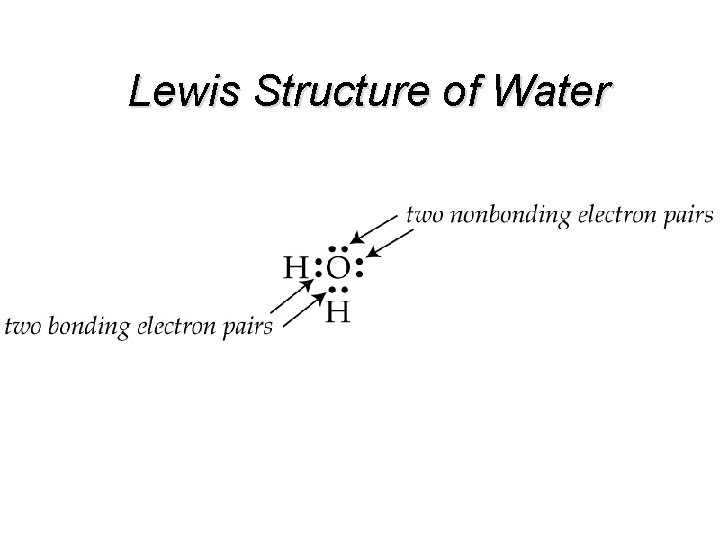
Lewis Structure of Water
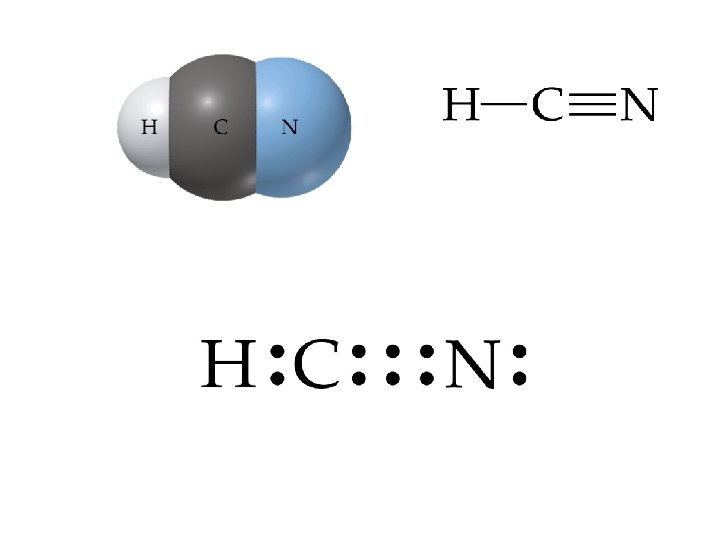


Fundamental Bonding Patterns Û Carbon • • • has a total of four bonds: 4 single bonds 2 single bonds plus 1 double bond 1 single bond plus 1 triple bond Û Oxygen has a total of 2 bonds plus 2 “free pairs of electrons”: • 2 single bonds • 1 double bond

Fundamental Bonding Patterns Û Nitrogen has a total of three bonds plus 1 “free pair of electrons”: • • • 3 single bonds 1 single bonds plus 1 double bond 1 triple bond Û Hydrogen only has 1 single bond. Û This is the total of almost all bonding arrangements in organic molecules.

Lewis Structures of Simple Molecules H N N C O . . H H Ammonia H . . . N. H Urea H

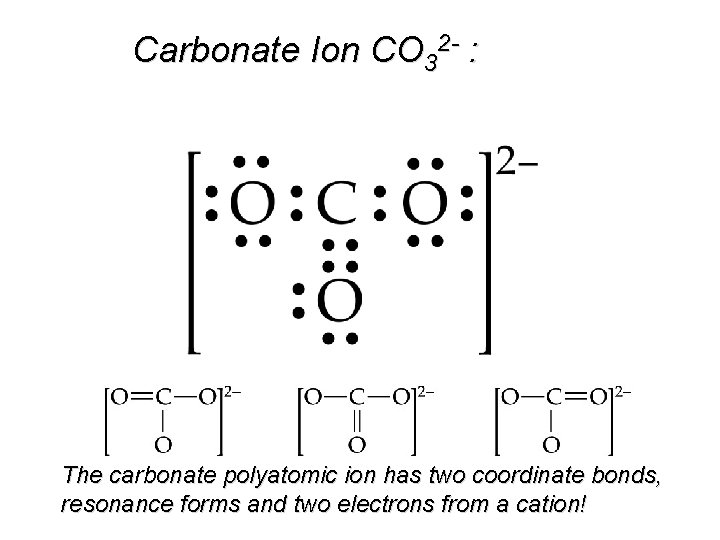
Carbonate Ion CO 32 - : The carbonate polyatomic ion has two coordinate bonds, resonance forms and two electrons from a cation!
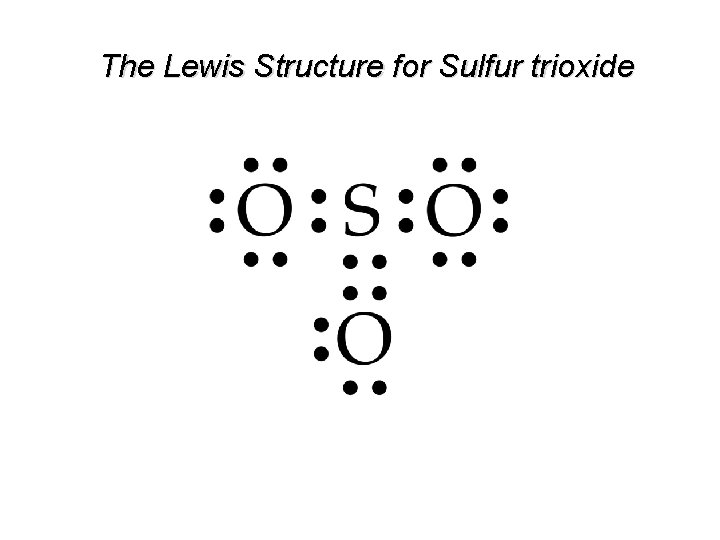
The Lewis Structure for Sulfur trioxide
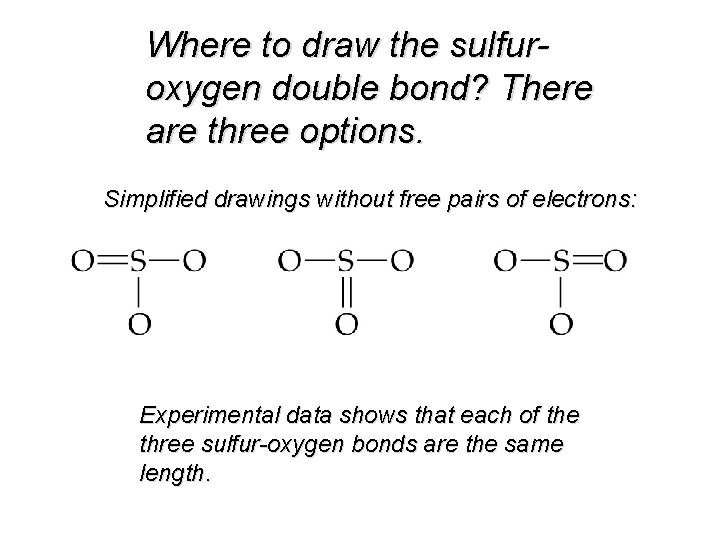
Where to draw the sulfuroxygen double bond? There are three options. Simplified drawings without free pairs of electrons: Experimental data shows that each of the three sulfur-oxygen bonds are the same length.

Resonance Û Occurs when more than one valid Lewis structure can be written for a particular molecule. Û These are resonance structures. The actual structure is an average of the resonance structures.
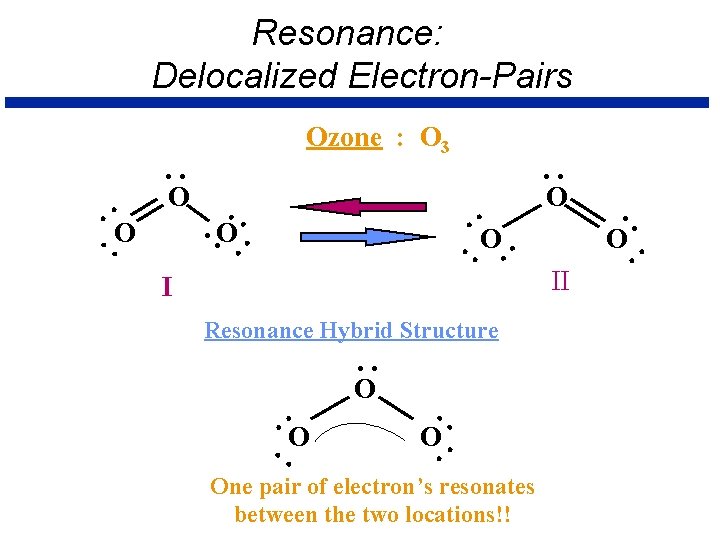
Resonance: Delocalized Electron-Pairs . O. I O O . . Resonance Hybrid Structure . . . O. . One pair of electron’s resonates between the two locations!! . . . . O. . . Ozone : O 3 II
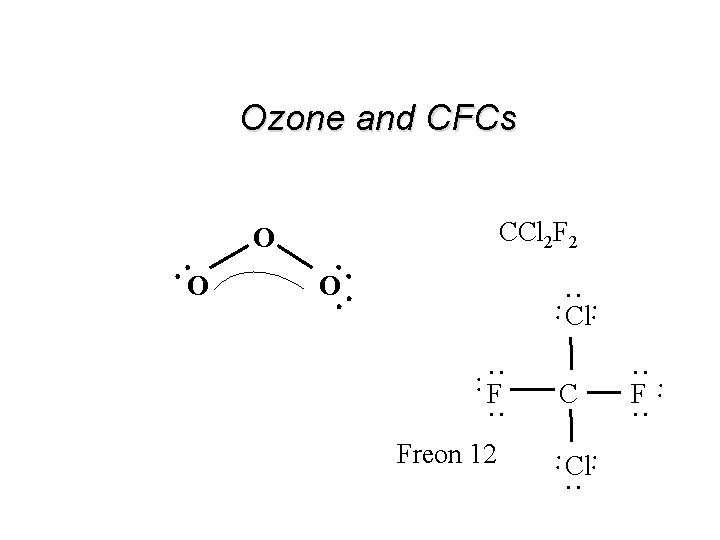
Ozone and CFCs CCl 2 F 2 . . O. . . . Cl Freon 12 Cl. . F. . C . . . . F . . O O

VSEPR Model Valence Shell Electron Pair Repulsion

VSEPR Model ÛThe molecular structure which surrounds a given atom is determined principally by minimizing electron pair repulsions through maximizing separations.

Molecular Models Û Computer Generated Models Ball and stick models of ammonia, water and methane. For many others see: http: //ep. llnl. gov/msds/pdb/ http: //ep. llnl. gov/msds/orgchem/Chem 226/Smell-Stereochem. html

- Slides: 41