References 1 Casarett Doulls Essentials of Toxicology 2























- Slides: 30

References 1. Casarett & Doull’s: Essentials of Toxicology, 2 nd Ed. , 2010 by Curtis Klaassen and John Watkins III 2. Poisoning and Drug Overdose, 6 th Ed. , 2012 by Kent R. Olson 3. Goldfrank's Toxicologic Emergencies, 10 th Ed. 2014 by Lewis R. Goldfrank 4. Emergency Toxicology, 2 nd Ed. , 1998 by Peter Viccellio

Toxicology (1203562) (2 credit hours) Dr. Khawla Abu Hamour Department of Biopharmaceutics & Clinical Pharmacy University of Jordan

Introduction: TOXICOLOGY IN PERSPECTIVE

DEFINITIONS & TERMINOLOGY • Toxicology: (toxicum) & (logia)…. the Study of Poisons (The science of poisons, including their source, chemical composition, action, tests, and antidotes). • Poisons: are drugs that have almost exclusively harmful effects • However, Paracelsus (1493– 1541) famously stated that “THE DOSE MAKES THE POISON” • a substance that, on ingestion, inhalation, absorption, application, injection, or development within the body, in relatively small amounts, may cause structural or functional disturbance. Called also toxin • biologic origin, ie, synthesized by plants or animals, in contrast to inorganic poisons (lead and iron) • Toxicology: is the branch of pharmacology that deals with the undesirable effects of chemicals on living systems

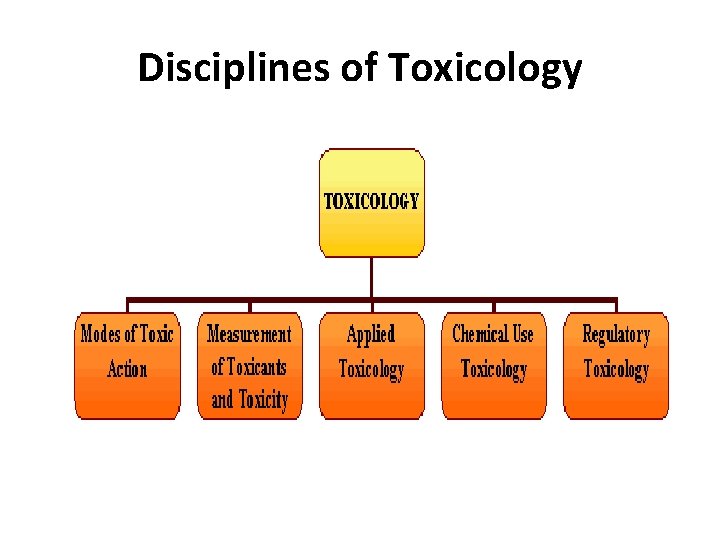
Disciplines of Toxicology

TOXICOLOGY DISCIPLINES Applied Toxicology. q Environmental toxicology: • study the effects of chemicals that are contaminants of food, water, soil, or the atmosphere q Industrial (occupational) toxicology: • Toxic exposure in the work place or during product testing q Clinical (medical) toxicology: focus on the diagnosis, management and prevention of poisoning or ADEs due to medications, occupational and environmental toxins, and biological agents

TOXICOLOGY DISCIPLINES q Veterinary toxicology** q Forensic toxicology: is the use of toxicology to aid medical and legal investigation of death q Nanotoxicology: is the study of the toxicity of nanoparticles (<100 nm diameter). Because of large surface area to volume ratio, (nanomaterials have unique properties compared with their larger counterparts)

What is a Poison? ? • “What is there that is not poison? All things are poison and nothing without poison. Solely, the dose determines that a thing is nota poison” Paracelsus (1493 -1541)

Water Intoxication? q Water poisoning…. fatal disturbance in brain functions when the normal balance of electrolytes in the body is pushed outside of safe limits (e. g. , hyponatremia) by overhydration q Water, just like any other substance, can be considered a poison when over-consumed in a specific period of time q Intravenous LD 50 of distilled water in mouse is 44 ml/kg q Intravenous LD 50 of isotonic saline in mouse is 68 ml/kg

What is a Poison? ? q Poisoning or exposure? ? q Many people consider that poisoning start the moment exposure occurs q In reality, we are exposed to a wide variety of toxic substances each day from food and water that we ingest, and air that we breath q We do not display toxic symptoms, we are not actually poisoned

• Exposure is defined as: • Actual or suspected contact with any substance which has been ingested, inhaled, absorbed, applied to, or injected into the body, regardless of toxicity or clinical manifestation. • Poisonings happen when the exposure results in an adverse health reaction; when a substance interferes with normal body functions after it is swallowed, inhaled, injected, or absorbed.

What is Response? q Change from normal state – could be molecular, cellular, organ, or organism level……the symptoms q The degree and spectra of responses depend upon the dose and the organism ü Immediate vs. Delayed (carcinogenic) ü Reversible vs. Irreversible (liver vs. brain, teratogenic effect) ü Local vs. Systemic ü Graded vs. Quantal……degrees of the same damage vs. all or none q Allergic Reactions & Idiosyncratic Reactions…. ADRs

Dose q The amount of chemical entering the body q This is usually given as: mg of chemical / kg of body weight = mg/kg q The dose is dependent upon: Ø The environmental concentration Ø The exposure pathway Ø The length of exposure Ø The frequency of exposure Ø The properties of the toxicant

Exposure: Pathways q Routes and Sites of Exposure: ü Ingestion (GIT), (first pass effect) § Ex. Lidocaine and Verapamil (antiarrhythmic drugs) ü Inhalation (Lungs): rapid absorption, because of large alveolar surface area ü Dermal/Topical (Skin), absorption varies with area of application and drug formulation, but usually absorption is slower than other routes ü Injection § Intravenous, intramuscular, intraperitoneal q Typical response of Routes and Sites of Exposure: i. v > inhalation > i. p > i. m > oral > topical

Exposure: Duration q Toxicologists usually divide the exposure of experimental animals to chemicals into 4 categories……: Acute < 24 hr Usually 1 exposure Sub-acute 1 month Repeated exposure Sub-chronic 1 -3 months Repeated exposure Chronic > 3 months Repeated exposure q Over time, the amount of chemical in the body can build up, it can redistribute, or it can overcome repair and removal mechanisms q Lethal injection by capital punishment…. .

The other time-related factor that is important in the temporal characterization of repeated exposures is the frequency of exposure Exposure = Intensity x Frequency x Duration Exposure = How much x How often x How long

Dose Response Relationship q The magnitude of drug effect depends on the drug concentration at the receptor site, which is in turn determined by the dose of drug administered and by factors of the drug pharmacokinetic profile q There is a graded dose-response relationship in each individual and a quantal dose-response relationship in a population

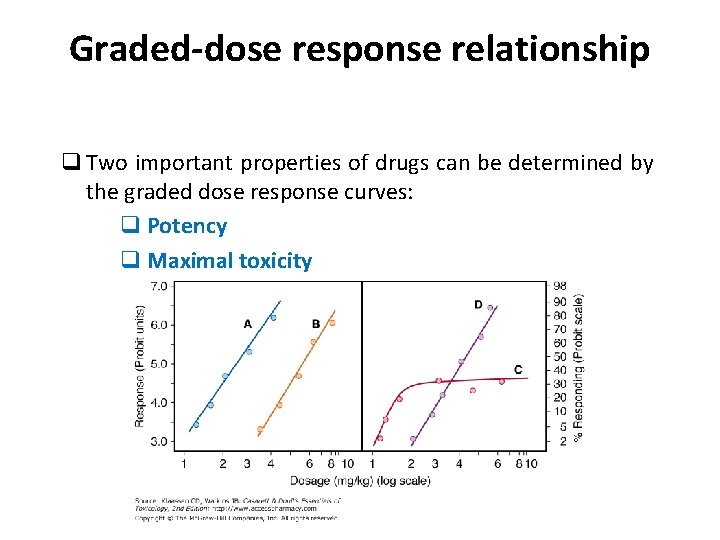
Graded-dose response relationship q The response to a drug is a graded effect, meaning that the measured effect is continuous over a range of doses q Graded dose response curves are constructed by plotting the magnitude of the response against increasing doses of a drug (or log dose)

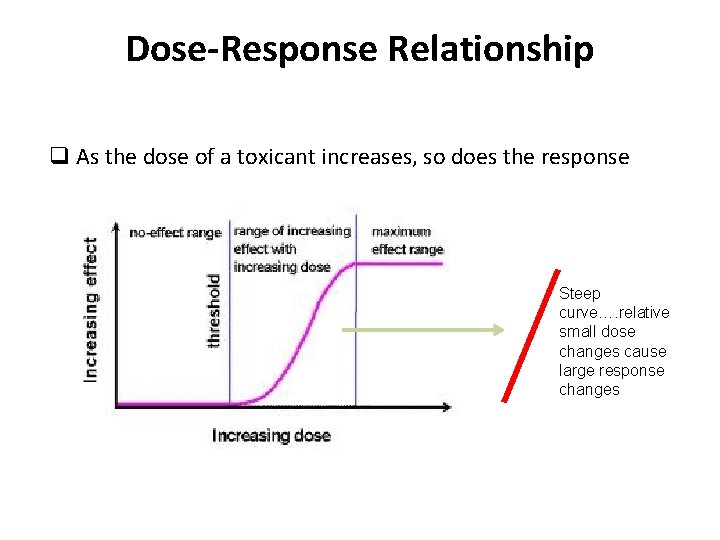
Dose-Response Relationship q As the dose of a toxicant increases, so does the response Steep curve…. relative small dose changes cause large response changes

Graded-dose response relationship q Two important properties of drugs can be determined by the graded dose response curves: q Potency q Maximal toxicity

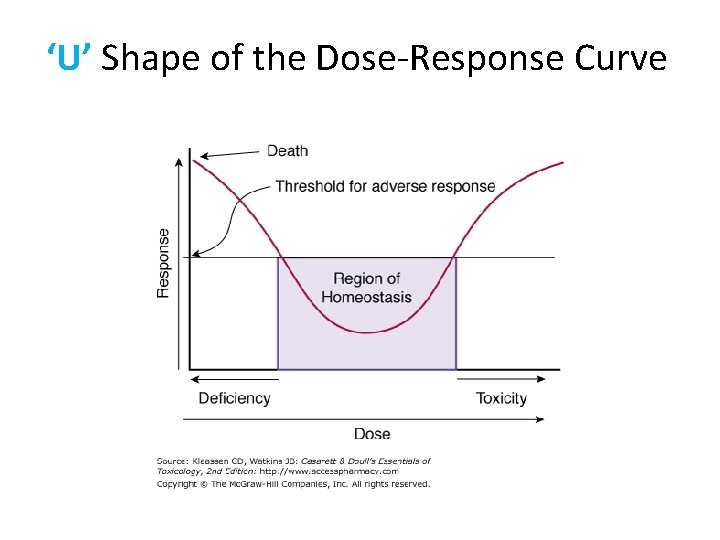
‘U’ Shape of the Dose-Response Curve

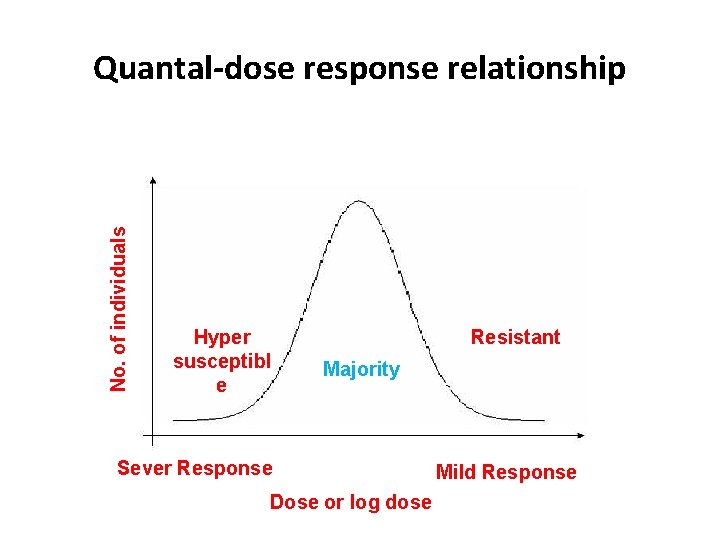
Quantal-dose response relationship q The quantal (all or none) dose-effect curve often characterizes the distribution of responses to different doses in a population of individual organisms q Median toxic dose(TD 50): the dose at which 50% of individuals/population exhibit a particular toxic effect q If the toxic effect is death of the animal, a median lethal dose (LD 50) may be experimentally defined q Median effective dose (ED 50) is the dose that produces a quantal effect (all or nothing) in 50% of the population that takes it

LD 50 q The dose of chemical required to produce death in 50% of the organism exposed to it q LD 50 is not an absolute description of the compound toxicity in all individuals…. . Variations q it’s possible to take more than the lethal dose and live, and take less of the lethal dose and die. q the LDLo (Lethal Dose Low) is the lowest dose known to have resulted in fatality in testing, whilst the LD 100 (Lethal Dose 100%) is the dose at which 100% of the test subjects are killed.

No. of individuals Quantal-dose response relationship Hyper susceptibl e Resistant Majority Sever Response Dose or log dose Mild Response

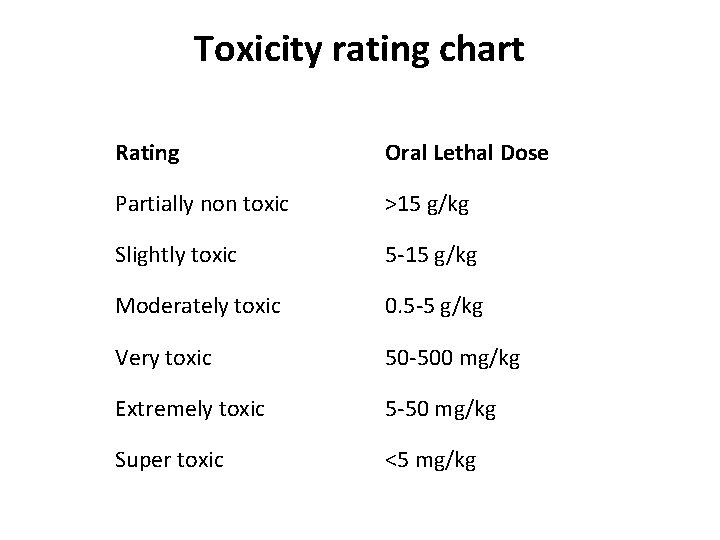
Toxicity rating chart Rating Oral Lethal Dose Partially non toxic >15 g/kg Slightly toxic 5 -15 g/kg Moderately toxic 0. 5 -5 g/kg Very toxic 50 -500 mg/kg Extremely toxic 5 -50 mg/kg Super toxic <5 mg/kg

Assumptions in Deriving the Dose. Response Relationship 1. Confirms that a chemical is responsible for a particular effect 2. Establishes the lowest dose for which an effect occurs – threshold effect (µg’s…. g’s) 3. Individuals vary in their response to a certain dose of xenobiotic 4. The magnitude of the response is related to the dose………!!!!!

!!!……Molecular Target Concept Agonist Antagonist

Toxicodynamics & Kinetics

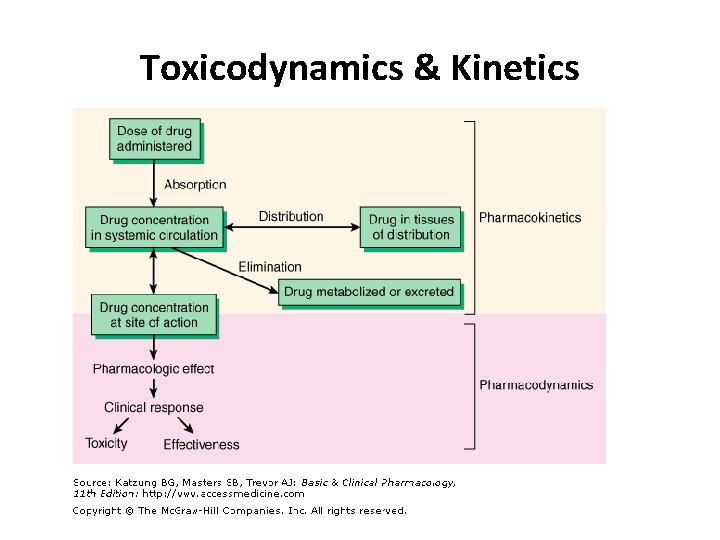
Toxicodynamics refers to the molecular, biochemical, and physiological effects of toxicants or their metabolites in biological systems These effects are result of the interaction of the biologically effective dose of the ultimate (active) form of the toxicant with a molecular target

Toxicokinetics: Disposition (ADME) • Toxicokinetics is the quantitation of the time course of toxicants in the body during the processes of absorption, distribution, biotransformation, and excretion or clearance of toxicants • In other words, toxicokinetics is a reflection of how the body handles toxicants as indicated by the plasma concentration of that xenobiotic at various time points • The end result of these toxicokinetic processes is a biologically toxic concentration of the toxicant/s