Limestone Limestone calcium carbonate WALT Describe what limestone

Limestone

Limestone (calcium carbonate) WALT: Describe what limestone is and what happens when it reacts with acids


What is limestone? Limestone is a rock that is largely the chemical compound calcium carbonate, Ca. CO 3. It is formed mainly from the remains of organisms that lived in ancient seas millions of years ago. Limestone is not just used for building. It can be processed into many useful chemicals. When it rains, acid in the rain can react with the limestone, wearing the rock away and releasing carbon dioxide.
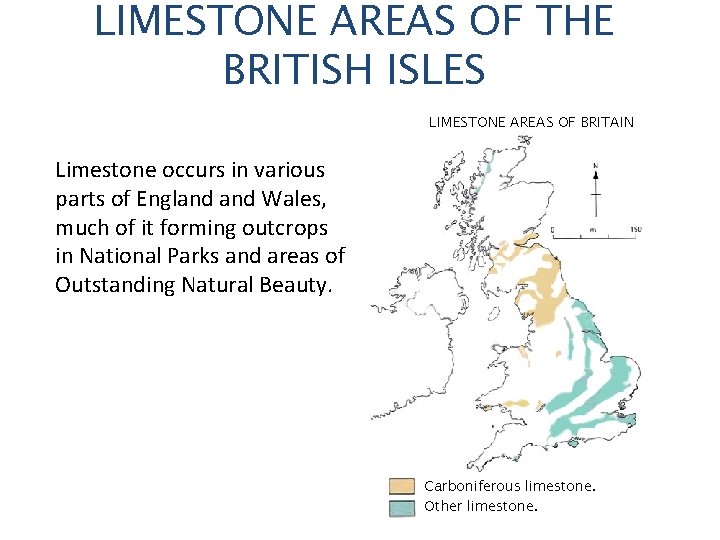
LIMESTONE AREAS OF THE BRITISH ISLES LIMESTONE AREAS OF BRITAIN Limestone occurs in various parts of England Wales, much of it forming outcrops in National Parks and areas of Outstanding Natural Beauty. Carboniferous limestone. Other limestone.


Limestone is taken out of the ground by quarrying.

Marble chips are mainly calcium carbonate • From Term 1, what did we learn about what happens when marble chips react with hydrochloric acid? What gas was given off? • What do we already know about acid rain?

You are going to mimic the effects of acid rain on limestone buildings and cliffs • Put a few marble chips into sulphuric (H 2 SO 4) acid. • Collect and test the gas • Note down if there was any fizzing heat • Draw a quick diagram, results and conclusion WHEN FINISHED, from KS 3 Science books, answer questions from p. 62 (start at Q 2), 63 and 64. Put a sub-heading with the book and page numbers.
- Slides: 10